Lens-Related Glaucomas
Efrem Mandelcorn
Neeru Gupta
Lens-related glaucomas, although rare, are important to consider in the evaluation of patients with elevated intraocular pressure. These are a diverse group of disorders with distinctly different management approaches depending on the diagnosis. Thus, it is essential to accurately identify the mechanisms leading to uncontrolled intraocular pressure. As they may develop by open- or closed-angle mechanisms (Table 54.1), careful clinical examination of the presenting patient will help to diagnose the type of lens-related glaucoma. Additional tests as part of the clinical workup may also be needed, including ultrasound biomicroscopy. In the case of lens-induced glaucoma with inflammation from phacolytic, phacoanaphylactic, or infectious causes, differentiating these conditions may be challenging 1,2, and so anterior chamber fluid or tissue evaluation for histology, cytology or microbiology may be helpful.
TABLE 1. Lens-Related Glaucomas | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
In the presence of an open angle, the trabecular meshwork can become blocked by lens proteins (phacolytic), lens material itself (phacotoxic or lens particle), inflammatory material secondary to an abnormal immune response to host lens proteins (phacoanaphylactic), or retained viscoelastic material in the early post-operative period. Lens-induced angle-closure glaucoma examples include phacomorphic glaucoma, ectopia lentis, and spherophakia, which may give rise to either pupillary block or vitreo-lenticular block due to the abnormal lens position. In the post-operative setting, anterior chamber intraocular lens implant without an adequate iridectomy can lead to angle-closure glaucoma. The possibility of combined mechanisms of glaucoma with a lens related component should also be considered.
Phacolytic Glaucoma
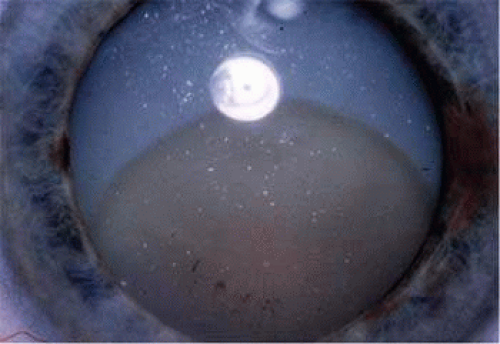
Phacolytic glaucoma is an inflammatory type of open-angle glaucoma in the presence of a mature or hypermature cataract. Patients present with acute onset of pain and redness, often with a history of poor vision due to cataract. The key finding on clinical examination is a mature or hypermature Morgagnian cataract 3,4 (Fig. 54.1) in the setting of high pressure. The angle is open and patients often have some anterior chamber reaction in the form of dense flare with mild to moderate cells (Fig. 54.2). This is different from phacoanaphylactic glaucoma in which keratic precipitates and a more marked inflammatory response with posterior iris synechiae are characteristic.
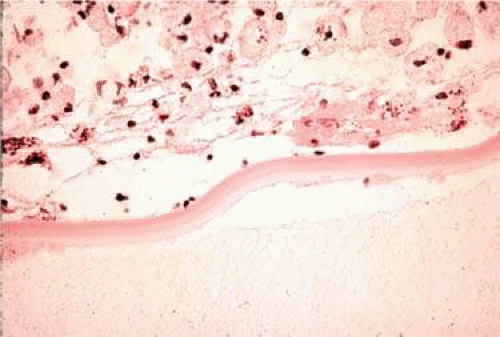
The term phacolytic, means lens destruction, due to macrophage breakdown or destruction of lens material. In phacolytic glaucoma, macrophages phagocytose lens proteins and these foamy macrophages have been thought to be the cause of obstructing the trabecular meshwork leading to decreased outflow and increased intraocular pressure.3,5 To obtain a specimen, an anterior chamber paracentesis can be performed using topical anesthetic, a lid speculum, and a fine needle on a 1-cc syringe at the slit-lamp. The specimen should be sent to pathology to look for foamy macrophages containing degenerated lens material that stains a pink color with PAS stain (Fig. 54.3). Detection of foamy macrophages under the microscope confirms the diagnosis, although their absence does not exclude the diagnosis.
In the presence of a mature or hypermature cataract, lens cortex liquefies and can leak out through the anterior capsule into the anterior chamber. These lens particles are able to exit an intact capsular bag in the form of soluble high-molecular-weight (HMW) proteins. Phacolytic glaucoma may also develop after traumatic or spontaneous rupture of the capsule.6 These HMW proteins are engulfed by macrophages and block the aqueous outflow pathways within the trabecular meshwork.3,7 There is evidence that outflow pathways may be obstructed directly by high-molecular-weight lens proteins rather than by lens-laden macrophages alone.8,9
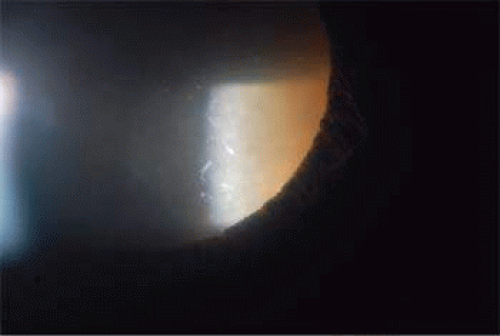
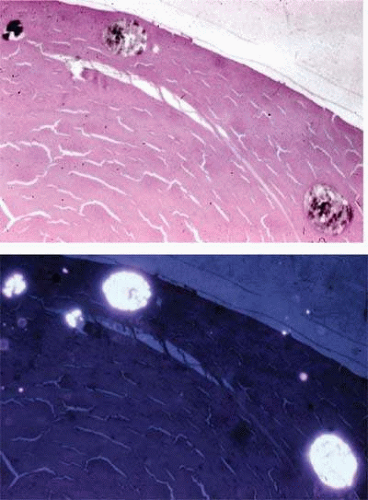
As the cortical material seeps out of the capsular bag, the lens loses volume and the capsular bag deflates, leading to the clinical finding of a wrinkled anterior capsule, and in some cases, iridescent or hyper-refringent crystals can be seen on the capsular bag surface (Fig. 54.4). These refractile particles that are being shed by the cataractous lens are composed of cholesterol or calcium oxalate (Fig. 54.5).10,11,12 The location of these refractile particles often points to the point of leakage through the capsular bag. Cortical white flocculent lens material may also leak out through an intact posterior capsule into the vitreous12,13 or may be found adherent to the lens capsule and this finding is also considered to be of diagnostic importance.14
Definitive management of phacolytic glaucoma is cataract extraction to remove the source of the lens proteins causing obstruction. Management also includes lowering intraocular pressure with topical and/or oral medications as needed. A topical steroid is also helpful in reducing inflammation and improving visibility to facilitate cataract removal. Poor visualization due to dense lens and hazy cornea and anterior chamber is a potential cause of surgical complications in this condition. A capsulorrhexis may be more easily completed once visualization of the anterior capsule has been enhanced with the use of trypan blue, a vital dye. The posterior capsule is often weak in cases of hypermature cataract, and the surgeon should be ready to perform an anterior vitrectomy to remove any residual lens material that might leak through a dehiscence in the posterior capsule into the vitreous cavity. For patients with poorly controlled preoperative intraocular pressure, cataract extraction can be combined with trabeculectomy.15
Lens Particle Glaucoma
Lens-particle glaucoma, also known as phacotoxic glaucoma, involves disruption of the anterior lens capsule during cataract surgery or following capsulotomy or ocular trauma.7,14 There are reports of lens-particle glaucoma secondary to liberation of lens cortical material from capsular remnants following intraocular lens dislocation.16 Lens material, either sequestered within the capsular bag or in other areas of anterior and posterior segments, along with secondary inflammatory debris, can block the aqueous outflow channels leading to open-angle glaucoma. Large lens pieces may also cause phacotoxic glaucoma as they break into smaller pieces that obstruct trabecular outflow. This is in contrast to phacolytic glaucoma in which HMW proteins, not lens cortical particles, block the trabecular meshwork.
Lens-particle glaucoma most often occurs within the first few weeks after the anterior capsule is compromised. There are, however, cases that present years after extracapsular cataract extraction after all residual soluble proteins have been reabsorbed lending to the theory that insoluble lens particles are responsible for outflow obstruction. The observation that anterior chamber washout to remove lens debris from the angle helps to control glaucoma supports this theory.
The degree of intraocular pressure elevation and inflammation varies depending on the amount of lens material liberated into the anterior chamber.17 In addition, individual patient factors, including outflow facility and immunologic variations may also explain why some patients tolerate retained lens cortex better than others and why the amount of cell, flare, and corneal edema varies so much among patients with this condition. Lens debris may be free floating in the anterior chamber or vitreous, may settle on the corneal endothelium, or may layer out in the anterior chamber and form a pseudohypopyon.
Management of lens particle glaucoma includes the use of corticosteroids and intraocular pressure lowering agents while trabecular meshwork cells work to clear residual lens particles. In cases of large amounts of lens material, persistent inflammation or elevated intraocular pressure, surgical removal of the lens fragments is indicated.
Retained Intravitreal Lens Fragments After Cataract Surgery
During cataract surgery, intraoperative loss of lens material into the vitreous cavity after posterior capsular break may lead to acute secondary open angle glaucoma in the early postoperative period.18,19,20,21,22,23 The anterior segment surgeon should be cautious whenever a break occurs in the posterior capsule, avoiding aggressive intraoperative maneuvers to remove posterior segment lens material to avoid complications such as retinal injury.24 Primary insertion of a posterior chamber intraocular lens during cataract surgery even in the presence of loss of cataract material into the vitreous may improve the outcome.25 Vitrectomy performed by a vitreoretinal surgeon offers the greatest chance for successful removal of the lens material and visual recovery. At the time of the surgery, any noted retinal tears or detachment can also be addressed.
Intravitreal lens cortex can be left alone without intervention, if they are small and consist of cortical material. Larger fragments of cortex, even small pieces of nucleus, usually require lensectomy by pars plana approach. Even if these particles are removed by vitrectomy and lensectomy, these patients may still experience long-term visual morbidity in the form of chronic uveitis with cystoid macular edema, chronic corneal edema, and late retinal detachment.26,27 There is some evidence that early pars plana lensectomy leads to a lower incidence of chronic glaucoma19 although other studies do not show any strong relationship between timing of vitrectomy surgery and long term glaucoma.21,22
Glaucoma Associated with Phacoanaphylactic Uveitis
Phacoanaphylactic glaucoma, also known as phacoanaphylactic endophthalmitis28 or phacoantigenic uveitis29 is the result of granulomatous reaction to retained lens material following lens capsule disruption, either from surgery or trauma.29,30 It may also occur in cases of retained lens material in the vitreous cavity. The term phacoanaphylactic is a misnomer since it is a granulomatous disease without an anaphylactic component. The onset of the granulomatous reaction is delayed so that patients often present with minimal pain that is slow to worsen. This is in contrast to the acute presentation of pain in phacolytic glaucoma. Phacoantigenic uveitis presents with signs of granulomatous uveitis, including mutton-fat keratic precipitates (KP), which also distinguishes it from phacolytic glaucoma where keratic precipitations are absent. Other clinical findings include synechiae as well as hypopyon. In contrast to the intact capsule in phacolytic glaucoma, the anterior lens capsule in phacoantigenic uveitis is classically described as ruptured, which gives the immune system access to lens material antigens that provoke a granulomatous inflammatory response. There are, however, clinical and histopathologic cases of phacoantigenic uveitis with no evidence of trauma or penetrating injury to the lens capsule.1,31,32
In contrast to phacolytic glaucoma, phacoanaphylactic uveitis is more commonly associated with hypotony (secondary to ciliary body shutdown) than increased intraocular pressure. In one study, hypotony was found in 58% of cases and glaucoma was diagnosed in only 17%.31 Elevation of intraocular pressure in phacoantigenic glaucoma can occur through a number of different mechanisms. Obstruction of outflow channels by inflammatory debris may lead to secondary open-angle glaucoma.33 Pupillary block from posterior synechiae may lead to angle-closure glaucoma. An anterior pull mechanism of secondary angle closure glaucoma may be the result of peripheral anterior synechiae from inflammatory or neovascular membranes. From chronic inflammation and glaucoma, these cases may progress to phthisis bulbi.
In enucleation specimens, phacoanaphylactic uveitis can be distinguished by zonal type of granulomatous reaction centered around residual lens material.34,35 The zone adjacent to the lens, zone 1, is composed of polymorphonuclear cells infiltrating the lens substance. In zone 2, epithelioid and multinucleated giant cells surround zone 1 and this makes up the granulomatous zone. Surrounding this is zone 3, a more chronic type of inflammatory component comprised of lymphocytes, plasma cells and granulation tissue.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree