Purpose
To investigate the utility of intraoperative optical coherence tomography (OCT) for Descemet membrane endothelial keratoplasty (DMEK) surgery.
Design
Prospective consecutive interventional case series.
Methods
DISCOVER (NCT02423213) is a prospective consecutive interventional case series examining the feasibility and utility of microscope-integrated intraoperative OCT in ophthalmic surgery. This report focuses on those eyes in the DISCOVER study undergoing DMEK surgery. The 8 cases were the first DMEK cases performed by the primary surgeon (J.M.G.) with microscope-integrated intraoperative OCT feedback. Qualitative OCT analysis was performed at multiple surgeon-defined time points, including host and donor tissue preparation, graft orientation, graft apposition, and tissue interface fluid dynamics.
Results
Correct graft orientation was confirmed by intraoperative OCT prior to unscrolling in 100% of cases. Seven of 8 grafts were fully attached at the conclusion of surgery and on postoperative day 1. One graft had a linear paracentral fixed area of interface separation corresponding to posterior stromal irregularities that was visible during surgery and unchanged on postoperative day 1. Two eyes developed significant peripheral graft dehiscence visible by the first postoperative week. Both grafts were successfully reattached with repeat gas injection. All eyes demonstrated improvement in best-corrected visual acuity and there was a 100% graft survival rate at last follow-up (minimum 4 months). Surgeon feedback indicated that intraoperative OCT provided valuable information in all 8 cases.
Conclusions
Real-time intraoperative OCT can provide useful information that may directly impact surgical decision making during DMEK surgery. Intraoperative OCT may facilitate the transition for novice DMEK surgeons by increasing surgeon confidence and reducing the risk of iatrogenic graft failure.
Descemet membrane endothelial keratoplasty (DMEK) offers several distinct advantages over Descemet stripping automated endothelial keratoplasty (DSAEK) for the treatment of corneal endothelial disease. Specifically, previous authors have demonstrated benefit with regard to faster visual rehabilitation, better visual acuity, and reduced rates of endothelial rejection. However, transitioning to DMEK surgery creates novel challenges for the corneal surgeon already skilled in DSAEK surgery. Difficulties and inconsistencies with donor tissue preparation, insertion, orientation, positioning, and unscrolling have all resulted in a steep “learning curve” for surgeons beginning DMEK surgery. Significant advances have been achieved in standardizing the steps and techniques used during DMEK surgery to reduce the frequency of intraoperative and postoperative complications. Despite these advances, many surgeons remain reluctant to transition to DMEK surgery owing to the technical and unique challenges of this surgical procedure.
One potential avenue for improved surgeon confidence and additional refinement of DMEK is the application of high-resolution, real-time intraoperative optical coherence tomography (OCT). This technology has already gained acceptance for its utility in visualizing the interface created during DSAEK. In 2014, the PIONEER study described the feasibility, utility, and safety of intraoperative OCT in ophthalmic surgery, including lamellar keratoplasty. In fact, the PIONEER study reported that intraoperative OCT enhanced surgeon understanding and potentially impacted surgical decision making in 40% of cases in lamellar keratoplasties. Similar to DSAEK, the implementation of intraoperative OCT for DMEK may provide information to the surgeon that helps improve tissue apposition and orientation while minimizing unnecessary and potentially dangerous manipulation of the donor scroll.
The DISCOVER study was initiated to assess the feasibility and utility of microscope-integrated intraoperative OCT for ophthalmic surgery with heads-up display surgeon feedback. The purpose of this report is to report the experiences and outcomes of a novice DMEK surgeon’s first consecutive cases performed with real-time intraoperative OCT from the DISCOVER study.
