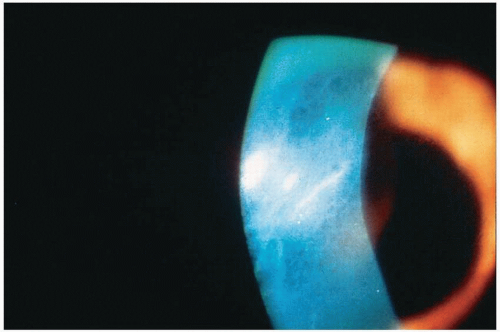
Thinning of the corneal stroma
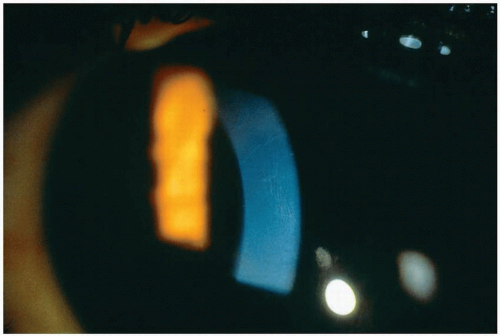
Breaks in the Bowman layer
Iron deposition in the basal layers of the corneal epithelium
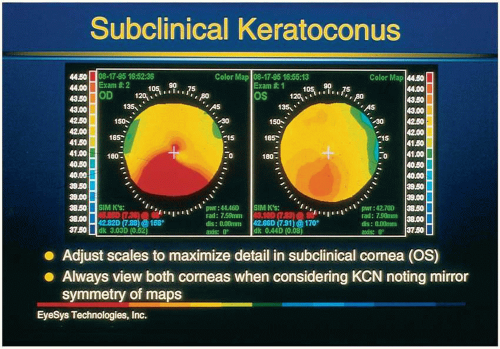
for refractive surgery and it has been found that as many as 5% to 7% of refractive surgery candidates have subclinical keratoconus.58,59
 ▪ FIGURE 18.2 A nipple cone is shown in (A), an oval or sagging cone in (B), and a globus or globoid cone in (C). |
TABLE 18.1 CONE TYPES AND PREVALENCE | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||
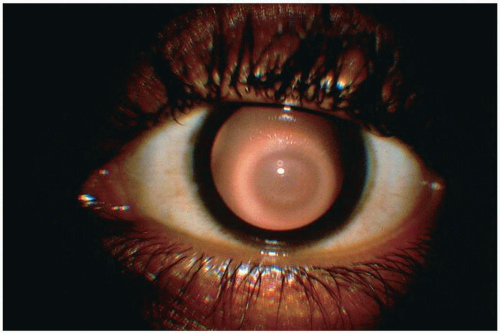
globe through the upper lid.64 The Fleischer ring can be observed in approximately 50% of all diagnosed cases of keratoconus.65 It is a yellow-brown to olive-green discoloration appearing in a broken or interrupted ring encircling the base of the cone (Fig. 18.4). It appears to outline the base of the cone and represents hemosiderin deposits in the deep epithelium near the Bowman membrane. Irregular superficial scars can form at the apex of the cone as the condition progresses. They begin as discrete dots in the Bowman membrane; fibrillar connective tissue invades the space between the opacities, and they proceed to increase and become opaque66,67 (Fig. 18.5).
technique called photodiagnosis has been useful in diagnosing and monitoring the size, shape, and location of advanced or severe cones. In this technique, the image of the cone is viewed against the red fundus reflex. With the dilated pupil, the examiner views the cornea through the direct ophthalmoscope at a distance of 2 feet. In advanced cases (>50 D), the cone can be easily observed against the red fundus reflex (Fig. 18.6). The image can also be recorded using a fundus camera with a high plus condensing lens.
in both conditions, CWS patients typically manifest signs of corneal hypoxia while rarely exhibiting corneal steepening beyond 50 D.69 In addition, the degree of mire irregularity or misalignment is typically less with CWS than in keratoconus. Videokeratoscopy will show a localized area of corneal steepening in keratoconus but not necessarily in CWS. Keratoconus can often be differentiated with a comprehensive biomicroscopy examination. Clinical signs such as corneal thinning, Fleischer ring, Vogt striae, and, in moderate to severe cases, corneal scarring are present in keratoconus and not associated with CWS, which is limited to changes in corneal contour.70 The affected corneal region is limited in keratoconus, often of variable size, encompassing some of (but not limited to) the central and inferior regions. This can be verified via photodiagnosis. Keratoconus, unlike CWS, is a progressive condition, typically over a 5- to 10-year period, often resulting in a protruding conical region, easily observable via Munson sign in advanced stages. Corneal warpage syndrome, conversely, is remediated and often results in a healthy, regular cornea via discontinuation of contact lens wear and/or refitting into a lens material with higher oxygen permeability (Dk) exhibiting a well-centered lens-to-cornea fitting relationship. The differences in keratoconus and corneal warpage syndrome are summarized in Table 18.3.
TABLE 18.2 KERATOCONUS CLINICAL SIGNS | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||
TABLE 18.3 CORNEAL WARPAGE SYNDROME VERSUS KERATOCONUS: DIFFERENTIAL DIAGNOSIS | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||
TABLE 18.4 KERATOCONUS CLASSIFICATION | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||
to them or their specific disease state. One excellent source is the National Keratoconus Foundation site available online at http://www.nkcf.org. Keratoconus patients can contact a large number of sources of information here and find answers to commonly asked questions. Likewise, practitioners can utilize this information as well. Additionally, several cities have keratoconus support groups in an effort to allow patients the opportunity to share their experiences with others.
TABLE 18.5 KERATOCONUS SEVERITY SCORE | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree