Purpose
To identify genetic mutations and study the corneal epithelium in Japanese patients with Meesmann corneal dystrophy.
Design
Laboratory investigation and prospective observational case series.
Methods
Slit-lamp biomicroscopy with fluorescein vital staining and in vivo confocal microscopy were performed. Mutation screening of the KRT3 and KRT12 genes was performed via polymerase chain reaction and direct sequencing for 5 patients in 2 families.
Results
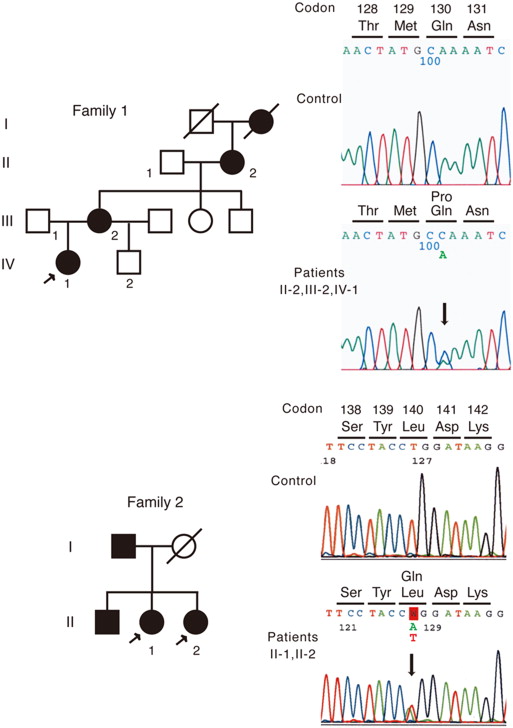
Slit-lamp biomicroscopy revealed multiple corneal intraepithelial microcysts in all patients. A clear zone was seen in the younger generation, whereas mild subepithelial opacity was seen in the older generation. In the in vivo confocal microscopy, numerous corneal intraepithelial microcysts and hyperreflective materials, which were believed to be degenerative cells, were detected closer to the basal layer of the corneal epithelium in older patients. The superficial layer contained more enlarged microcysts, and the hyperreflective materials showed atrophic changes, as compared to the basal layer. The demarcation line between the microcysts and normal epithelial cells was clearly visualized by in vivo confocal microscopy and corresponded to the demarcation line of the clear zone observed by the slit-lamp examination. Two heterozygous mutations (Q130P, L140Q) in the KRT12 gene, one of which (L140Q) was novel, were identified only in the affected patients of the families.
Conclusions
We identified a novel missense mutation of the KRT12 gene in Meesmann corneal dystrophy. The in vivo confocal microscopy examinations revealed previously unreported depth-dependent ultrastructural changes in the living cornea of Meesmann corneal dystrophy patients.
Meesmann corneal dystrophy is a rare dominantly inherited corneal disease with myriad microcysts in the corneal epithelium. Since the microcysts move to the surface and form corneal erosions, it is not unusual for Meesmann corneal dystrophy to be misdiagnosed and treated as dry eye syndrome. Patients with Meesmann corneal dystrophy report foreign body sensations, dry eye symptoms, and photophobia; however, some patients have no symptoms. In Meesmann corneal dystrophy, the corneal stromal or endothelial cells are not usually damaged.
In 1997, genetic analyses revealed that mutations in the cornea-specific keratin 3 ( KRT3 ) and keratin 12 ( KRT12 ) genes are responsible for Meesmann corneal dystrophy. To date, 3 mutations in the KRT3 gene and 20 mutations in the KRT12 gene have been reported. Six mutations (R135I, R135G, A137P, L140R, Y429D, and L433R) in the KRT12 gene have been identified exclusively in Japanese patients with Meesmann corneal dystrophy.
Laser scanning in vivo confocal microscopy is a noninvasive imaging tool for investigating microstructural changes in corneal disorders. A few studies have used in vivo confocal microscopy to investigate the microstructure of the corneal epithelium in patients with Meesmann corneal dystrophy. In the current study, we report clinical features, including in vivo confocal microscopy findings, in 5 patients with Meesmann corneal dystrophy from 2 Japanese families and identify a novel KRT12 mutation (L140Q) in 1 of the families.