Methods
The DISCOVER study is a single-site, prospective multi-surgeon investigational device consecutive interventional case series. The study was approved by the Cleveland Clinic Institutional Review Board and adhered to the tenets of the Declaration of Helsinki. Informed consent was obtained from all participants. The purpose of the DISCOVER study is to examine the feasibility and utility of a microscope-integrated intraoperative OCT system in both anterior and posterior segment surgery and identify when the system helps to improve surgical outcomes. The integrated systems used in the DISCOVER study are able to provide real-time information regarding instrument-tissue interactions. For this report, the primary inclusion criterion was enrollment in DISCOVER for DMEK surgery.
Donor Tissue
All donor tissue was prestripped and “S-stamped” for DMEK surgery by Midwest Eye-Bank (Ann Arbor, Michigan, USA) as previously described (Veldman PB et al, “Eliminating the possibility of upside-down DMEK grafts: a novel stromal side S-stamp technique for DMEK.” Presented at Cornea Society Fall Education Symposium. November 15, 2013; New Orleans, Louisiana). In brief, the technician initiated a 9.5 mm partial trephination and stripped Descemet membrane (DM) with tying forceps. After the tissue was 80% stripped and elevated, a 2 mm circular trephination was performed eccentrically in the underlying stromal bed. DM was reapposed and the corneoscleral rim was placed upright. The exposed anterior surface of DM was dry-marked with gentian violet on a micro “S” stamp (Moria SA, Antony, France) and the 2 mm corneal button was replaced. The tissue was placed in Optisol GS (Bausch & Lomb, Rochester, New York, USA) storage media until the time of surgery.
Surgical Technique
Host DM was stripped with a reverse Terry-Sinskey hook (Bausch and Lomb, St Louis, Missouri, USA) with Healon (Abbott Laboratories Inc, Abbott Park, Illinois, USA) filling the anterior chamber. A minimum descemetorrhexis of 8 mm was performed. Healon was evacuated with an irrigating/aspirating handpiece and intraocular Miochol (Bausch & Lomb, Rochester, New York, USA) was injected to constrict the pupil. To optimize the red reflex during descemetorrhexis, Miochol was used intraoperatively rather than pilocarpine preoperatively. An inferior peripheral iridectomy was created at the 6 o’clock position with capsulorrhexis forceps through a dedicated vertical paracentesis site at the limbus.
In all cases, the donor tissue was punched by the surgeon with a 7.75 mm single-use corneal vacuum punch (Moria SA). Vacuum suction was applied to the epithelial surface but had to be aborted in 2 cases owing to unexpected retraction of the DMEK tissue into the punch site for the “S” stamp. In both of these cases the suction was released and the tissue was successfully repositioned and subsequently cut without suction. The tissue was stained for 3 minutes in 0.06% trypan blue solution. The trypan blue was replaced with balanced salt solution and the graft was then drawn into a modified Jones tube for DMEK (Gunther Weiss Scientific, Portland, Oregon, USA) attached to a 3 cc syringe. The tissue was injected into the anterior chamber through a 3.5- to 4-mm-wide triplanar corneal tunnel. The tissue was centered and oriented by a combination of external taps and successive fluid infusions through the paracentesis sites.
Once a properly centered and oriented double scroll was achieved, the tissue was unscrolled by anterior chamber collapse followed by external taps ( Supplemental Video 1 , available at AJO.com ). After the tissue was completely unscrolled, 20% sulfur hexafluoride gas was injected beneath the graft to elevate it against the posterior corneal surface ( Supplemental Video 2 , available at AJO.com ). The pressure was increased to approximately 40–50 mm Hg and the cornea was then swept in a center-to-periphery fashion to evacuate any remaining interface fluid. After the 10 minutes with a complete gas fill, a partial gas-fluid exchange was performed until an approximately 75%–90% gas fill was obtained and the gas was no longer obstructing the inferior peripheral iridectomy.
Use of Intraoperative Optical Coherence Tomography
Intraoperative imaging was performed using a microscope-integrated intraoperative OCT system (RESCAN 700; Carl Zeiss Meditec, Oberkochen, Germany), as previously described. This integrated system includes a “heads-up display” with a visible transparent overlay of the OCT data stream in the surgeon’s right ocular and an external video display panel. Surgeon foot-pedal control or external assistant control is possible. Intraoperative OCT was employed for select surgical steps, including host descemetorrhexis, graft orientation, and graft apposition. Both real-time and static intraoperative OCT feedback were used by the surgeon. A surgeon feedback form was used to evaluate the impact of intraoperative OCT on surgeon decision making as part of the DISCOVER study.
Results
Clinical Results
Eight eyes of 7 patients (4 female, 3 male; mean age 74 years, range 49–88 years) that underwent DMEK in the DISCOVER study were included in this analysis. All surgeries were performed by a single surgeon (J.M.G.). Of note, these were the first DMEK cases performed by this surgeon (with or without intraoperative OCT). The median “un-scrolling time” (onset of intraocular graft manipulation until full apposition with 100% gas fill) was 6 minutes and 15 seconds (range 2:25–27:36). The median duration of postoperative follow-up was 7 months (range 4–9 months).
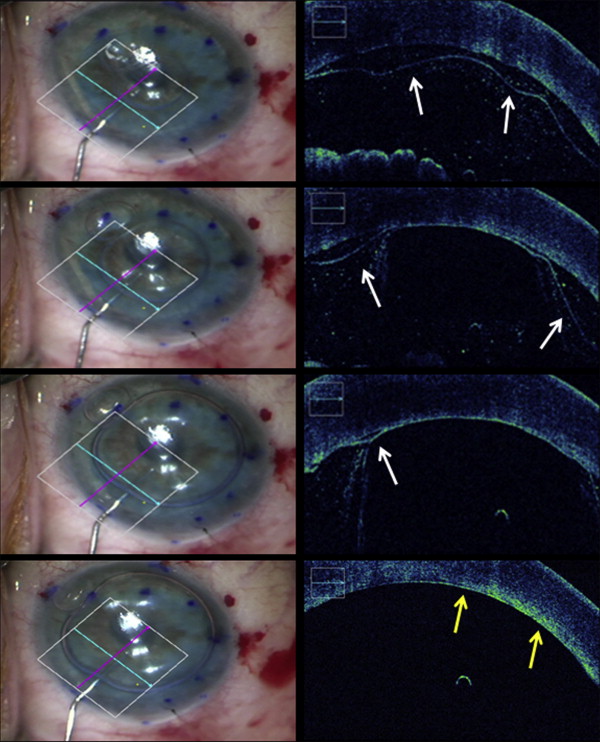
Seven of 8 grafts were fully attached on intraoperative OCT at the conclusion of the initial surgery. One graft had a linear area of nonadherence corresponding to a significant posterior stromal irregularity, which was visible on intraoperative OCT. There was no change in the adherence in any of the grafts on postoperative day 1. At week 1, 2 eyes developed partial graft detachments. Both grafts underwent successful rebubbling procedures and remained attached. All DMEK eyes experienced an improvement in best-corrected visual acuity and there was a 100% graft survival rate at last follow-up.
Intraoperative Optical Coherence Tomography Utility and Surgeon Feedback
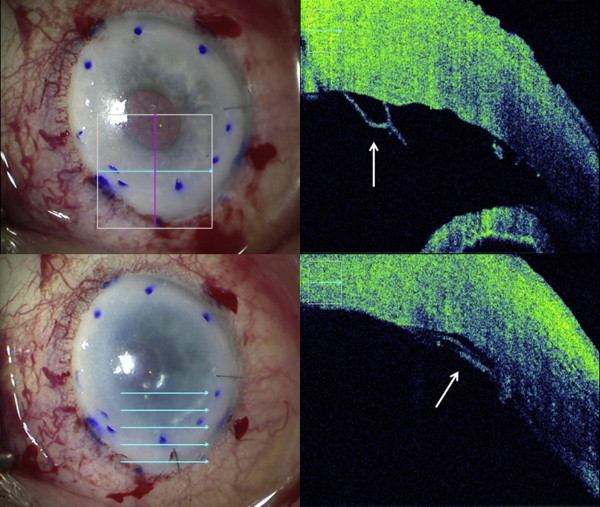
During descemetorrhexis, intraoperative OCT allowed identification of the presence of DM fragments and posterior stromal irregularities. In 2 of 8 cases, significant irregularities were visible after descemetorrhexis (Cases 1 and 2, Figures 1 and 2 ) and both grafts developed partial postoperative graft detachment requiring rebubbling.
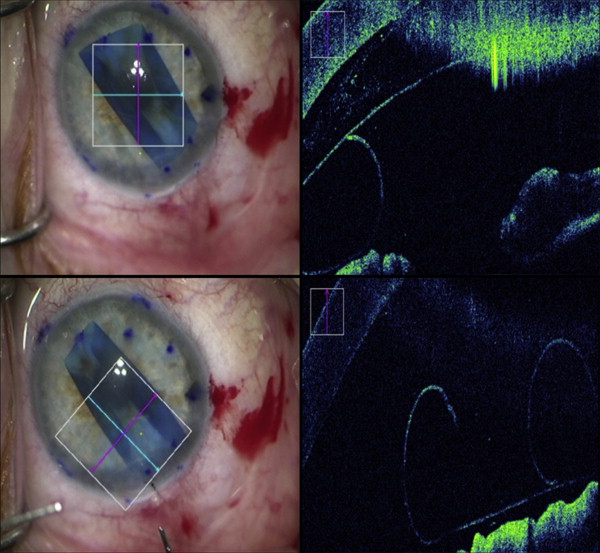
The graft orientation was rapidly identifiable with intraoperative OCT in 100% of cases based on the rolling behavior of the graft ( Figure 3 ). Real-time intraoperative OCT feedback contradicted surgeon’s immediate initial “en face” impression of tissue orientation in 4 of 8 cases. Of note, however, no concerted effort was made to identify the S-stamp prior to intraoperative OCT verification. The properly oriented “S” stamp was clearly visible in 6 of 8 cases at the conclusion of surgery. In 2 cases, the “S” stamp could not be clearly identified: 1 case in which the anterior chamber visibility was extremely poor owing to severe edema and dense arcus senilis, and 1 case in which the S-stamp was already barely discernible prior to graft preparation.
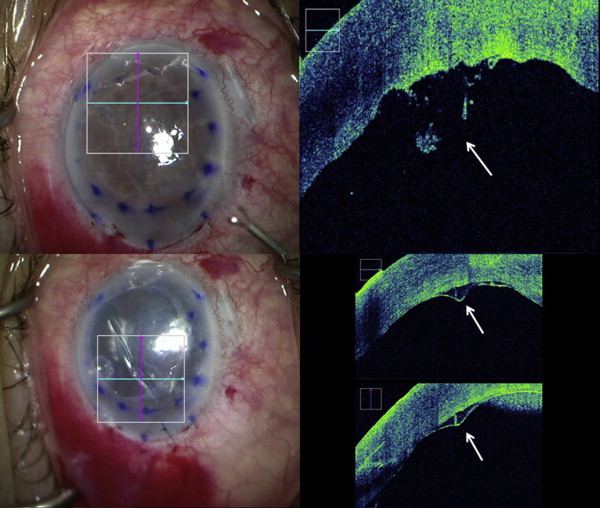
Intraoperative OCT was also used to evaluate graft apposition and the impact of external sweeping on interface fluid, when visible ( Figures 1 and 4 ). Six grafts were completely apposed on intraoperative OCT after gas injection alone. In 2 cases, intraoperative OCT identified areas of incomplete apposition. In 1 case, additional manipulation had no effect on the area of nonapposition. In the other case, external sweeping resulted in complete elimination of the interface separation.