first. Because they are smaller, they move more on the eye. The lids sense this movement initially and gradually adapt, typically resulting in comfortable lens wear.” The use of the term gas permeable as compared to the previous rigid gas permeable terminology was recently proposed by the Contact Lens Manufacturers Association (CLMA) to minimize the focus on “rigid” lenses.
at inhibiting prostaglandin synthesis appears to be Voltaren. A recommended NSAID dosage for GP lens adaptation is as follows10:
Instill one drop of Voltaren in each eye 30 minutes and then 15 minutes before lens insertion.
Instill a third drop just before lens insertion.
A fourth drop can be instilled 1 hour after insertion.
This regimen can be maintained for 3 to 5 days or until adaptation is completed.
practitioners the opportunity to evaluate the lens-to-cornea fitting relationship and to make the changes necessary to obtain a good fit and provide acceptable vision to the patient. It is also apparent that the fitting visit provides patients with an opportunity to become familiar with their particular lenses. Finally, such factors as lens centration and residual astigmatism can be evaluated.18 The primary limitations pertain to (likely) not leaving the office with lenses, as compared with most soft lens patients, and the fact that, in most cases, satisfactory vision will not be obtained with the first pair of GP lenses applied. The 6 D myopic patient who is fitted with 3 D power diagnostic GP lenses will not only experience initial awareness, but also blurred vision. Nevertheless, with most special design GP lenses, including bifocal, keratoconic, and postsurgical designs, it is important to use diagnostic fitting sets because of the greater challenges involved in the fitting process and the more custom nature of the designs. When diagnostic fitting sets are to be used, it is important that the lenses are in the same design and material as the lenses to be ordered. A comparison of the factors involved in deciding between empirical and diagnostic fitting is provided in Table 5.1.
appropriate center thicknesses; these parameters—and especially edge shape—should be verified upon receiving them from the laboratory. With the increasing popularity of ultrathin designs, a diagnostic set of this design in a minus power (i.e., −3.00 D) would be recommended as well.
TABLE 5.1 DIAGNOSTIC FITTING VERSUS EMPIRICAL FITTING | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
TABLE 5.2 RECOMMENDED PARAMETERS FOR A 20-LENS DIAGNOSTIC SET, LOW-AND HIGH-DK GAS-PERMEABLE MATERIALS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 5.3 PARAMETERS FOR GAS-PERMEABLE INVENTORY LENS SET | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Sterility.
Slightly alkaline in pH, which assists in fluorescence.
Reduced risk of burning and stinging caused by pH.
Less viscous than use of a wetting solution (which may result in an abnormally thick tear layer).
Inexpensive.
Easy to use.
Overall field of view and ability to directly compare fluorescein pattern of both eyes simultaneously.
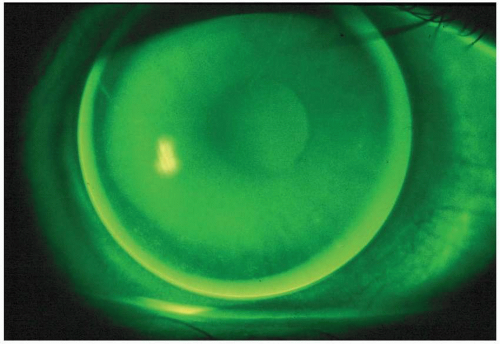
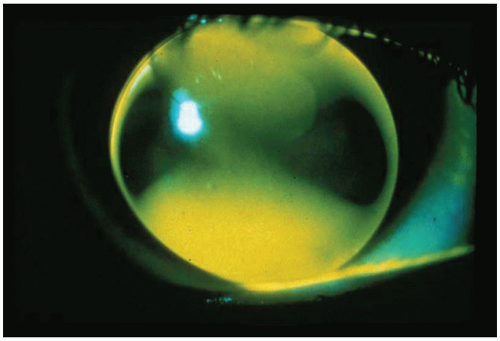
with the instillation of fluorescein (Fig. 5.5). Excessive apical bearing can potentially result in corneal molding with resultant distortion or warpage. In addition, the gradual formation of a central corneal abrasion is also possible.
Corneal topography. This varies between patients; for example, a patient with a small corneal cap (defined as the region within 0.50 D of the corneal apex) will exhibit a somewhat steeper fluorescein pattern than a patient with a larger-than-average cap.29,30
Selection of a steep BCR may result in poor tear exchange and a misleading small amount of fluorescein centrally.
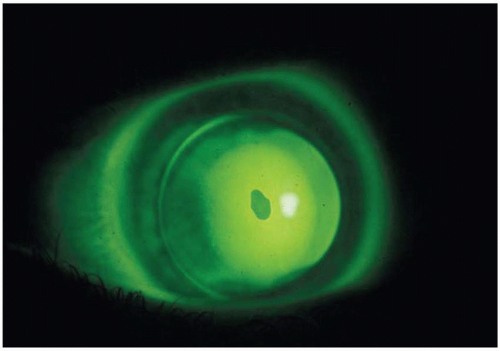
If the peripheral curve is too steep, peripheral seal-off can occur and the fluorescein pattern can exhibit apical clearance.
In certain individuals—particularly dry-eye patients—the fluorescein will dissipate quickly and may create a “pseudoapical flat” relationship; therefore, the pattern should be evaluated immediately after fluorescein instillation.
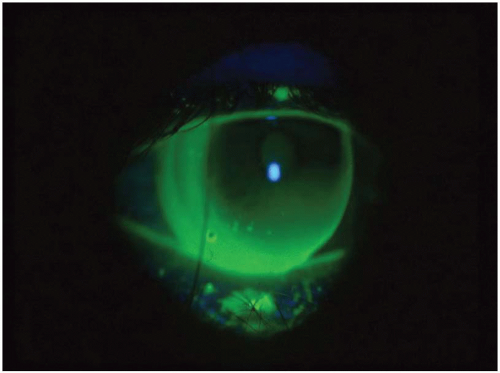
A “pseudosteep” pattern has been reported in high minus fluoro-silicone/acrylate (F-S/A) lenses.31 Apparently the edge thickness blocks the fluorescence, giving an appearance of central pooling. Likewise, one would expect that a high plus lens may demonstrate a flatter than actual base curve fitting relationship because the thick center would attenuate the light more.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree