Intrastromal Corneal Implants
Jonathan H. Talamo
Mahnaz Nouri
Azhar N. Rana
HISTORY AND BACKGROUND
Additive keratorefractive surgery has undergone many changes since the early efforts of Barraquer (1), Stone and Herbert (2), Belau et al. (3), and Choyce (4) in the 1940s, 1950s, and 1960s. As technology and understanding have progressed, a variety of materials have been evaluated for implantation within the corneal stroma to correct refractive errors, and have included synthetic materials (2, 3, 4) as well as autologous donor tissue (1). New refractive techniques are being developed for the treatment of corneal ectasia and presbyopia with corneal implants. This chapter addresses the use of intrastromal implants for myopia, hyperopia, and corneal ectatic conditions.
Synthetic Materials
Belau (3) studied the use of several materials for implantation within corneal stroma, experimenting with polysulfone, polymethymethacrylate (PMMA), polyprolene, and silicone oil. Polysulfone is a thermoplastic compound first synthesized and for use as a corneal inlay (4,5). Due to the very high refractive index of polysulfone, thin lenses can be made to allow correction of large refractive errors. Unfortunately, clinical trials of polysulfone implants resulted in corneal haze and degenerative changes with thinning and necrosis of the overlying corneal stroma and epithelium (6,7). Years later, hydrogel (hydroxyethyl methacrylate) lenses were developed (8). Hydrogel lenses are permeable to water, oxygen, glucose, and other metabolites. Although hydrogel is well tolerated by the human cornea, early refractive results were not very predictable (9,10). Revised models of these inlays are currently in clinical trials.
Animal studies showed that use of impermeable corneal implants such as PMMA blocked the flow of metabolites and water, thus causing thinning and necrosis of corneal stroma (11). It was later discovered that reducing the diameter and increasing the depth of placement for impermeable inlays would improve the diffusion of metabolites to the adjacent corneal tissue. Thus, the diameter and depth of a stromal implant appeared to be of critical importance if an impermeable material was to be used. These insights resulted in the development of intrastromal ring segments (Intacs), conceived by Reynolds in 1978 and first studied in the late 1980s (12). Although originally conceived and studied as a 360-degree implant, this device has evolved into two 150-degree arcuate segments that are easier to implant and manipulate within the peripheral cornea and cause fewer healing problems.
Autologous Donor Corneal Tissue
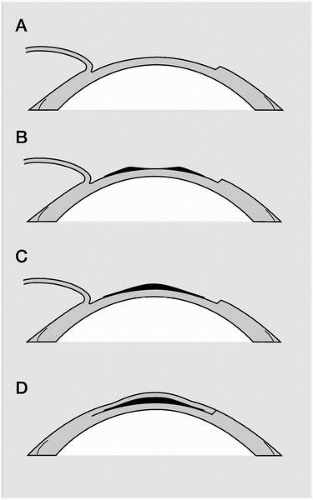
Keratophakia was introduced by Barraquer (1) in 1949. Keratophakia (kera: cornea; phacos: lens) refers to the placement of donor lenticules with refractive power within the stromal lamellae to correct either myopia or hyperopia. The lenticule is a piece of autologous donor corneal stroma that is frozen and shaped by a cryolathe (13) or harvested fresh by a microkeratome (14). Once implanted within the central stroma, the lenticule alters the anterior corneal curvature and thus its dioptric power (Fig. 74-1).
The keratophakia procedure as performed by Barraquer in the 1970s with cryolathe techniques or with early microkeratomes was skill-intensive and technically complex, and as a result was never widely adopted. With the advent of safer, more flexible microkeratome systems and artificial anterior chambers, autologous keratophakia may in the future claim a place within the practice of mainstream refractive surgery.
CURRENT APPROACHES
There are many strategies for the correction of myopia, hyperopia, and astigmatism via the alteration of corneal shape using additive surgical procedures. As with any elective refractive surgery, such procedures should be safe, effective, predictable, and stable. The potential adjustability and reversibility of corneal implant surgery are additional positive attributes. We will describe the procedures currently
used in clinical practice to correct myopia and hyperopia along with clinical indications, surgical techniques, published results, and future directions.
used in clinical practice to correct myopia and hyperopia along with clinical indications, surgical techniques, published results, and future directions.
