, Yasuya Nomura2 and Yasuya Nomura3
(1)
The Society for Promotion of International Oto-Rhino-Laryngology, Tokyo, Japan
(2)
The University of Tokyo, Tokyo, Japan
(3)
Showa University, Tokyo, Japan
Abstract
All sensory hair cells of the inner ear exist in extracellular fluid, but in some areas the fluid has the characteristics of intracellular fluid, as far as concentrations of K+ and Na+ are concerned. The membranous labyrinth contains the intracellular fluid-like endolymph, and is surrounded by a distinct extracellular fluid, the perilymph. The sensory hair cells of the cochlea are surrounded by extracellular fluid called cortilymph in a separate compartment. Excessive accumulation or leakage of any of these fluids may affect the functions of the whole or partial inner ear.
This chapter is concerned with perilymphatic fistula, signs and symptoms of which mimic Meniere’s disease and sudden deafness. Because the pathophysiology of perilymphatic fistula formation is not clear, we have attempted to experimentally induce leakage of perilymph from the cochlear windows of guinea pigs by injecting artificial perilymph into the subarachnoid space or by suctioning a small amount of perilymph through the round window membrane. In these studies, the membranous labyrinth demonstrated various changes. The cochlea developed endolymphatic hydrops. The vestibular membranous labyrinth collapsed to a greater or lesser degree. In some experimental conditions, the organ of Corti showed cortilymphatic hydrops.
Keywords
Cerebrospinal fluid pressureCochlear aqueductCortilymphatic hydropsExperimental perilymphatic fistulaPerilymphPerilymphatic fistula2.1 Inner Ear Fluid
2.1.1 Perilymph and Endolymph
In 1760, Domenico Cotugno (Italian anatomist, 1736–1822) discovered the presence of fluid in the inner ear, which was thereafter called the “liquid of Cotugno.” Later, Henri Marie Ducrotay de Blainville (French anatomist, 1777–1850) named the fluid “perilymph.” The fluid within the membranous labyrinth was called “endolymph of Breschet” (Gilbert Breschet, French anatomist, 1784–1845). The vestibular membrane was described in 1854 by Ernst Reissner (German anatomist, 1824–1878), and has been called Reissner’s membrane since that time.
2.1.2 Cortilymph
A third fluid space exists within the organ of Corti. Morphological studies have found no direct communication between the endolymphatic or perilymphatic spaces and the fluid within the organ of Corti. In 1960, Hans Engström [1] proposed the name “cortilymph” for the fluid filling the tunnel of Corti, the space of Nuel, the region around the hair cells, and the outer tunnel (Fig. 2.1).
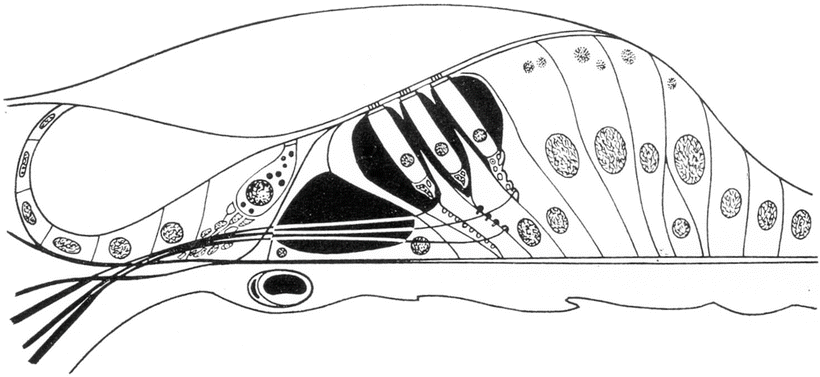

Fig. 2.1
The cortilymph occupies the space within the organ of Corti [1]. Engström H (1960) Acta Morphol Neerl Scand 3:195 Taylor & Francis Ltd. www.tandfonline.com
Schuknecht and Seifi [2] perfused the scala tympani of a cat with a histochemical acetylcholinesterase staining solution, and found deposits of reaction product extending from holes in the lower shelf of the osseous spiral lamina to inside the organ of Corti (Fig. 2.2). Schuknecht believed that these were communicating perilymph channels from the scala tympani to the organ of Corti. The holes in the lower shelf of the osseous spiral lamina were named the “canaliculi perforantes Schuknechtii.” They are located 0.2 mm from the habenula perforata in the cat. The staining solution flowed from the osseous spiral lamina through the habenula perforata into the organ of Corti. In a frozen specimen, however, no acetylcholinesterase reaction deposit could be found in the lower shelf of the osseous spiral lamina (Fig. 2.3).
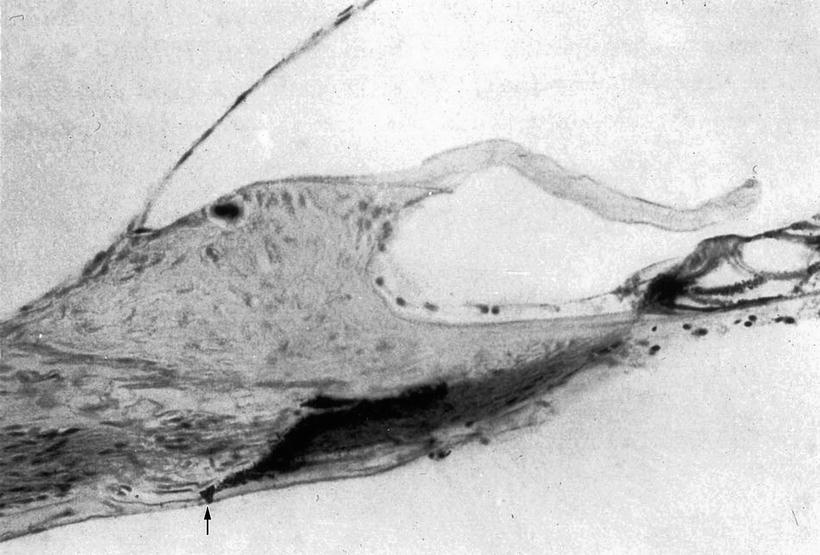
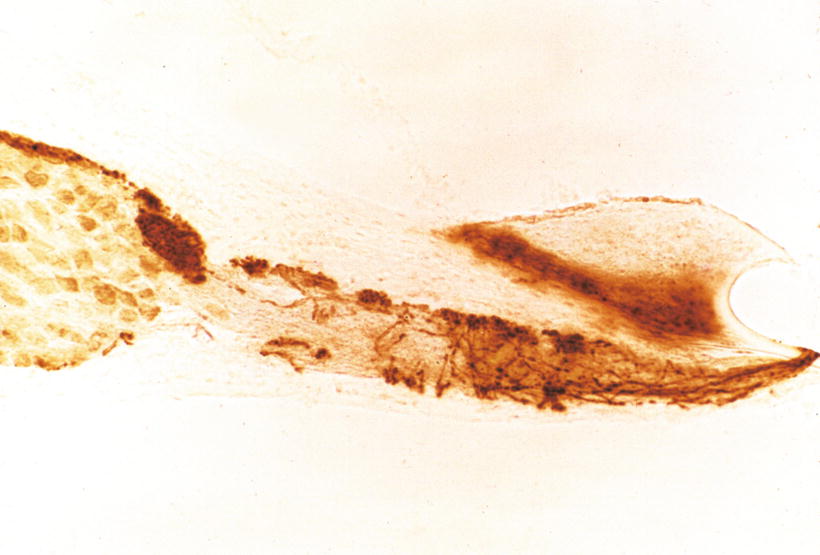

Fig. 2.2
Staining solution for acetylcholinesterase enters the osseous spiral lamina of a cat through a hole in the lower shelf. Reaction product is observed from the osseous spiral lamina to the organ of Corti. Arrow: Canaliculi perforantes Schuknechtii. Koelle’s method (Courtesy of Dr. Schuknecht)

Fig. 2.3
Acetylcholinesterase activity in efferent fibers within the osseous spiral lamina of the cat. Strong activity is found in the intraganglionic spiral bundle and in the many small bundles adjacent to the upper and lower bony shelves. Frozen section, Gomori’s method (original ×6.5)
Masuda et al. [3] demonstrated perilymphatic communication routes between the perilymph of the scala tympani and the cortilymph in guinea pigs by injecting inulin-methoxy-H3 through the round window membrane. Inulin accumulated not only in the habenula perforata and basilar membrane, but also in the cortilymph spaces, suggesting that the habenula perforata and basilar membrane are communication routes between the two spaces.
Nomura [4] perfused the scala tympani of guinea pigs using staining solution for succinic dehydrogenase with phenazine methosulfate. There were numerous holes in the lower shelf of the osseous spiral lamina through which the solution penetrated into the osseous spiral lamina (Fig. 2.4). Formation of reaction product, formazan, was rapid with phenazine methosulfate. Staining was observed along nerve fibers in the osseous spiral lamina. The staining solution passed from the osseous spiral lamina through the habenula perforata to the space in the organ of Corti (Fig. 2.5). The entire basilar membrane was eventually stained blue. But the first very rapid passage of solution into the organ of Corti was via a passageway between Deiters’ cells and Hensen’s cells (Fig. 2.6) [4]. According to Beagley [5], normal histological preparations showed fine canal-like intercellular spaces between Deiters’ cells and Hensen’s cells. Near the membrana reticularis, however, strong desmosomes join Hensen’s cells to the outermost phalangeal process of Deiters’ cells and this area seemed quite stable under the stress of acoustic trauma. By contrast, the base of the Hensen-Deiters’ junction showed a wide cleft in some acoustically stimulated guinea pigs. Beagley was of the opinion that the Hensen-Deiters’ split had protected the hair cells from acoustic stimuli.
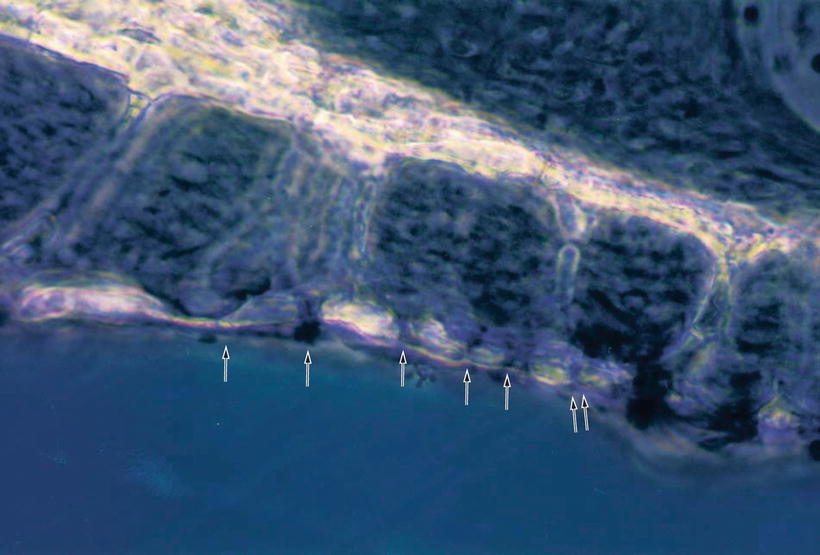
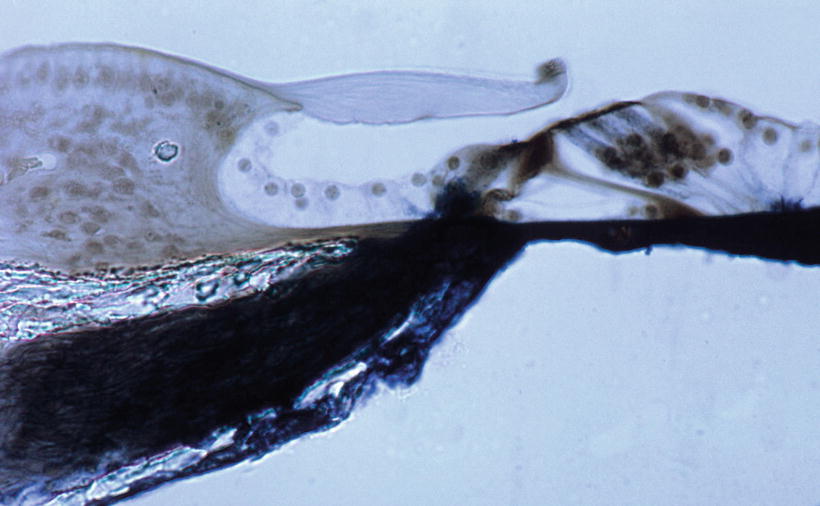
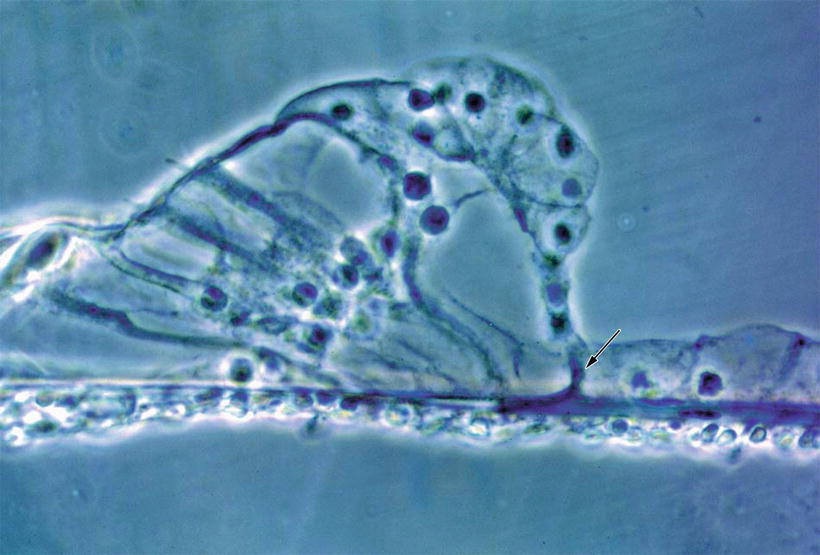

Fig. 2.4
Numerous holes (arrows) seen in the lower shelf of the osseous spiral lamina of the guinea pig. Staining solution for succinic dehydrogenase with phenazine methosulfate was introduced in the scala tympani (original ×16) [4]

Fig. 2.5
Fluid passageway from the scala tympani to the organ of Corti. The solution entered through holes in the lower osseous spiral lamina to the inside the osseous spiral lamina of the guinea pig. The nerve fibers were stained. The solution entered the organ of Corti through the habenula perforata and basilar membrane (original ×6.5)

Fig. 2.6
Fluid passageway through the basilar membrane. Marker solution rapidly passed through the localized area of basilar membrane, and entered the organ of Corti through the space between Deiters’ cell and Hensen’s cell in the guinea pig (arrow). Arrow indicates a route for fluid into the organ of Corti (original ×40) [4]
From an electron microscopic study, Ishiyama and Ishiyama [6] noted that there was a wide gap between Deiters’ cells and Hensen’s cells adjacent to the basilar membrane (Fig. 2.7).
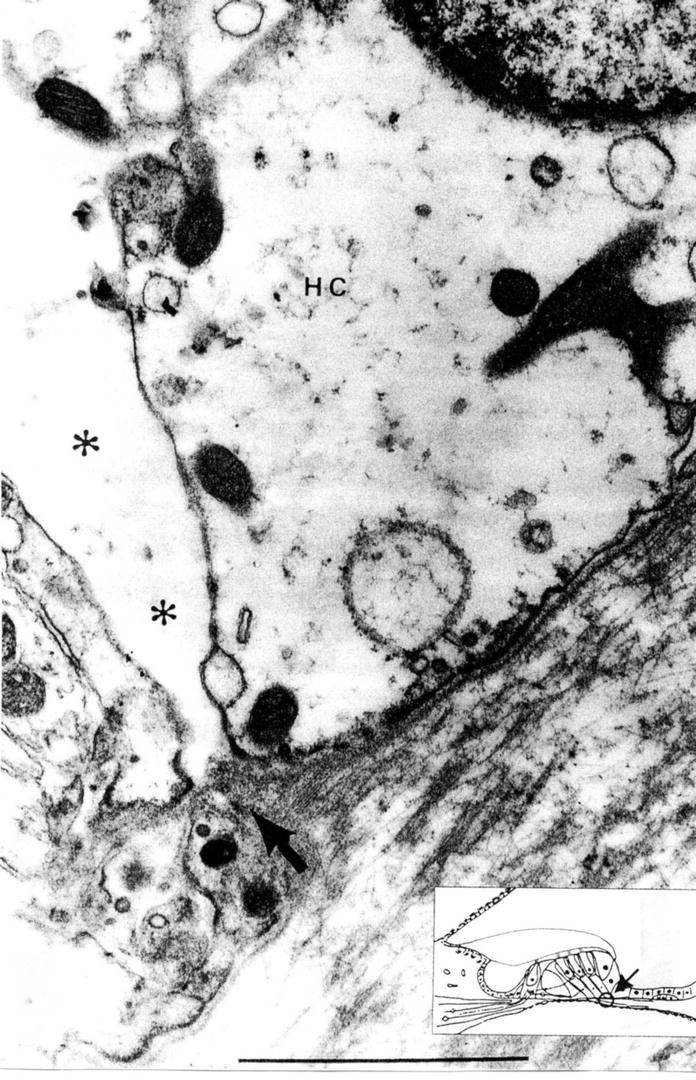

Fig. 2.7
There is a wide space between the junction (asterisk) of Deiters’ cell and Hensen’s cell. The inset shows the area of observation. Arrow indicates a route for fluid into the organ of Corti. HC Hensen’s cell, scale: 1 μm (Courtesy of Drs. Ishiyama and Ishiyama) [6]
In Nomura’s study, after perfusion of the solution containing phenazine methosulfate, the organ of Corti became hydropic. Hensen’s cells were pushed laterally, detached from the basilar membrane, and ballooned, so that there was a large space in the lateral part of the organ of Corti including the outer tunnel. This is the initial sign of formation of cortilymphatic hydrops. If hydropic conditions progress, the lateral portion of the membrana reticularis will be lifted (Fig. 2.8).
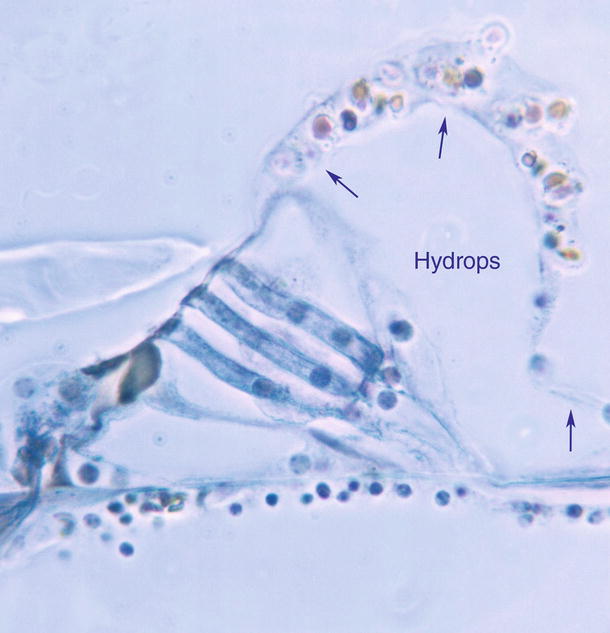

Fig. 2.8
Cortilymphatic hydrops. The organ of Corti is stained blue. Hensen’s cell is detached from the basilar membrane. The split between Deiters’ cell and Hensen’s cell is enlarged. Hensen’s cells are ballooned (arrows). Lateral part of the membrana reticularis is lifted. The inner pillar cell is crushed. Perfusion of the solution for staining succinic dehydrogenase with phenazine methosulfate, Guinea pig, original ×40
The pillar cells and Deiters’ cells contain large numbers of tonofibrils. The pillar cells are firmly fixed to the basilar membrane. These cells together with the membrana reticularis strongly support the architecture of the organ of Corti. Hensen’s cells have no contact with Deiters’ cells at the base. The lateral aspect of the organ of Corti, where Hensen’s cells are found, is the weakest and least able to tolerate increased pressure. Although the functions of Hensen’s cells are not well understood, one role may be to release pressure within the organ of Corti.
As hydrops of the organ of Corti progresses with increasing osmotic pressure and/or diffusion from the scala tympani, the phalangeal processes of Deiters’ cells and the pillar cells become slender, and hair cells disappear. The membrana reticularis and Hensen’s cells become thin, but as a tough membrane they maintain the boundary of the organ of Corti (Fig. 2.9).
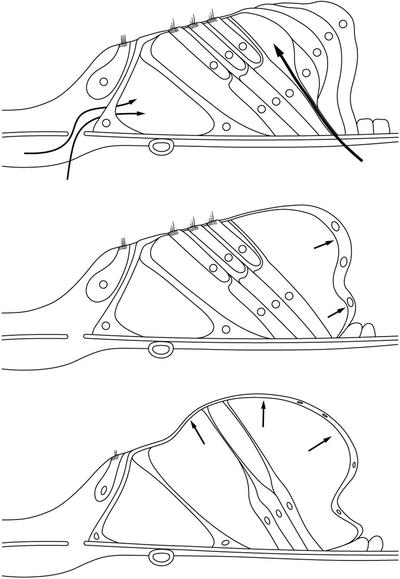

Fig. 2.9
Schema showing the process of cortilymphatic hydrops formation. Fluid enters the organ of Corti through the Deiters–Hensen’s cell slit and habenula perforata. As the pillar cells and Deiters’ cells are hardy, Hensen’s cells are pushed laterally. As hydrops progresses, the pillar cells and Deiters’ cell become elongated. The outer hair cells disappear
Djeric and Schuknecht [7] reported two cases of Hensen’s cell cyst of the organ of Corti in human temporal bones. The cysts were bilaterally symmetrical. The authors postulated that the cysts might represent coalesced lipid droplets from Hensen’s cells.
2.1.3 The Cochlear Aqueduct
The cochlear aqueduct and fundus of the internal auditory canal are communicating routes between the cerebrospinal fluid (CSF) and the perilymph (Fig. 2.10). The internal aperture of the cochlear aqueduct is located near the round window and the inferior cochlear vein (Fig. 2.11).
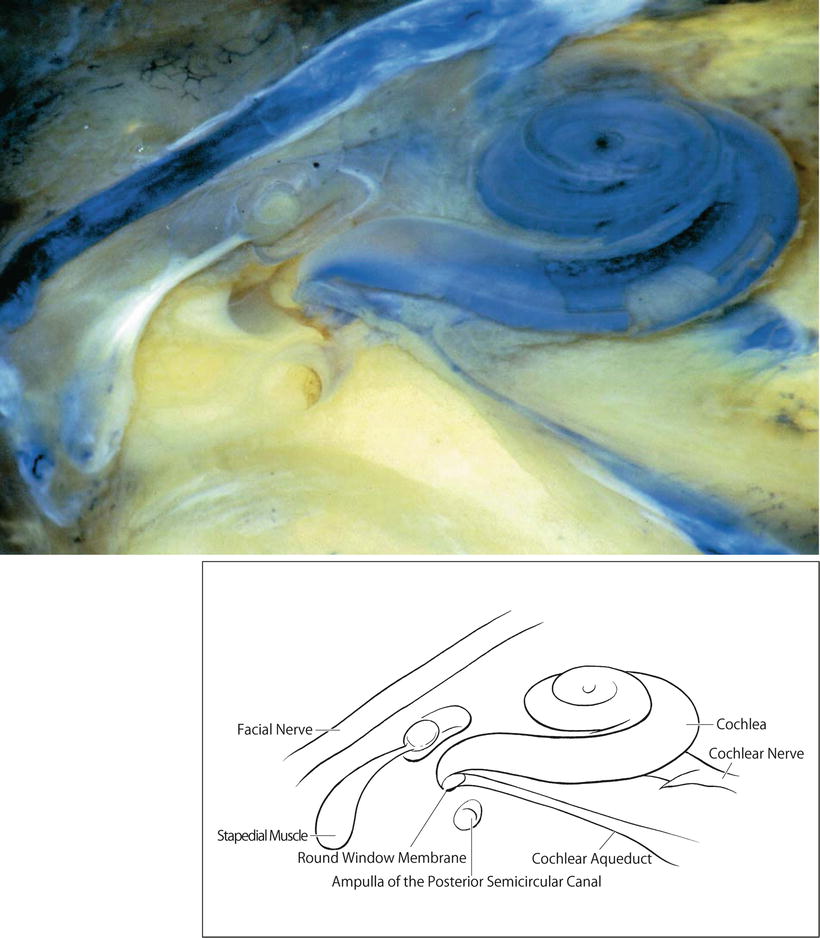
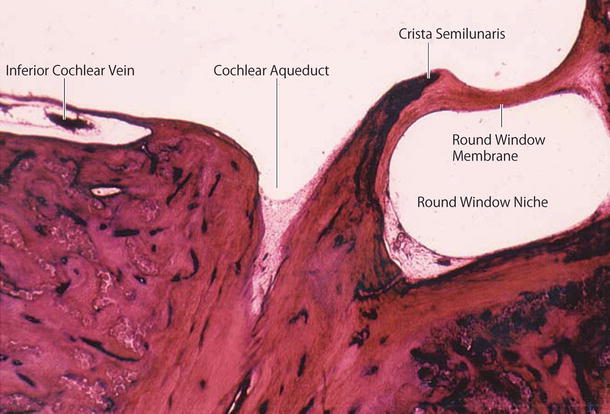

Fig. 2.10
Human cochlea and cochlear aqueduct. The internal aperture of the cochlear aqueduct is near the round window. Sudan Black B stain (original ×2.5)

Fig. 2.11
The cochlear aqueduct’s internal aperture near the round window. The inferior cochlear vein runs within the canal of Cotugno (original ×6.5)
Gopen et al. [8] evaluated 101 temporal bones from subjects aged 0–100 years, and reported that the mean diameter of the narrowest portion of the cochlear aqueduct was 138 ± 58 μm, which occurred 200–300 μm from the cochlear end of the aqueduct. They noted four types of aqueduct patencies: central lumen patent throughout the length of the aqueduct (34 %), lumen filled with loose connective tissue (59 %), lumen occluded by bone (4 %), and obliteration of the aqueduct (3 %). There was no correlation between age and narrowest diameter, or between age and type of patency.
The cochlear aqueduct is not a route for cerebrospinal fluid in cases of stapes gusher. Stapes gusher occurs in the cochlea when the fundus of the internal auditory canal is missing, and a wide communication exists between the internal auditory canal and the scala vestibuli (Fig. 2.12).
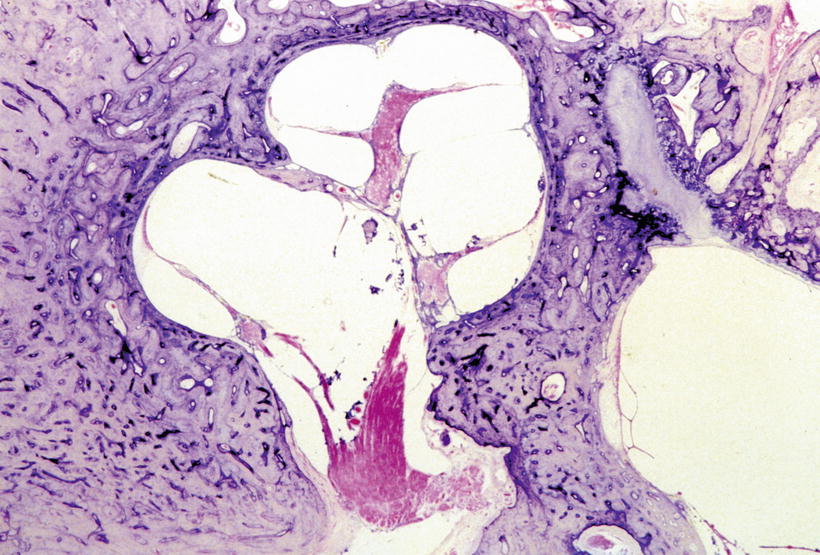

Fig. 2.12
This temporal bone is from a patient with otopalatodigital syndrome. The bone shows fixation of the stapes, scala communis, defective modiolus, and a large communication between the internal auditory canal and scala vestibuli. No fundus of the internal auditory canal is observed. There is a risk of stapes gusher occurrence (Courtesy of Dr. Schuknecht)
2.1.4 Perilymph and the Limbus Spiralis
The portion of the limbus spiralis facing the scala vestibuli was named the vasculoepithelial zone by Borghesan [9]. The zone is believed to be related to the production of perilymph constituents and is lined by osmiophilic cells [6, 10, 11]. The cells have also been shown to have Na+/K+-ATPase activity [12].
Hsu and Nomura [13] demonstrated strong carbonic anhydrase activity in the vasculoepithelial zone (Fig. 2.13). Carbonic anhydrase is an enzyme that assists the rapid conversion of carbon dioxide and water into carbonic acid, hydrogen, and bicarbonate ions.
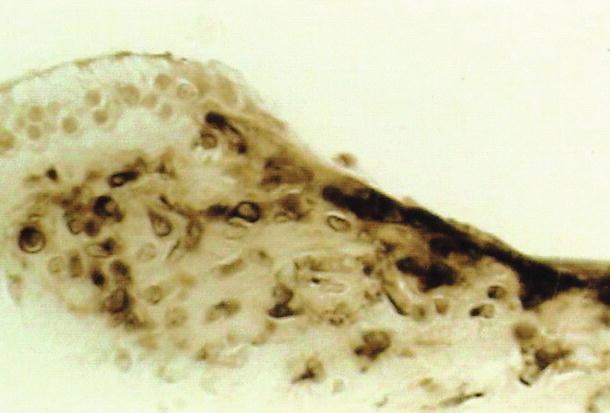
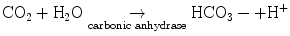

Fig. 2.13
Carbonic anhydrase activity in the limbus spiralis. The vasculoepithelial zone demonstrates strong enzyme activity. The interdental cell and its vicinity show no reaction. Guinea pig [13]
Carbonic anhydrase is an important enzyme in the regulation of pH and fluid balance in different parts of the body, including the inner ear. Light and electron microscopic studies have shown diverse distribution of the enzyme in the inner ear. The stria vascularis and dark cells have strong carbonic anhydrase activity [13]. The neuroepithelia of the crista ampullaris show strong activity in the supporting cells, but sensory cells with nerve chalices show no activity, although the reaction products are found on the stereocilia (Fig. 2.14).
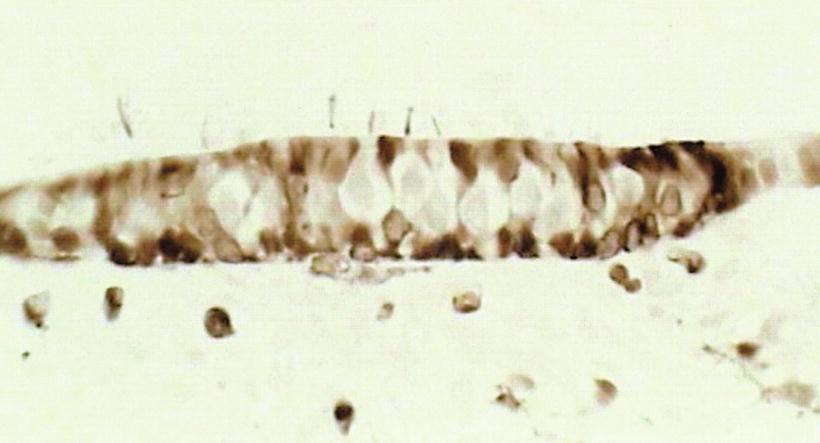

Fig. 2.14
Carbonic anhydrase activity in the neuroepithelia of the crista ampullaris. Supporting cells show carbonic anhydrase activity. The sensory cell and nerve chalice show no activity. The stereocilia show deposits of reaction products. Guinea pig (original ×40) [13]
The inner ear maintains its functions through the actions of various cells and of enzymes such as carbonic anhydrase, Na+/K+-ATPase, and adenyl cyclase.
2.1.5 Endolymph
Endolymph is produced mainly in the stria vascularis and dark cell areas. Electrolyte concentrations in the endolymph are quite different from those in the perilymph. In 1954, Smith et al. [14] demonstrated a high potassium ion concentration and a low sodium ion concentration in the endolymph, while the reverse was found in the perilymph.
Unlike the utricle and ampullae of the semicircular canals, the saccule has no dark cell areas. Hydrops of the saccule is often quite marked in patients with Meniere’s disease. The origin of the endolymph in these cases is probably the stria vascularis.
The membrana reticularis divides the cortilymph from the endolymph. When the cuticular plate of the outer hair cells disappears, repair of the membrana reticularis will take place simultaneously. The area of the cuticular plate is tightly enclosed by the surrounding phalanxes of Deiters’ cells and the heads of the pillar cells, so that no contamination occurs between the endolymph and the cortilymph. When the stria vascularis is damaged by laser irradiation, rapid repair takes place to maintain the scala media.
Endolymphatic hydrops is described in the chapter on Meniere’s disease.
2.2 Perilymphatic Fistula
2.2.1 Clinical Aspects
Perilymph may leak into the tympanic cavity from the inner ear in cases of temporal bone trauma, including ear surgery. However, leakage can occur without apparent cause, leading to signs and symptoms of inner ear malfunction. This condition is called spontaneous perilymphatic fistula.
Perilymphatic fistula is suggested if any of the following symptoms is evident:
Tinnitus, hearing loss, fullness of the ear, dizziness, and disequilibrium, occurring immediately after straining, nose blowing, or any other physical activity that causes a rapid increase of cerebrospinal fluid (CSF) pressure. The same symptoms can also occur with a rapid change of atmospheric pressure that affects the middle ear pressure.
Sensorineural hearing loss developing over a period of hours or a few days.
Tinnitus or a sensation of running water within the ear.
Dizziness when pressure in the external ear is artificially increased or decreased.
Fullness of the ear, hearing loss, tinnitus, dizziness, and disequilibrium following a popping sound.
Perilymphatic fistula can also occur without hearing loss or dizziness. A definitive diagnosis can be made by confirming perilymph leakage or fluid from the oval and/or round window at exploratory tympanotomy or endoscopic inspection. The pathophysiology of perilymphatic fistula remains unclear, mainly because of the paucity of histopathological studies of the inner ear in patients with the disorder.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