Methods
Clinical Studies
This prospective study was approved by the Institutional Review Board of The Jikei University School of Medicine and Tokyo Dental College. The protocol adhered to the tenets of the Declaration of Helsinki, and informed consent was obtained from all participants before entry into this study.
Two Japanese families (Family 1 at The Jikei University and Family 2 at Tokyo Dental College) were diagnosed with Meesmann corneal dystrophy ( Figure 1 ). Patients from 3 generations (a 16-year-old female proband, IV-1; the 53-year-old mother of the proband, III-2; and the 81-year-old maternal grandmother of the proband, II-2) were studied in Family 1 ( Figure 1 , Top). Two affected sisters (a 63-year-old woman, II-1; and a 62-year-old woman, II-2) were studied in Family 2 ( Figure 1 , Bottom). All patients were examined by slit-lamp biomicroscopy including fluorescein dye vital staining and laser scanning in vivo confocal microscopy (Heidelberg Retina Tomograph II Rostock Cornea Module; Heidelberg Engineering, Heidelberg, Germany).
Molecular Genetic Studies
Genomic DNA was extracted from venous blood samples using a Gentra Puregene Blood kit (Qiagen, Tokyo, Japan), and family members including all 5 patients underwent molecular analysis. All coding exons of the KRT3 and KRT12 genes were amplified via polymerase chain reaction (PCR) with previously reported primers, all of which were produced by Operon Biotechnologies (Tokyo, Japan). The PCR products were purified with a QIAquick PCR Purification kit (Qiagen) and used as the template for sequencing. Both strands were sequenced on an automated sequencer (3730 xl DNA Analyzer; Applied Biosystems, Foster City, California, USA) using a BigDye Terminator kit V3.1 (Applied Biosystems).
Results
Clinical Findings
Family 1
Patient IV-1 : The proband experienced intermittent photophobia and foreign body sensations. The decimal best-corrected visual acuity (BCVA) was 1.2 in both eyes. Intraocular pressure was within the normal range in each eye. Findings of lens and fundus examination were unremarkable. When the proband was 9 years old, myriad microcysts were detected in the corneal epithelium of her right ( Figure 2 , Row 1, left) and left eyes ( Figure 2 , Row 1, right). Fluorescein punctate staining was positive in both eyes. No gray lines were observed in the corneal epithelium. Both eyes contained irregularly shaped clear areas without microcysts ( Figure 2 , Row 1). At the age of 16 years, the clear zones in the patient’s right ( Figure 2 , Row 2, left) and left eyes ( Figure 2 , Row 2, right) had changed in shape.
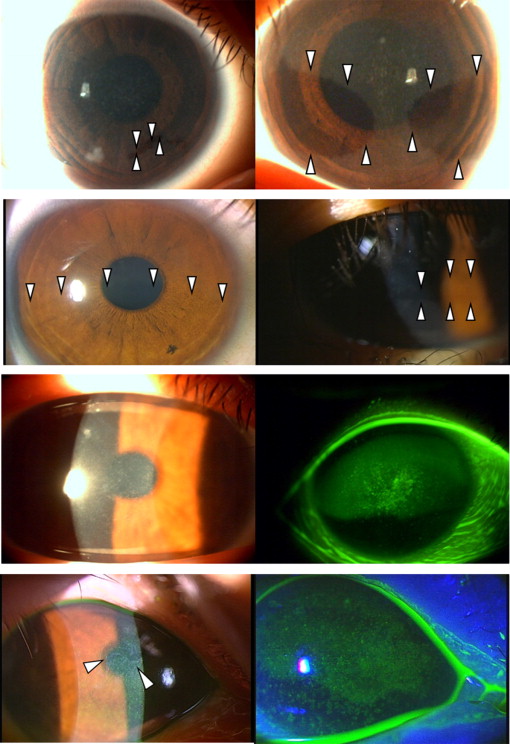
Patient III-2 : The patient experienced intermittent photophobia and foreign body sensations. The BCVA was 0.6 in the right eye and 1.0 in the left eye. Intraocular pressure was within the normal range in each eye. Findings of lens and fundus examination were unremarkable. Myriad microcysts with positive fluorescein punctate staining were observed in the right ( Figure 2 , Row 3) and left eyes. No clear zones or gray lines were observed in the corneal epithelium.