Space does not permit an extensive discussion of patient selection and preoperative evaluation, and these issues are discussed at length elsewhere in this text. However, important components of any elective keratorefractive presurgical evaluation include a complete past medical and ocular history with specific attention to the history of the refractive state of the eye as well as ocular and systemic conditions that may be contraindications to corneal surgery in general. The complete preoperative ophthalmic exam should include as a minimum the following: visual acuity [uncorrected visual acuity (UCVA) and best spectacle-corrected visual acuity (BSCVA)], manifest and cycloplegic refraction, slit-lamp examination (with particular attention to the health of the ocular surface and adnexa), intraocular pressure measurement, and fundus examination. Corneal topography and pachymetry are important adjunctive studies, as they help exclude corneal ectatic conditions and are important for surgical planning. If there is any question as to the health of the corneal endothelium, analysis by specular microscopy is mandatory.
Intrastromal Corneal Ring and Ring Segments (ICR/ICRS-Intacs)
The 360-degree intrastromal corneal ring (ICR) and the 150-degree intrastromal corneal ring segment (ICRS) implants (Intacs, Addition Technology, Inc., Des Plaines, IL) are made of PMMA and may be used for the correction of low to moderate myopia of −1 to −4 D, although their optimal range seems to be from −1 to −3 D. The use of the 360-degree ring was abandoned after early clinical trials, as it became apparent that it was difficult to implant and more prone to healing problems near the incision site. The use of multiple, small peripheral segments is being investigated for the treatment of low hyperopia up to 2 D.
Myopic ICRS (Fig. 74-2) have an arc length of 150 degrees, an inner diameter of 7 mm, an outer diameter of 8.1 mm, and thickness in the range of 0.25 mm to 0.45 mm with increments of 0.05 mm. A 0.21-mm segment is also available for corrections of less than −1 D. Only the 0.25-, 0.30-, and 0.35-mm rings are approved for use in the United States. Table 74-1 describes the recommended prescribing range for Intacs and summarizes the refractive effects achieved with each segment diameter in the larger U.S. and European multicenter clinical trials.
When implanted within the peripheral cornea, ICRS flatten the anterior surface of the cornea by increasing the arc length curvature of the central cornea, with resultant flattening (Fig. 74-3). The advantage of ICRS over the other myopic keratorefractive procedures include surgery that spares the central cornea, maintenance of prolate asphericity of the cornea (15, 16, 17), and the reversibility of surgical effect with removal of segments (18, 19, 20).
Patient Selection
Implantation of ICRS is indicated for stable myopia of a mild degree (−1 to −3 diopters spherical equivalent), and a stable refraction. Good candidates have refractive cylinder of 0.5 D or less, as astigmatism is not readily correctable with this procedure. ICRS are contraindicated for individuals with autoimmune disorders, collagen vascular disease, and pregnant or nursing women. Ocular contraindications include the conditions described for other keratorefractive procedures, including recurrent corneal erosion syndrome and some corneal dystrophies, history of herpetic eye disease, and uncontrolled ocular surface disorders. Patients using such medications as amiodarone, sumatriptan, and isotretinoin are also not considered candidates for ICRS. Select patients with mild to moderate keratoconus may benefit from implantation of Intacs or
Ferrara rings (another type of ICRS), and this topic is discussed further below.
Ferrara rings (another type of ICRS), and this topic is discussed further below.
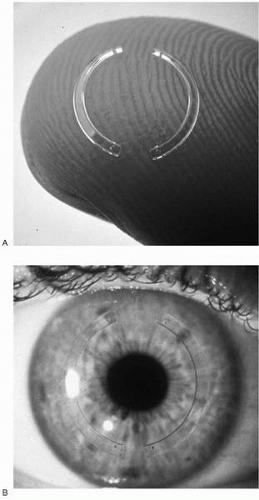 FIGURE 74-2. Intrastromal corneal ring segment (Intacs) 150-degree segments for myopia ex vivo (A) and in vivo (B).(see color image) |
TABLE 74-1. INTRASTROMAL RING SEGMENTS (INTACS) FOR MYOPIA: CLINICAL RESULTS AND PRESCRIBING INFORMATION | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree