Fig. 2.1
Aqueous outflow. Aqueous exits the eye through either trabecular or uveoscleral channels. Trabecular outflow, seen in red, is also termed pressure-dependent outflow and handles most of the flow of aqueous, estimated to be approximately 75 %. Uveoscleral, or nonpressure-dependent, outflow is nontrabecular and comprises flow through the uveoscleral tissues
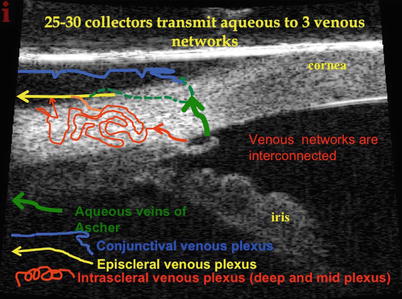
Fig. 2.2
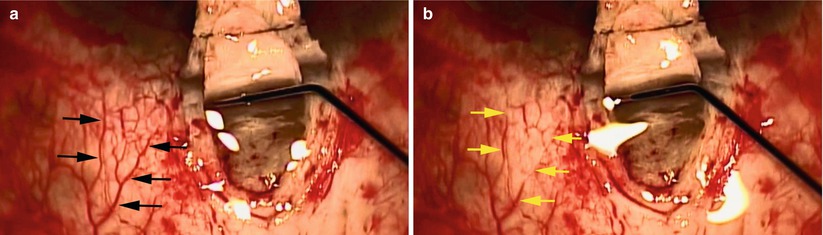
Aqueous collectors and venous networks. How does aqueous reach the distal collectors from the anterior chamber? There are approximately 25–35 collector channels that drain aqueous from Schlemm’s canal into three venous networks: the intrascleral plexus (deep and mid), the episcleral plexus, and the conjunctival plexus. The approximately 30 collectors are comprised of mainly smaller collectors that drain into the deep plexus, then drain into the mid scleral plexus, and then into the episcleral venous plexus. There is a small group of larger veins (8 of the 30) that largely bypass the reticulated intrascleral plexus and drain directly into the episcleral and/or conjunctival networks. These are known as the veins of Ascher and may be seen during slit lamp examination (see reference [3])
Mapping the Conventional Aqueous Collector System
Ashton’s Doyne’s memorial lecture revealed groundbreaking work on the canal of Schlemm and outlet channels [5]. His neoprene cast worked and confirmed an irregular number of collector channels, 17–35 in number, that vary considerably in size. Immediately after leaving the canal, the smaller collectors form a deep scleral plexus located just external to the canal. The aqueous is then transmitted to the mid-scleral plexus, together with the deep scleral plexus, forming the intrascleral venous plexus. This drains into the episcleral venous plexus. In addition, Ascher described larger collector vessels that span from Schlemm’s canal directly to the episcleral veins and/or the conjunctival plexus and largely bypass the intrascleral plexus. Thus, the aqueous exits Schlemm’s canal through approximately 30 collectors passing into three venous systems: the intrascleral, episcleral, and conjunctival networks. Six to eight of the larger collectors comprise the veins of Ascher, which may be seen at the slit lamp. Ascher described the aqueous veins in his landmark 1942 article and defined them as biomicroscopically visible pathways of blood-vessel-like appearance containing a clear colorless fluid or diluted blood and intercalated between Schlemm’s canal and conjunctival and subconjunctival veins.
Schlemm’s canal is an endothelial-lined circular tube that transmits aqueous from the trabecular meshwork to the distal collector outflow system. It is approximately 36 mm in circumference, 300 μm wide (meridional), and 25 μm in height with the majority of the roughly 30 collector channels located mainly nasally and then inferiorly. This location is quite fortunate for canal surgeons from an accessibility issue. The majority of the collector channels are small, around 20–40 μm, but up to eight of these vessels may be considerably larger and connect directly to the canal (Ascher’s veins) and avoid the plexus by communicating directly with either an episcleral or conjunctival vein.
Assessment of the Conventional Outflow System
Preoperative Assessment of the Conventional Collector System
It is difficult to study the collector outflow system in vivo, much less a diseased one or an outflow path that has been altered due to canal surgery. From a historical viewpoint, the first in vivo assessment of the collector system was truly the identification of blood in the canal of Schlemm. Investigators eventually discovered that blood could reflux into the canal by applying episcleral pressure with a flanged gonioprism or suction gonioprism. This raised EVP and reversed the pressure gradient causing the blood to reflux. In retrospect, this was an evaluation of the collector system between the canal and some of the distal collectors. The other way to reverse the pressure gradient is to artificially lower IOP below the EVP, and blood will collect in Schlemm’s canal in normal eyes and probably less so in glaucomatous eyes. This information in conjunction with Ascher’s studies of aqueous veins helped establish the path of aqueous once it left the canal of Schlemm. Therefore, preoperative gonioscopic maneuvers to cause blood to reflux into Schlemm’s canal may help predict that there is a communication from the distal to more proximal collectors, but the gonioprism induces obvious mechanical and pressure distortion. OCT with special imaging attachments and altered wavelengths are improving the ability to image the collector system. However, a preoperative commercially available imaging method of the collector system is not widely available. It is possible to see Schlemm’s canal with ultrasound biomicroscopic devices [6], but the smaller collectors are very difficult to see.
Intraoperative Assessment of the Aqueous Channels
Canal surgery presents a unique opportunity to evaluate and explore the conventional aqueous outflow system. Why is it that a canaloplasty, trabectome, or iStent procedure fails? If we were able to assess the entirety of the channel anatomy at the time of surgery and it appeared to be patent but the procedure failed, it would more likely be due to postoperative scarring in or adjacent to the canal, not the fact that the downstream collector system was atrophic and/or unsalvageable. This is imperative to understand in order to advance the field of canal surgery and make pathophysiologic correlations. Gonioscopy or endoscopy is still a key method of visualizing the trabecular meshwork and canal of Schlemm intraoperatively for these structures are large enough to appreciate, but what is happening in the downstream aqueous channels is difficult to ascertain. Typically, we are unable to appreciate if the downstream aqueous channels are intact or totally atrophic from years of glaucomatous downstream collector atrophy from lack of aqueous flow. The other possibility is that the glaucomatous process not only involves the proximal trabecular meshwork but also, in some patients, involves the mid or distal system of collectors. Of course, this might vary depending on the genetic makeup of the individual with primary open-angle glaucoma (POAG).
When viscocanalostomy was introduced, it was not uncommon to see laminar flow into adjacent collectors and episcleral veins while injecting balanced salt solution (BSS) into the orifice of the canal. This phenomenon was also observed during canaloplasty (Fig. 2.3). This exciting observation verified the correct location of the canal and demonstrated flow through the collectors. Intraoperatively, some investigators have infused dye into the anterior chamber adjacent to an iStent with identification of various episcleral collector structures, but we await further published reports on this technique. Grieshaber [7] and colleagues published the intraoperative evaluation of aqueous channels during canaloplasty. They evaluated blood reflux into Schlemm’s canal and fluorescein tracer channelography during canaloplasty. The blood reflux in the canal of Schlemm was observed after acutely lowering IOP by paracentesis. They found that excellent blood reflux into Schlemm’s canal was associated with a lower preoperative IOP and poor to no reflux was associated with a high preoperative IOP. The tracer was injected directly into the canal through the iTrack microcatheter during canaloplasty (iScience Interventional, Menlo Park, CA). The investigators observed egress of dye through the episcleral venous plexus and graded the dispersion of dye. A broader dispersal of dye into the aqueous channels was associated with a lower postoperative IOP. The authors felt that provocative gonioscopy and channelography may reflect the function of the outflow pathway and correlate with canaloplasty outcome. This intuitively makes sense because a higher preoperative IOP is more likely associated with collapse of the canal of Schlemm along with distal compromise of the collectors.