Patient II-2 : The patient experienced intermittent photophobia and foreign body sensations. The BCVA was 0.3 in both eyes. Senile cataracts were observed, but fundus examination was unremarkable in both eyes. Intraocular pressure was within the normal range in each eye. Myriad microcysts with positive fluorescein punctate staining and mild subepithelial opacities were observed in the right ( Figure 2 , Row 4) and left eyes. No clear zones or gray lines were observed in the corneal epithelium.
Family 2
Patient II-1 : The patient experienced foreign body sensations. The BCVA was 0.3 (−14.0 diopters [D]) in the right eye and 0.3 (−8.00 D/−3.00 D × 170 degrees) in the left eye. She had cataracts and myopic retinal change in both eyes. Intraocular pressure was within the normal range in each eye. Myriad microcysts in the corneal epithelium and mild subepithelial opacities were detected in the right ( Figure 3 , Top left and Top center) and left eyes. Punctate fluorescein staining of the cornea was present ( Figure 3 , Top right). A clear zone or gray line was not observed.
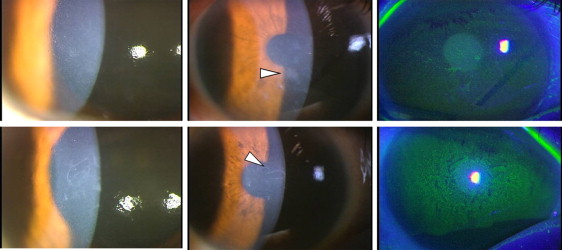
Patient II-2 : The patient experienced foreign body sensations. The BCVA was 0.4 (+0.50 D) in the right eye and 0.5 (+1.50 D/−1.00 D × 105 degrees) in the left eye. She had cataracts in both eyes. Fundus examination was unremarkable. Intraocular pressure was within the normal range in each eye. Myriad microcysts, mild subepithelial opacities, and punctate fluorescein staining were detected in the right ( Figure 3 , Bottom) and the left eyes. A clear zone or gray line was not observed.
In Vivo Confocal Microscopic Findings
The summary of the in vivo confocal microscopic findings is shown in the Table .
| Patient | Age/Sex | Deepest Location of Hyperreflective Materials | High-Density Objects | Clear Zone | Low-Density Spaces | |
|---|---|---|---|---|---|---|
| Family 1 | IV-1 | 16/Female | 28 μm | – | + | + |
| III-2 | 53/Female | 48 μm | – | – | + | |
| II-2 | 81/Female | 57 μm | + | – | + | |
| Family 2 | II-1 | 63/Female | 57 μm | + | – | + |
| II-2 | 62/Female | 42 μm | + | – | + |
Family 1
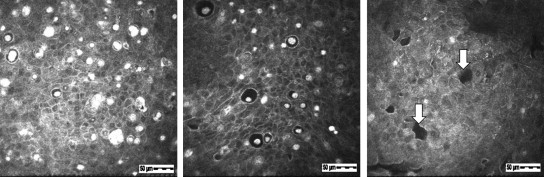
Patient IV-1 : The in vivo confocal microscopy images in the right eye showed intraepithelial microcysts ( Figure 4 , Row 1). The majority of microcysts were round, and some were oval ( Figure 4 , Row 1, left). Most microcysts were located between the depths of 7 μm and 20 μm from the corneal surface. The size of the microcysts, which had well-defined edges, ranged from 10-40 μm in diameter. Enlarged microcysts more than 60 μm in diameter were rarely seen ( Figure 4 , Row 1, center). Most microcyst edges had high intensity, but some edges had low intensity ( Figure 4 , Row 1, left). The inside of the microcysts showed low intensity, and most microcysts contained hyperreflective materials ( Figure 4 , Row 1, left). The hyperreflective materials in the microcysts were large and occupied most of the microcyst ( Figure 4 , Row 1, left), but some were small ( Figure 4 , Row 1, left) or plural ( Figure 4 , Row 1, center). Many independent hyperreflective materials without microcysts were also visible ( Figure 4 , Row 1, center). These independent hyperreflective materials were round or irregularly shaped ( Figure 4 , Row 1, center) and were relatively larger than the hyperreflective materials within microcysts ( Figure 4 , Row 1, center). High-intensity and indistinct regions were sometimes seen around the concentrated microcyst assembly ( Figure 4 , Row 1, right). An area in which microcysts suddenly disappeared at the same depth was detected only in this patient, and the boundary with the normal epithelium was clearly observed ( Figure 4 , Row 2, left). The boundary line corresponded to the clear zone border. The epithelial cells in the area without microcysts were the same as epithelial cells of a normal individual. In the deeper layers (closer to the basal layer), the number of microcysts decreased. The deepest individual hyperreflective materials were confirmed at a depth of 28 μm. The findings of the left eye were the same as those of the right eye.
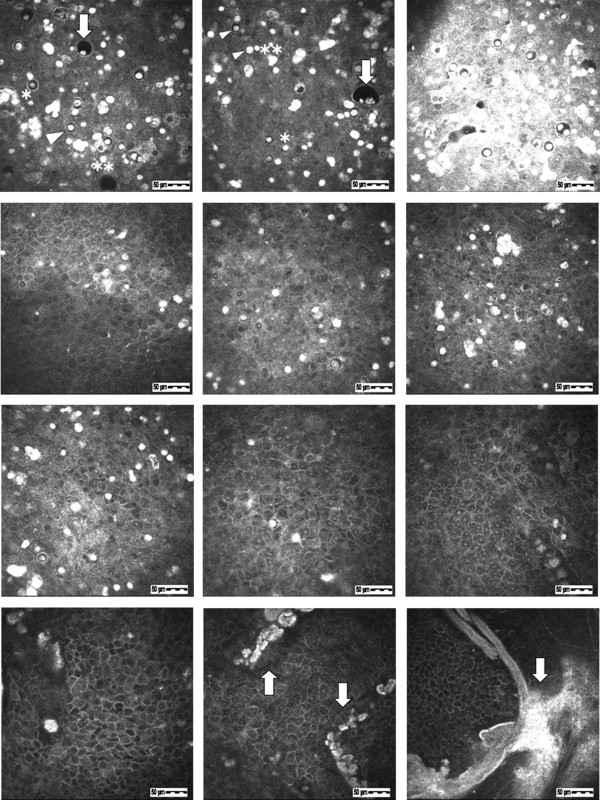
Patient III-2 : The images in the right eye showed intraepithelial microcysts at a depth of 16 μm from the corneal surface ( Figure 4 , Row 2, center). Characteristic findings of the microcysts and hyperreflective materials were very similar to those in Patient IV-1. In the deeper layers, the number of microcysts and hyperreflective materials decreased. The deepest individual hyperreflective materials were confirmed at a depth of 48 μm. The findings of the left eye were the same as those of the right eye.
Patient II-2 : The images in the right eye showed intraepithelial microcysts ( Figure 4 , Row 2, right). In details, the images showed microcysts at depths of 12 μm ( Figure 4 , Row 3, left), 29 μm ( Figure 4 , Row 3, center), 40 μm ( Figure 4 , Row 3, right), and 57 μm ( Figure 4 , Row 4, left) from the corneal surface. The individual hyperreflective materials and microcysts containing hyperreflective materials were mixed in the anterior layer ( Figure 4 , Row 3, left). In the deeper layers, large microcysts disappeared, and the number of microcysts decreased. The ratio of the hyperreflective materials to microcysts increased in the deeper layers ( Figure 4 , Row 3, center). In the area deepest to the basal layer, only individual hyperreflective materials were detected ( Figure 4 , Row 3, right). The deepest individual hyperreflective materials were confirmed at a depth of 57 μm ( Figure 4 , Row 4, left). The epithelial cells surrounding microcysts or individual hyperreflective materials were normal at all depths. High-density objects were seen at depths of 40-80 μm in the epithelium near the Bowman layer ( Figure 4 , Row 4, center and right). The objects overlapped with each other and appeared as arc-shaped objects larger than 400 μm ( Figure 4 , Row 4, right). The findings of the left eye were the same as those of the right eye. As the patient age increased, the lesions were confirmed at deeper layers.
Family 2
Patient II-1 : The images in the right eye showed intraepithelial microcysts and hyperreflective materials at a depth of 12 μm from the corneal surface ( Figure 5 , Left). Characteristic findings were very similar to those in Patient II-2 from Family 1.