Purpose
This study sought to determine the cost effectiveness of deep anterior lamellar keratoplasty (DALK) compared with penetrating keratoplasty (PK) for the treatment of keratoconus. DALK is associated with lower rates of postoperative complications compared with PK, but is a more expensive procedure; whether it is cost effective compared with PK is unknown.
Design
The study was an incremental cost-utility analysis from a health systems perspective using retrospective data on a cohort of patients with severe keratoconus.
Methods
The analysis was conducted using 1-year cost and outcomes data from 148 keratoconus patients, representing 102 PK and 46 DALK cases, seen for corneal grafts between January 1991 and January 2009 at the Singapore National Eye Center. The main outcome assessed was the incremental cost-utility ratios associated with PK and DALK for the treatment of keratoconus.
Results
Over a 20-year period, PK was the lower cost procedure and had an incremental cost-utility ratio of $3750 per quality-adjusted life-year compared with no surgical treatment. Compared with PK, DALK has an incremental quality-adjusted life-year gain of 0.8 and an incremental cost of $2420, for an incremental cost-utility ratio of $3025 per quality-adjusted life-year.
Conclusions
Compared with PK, DALK has a favorable cost-effectiveness ratio and, resources permitting, should be considered as a first-line treatment for keratoconus. Additional studies are needed to confirm the expected advantages of DALK over PK when it comes to long-term graft failure rates.
The surgical correction of keratoconus involves replacement of the diseased cornea with an allograft cornea. Penetrating keratoplasty (PK), a full-thickness replacement of the cornea, currently is the most widely used corneal transplantation technique worldwide; its safety and efficacy in the treatment of advanced keratoconus have been validated over the past few decades. One study has demonstrated its cost efficacy as a treatment for keratoconus.
The use of PK, however, has been associated with numerous complications, the most important of which are graft rejection and failure, but which also include astigmatism, microbial endophthalmitis, and development of cataracts or glaucoma. Long-term studies have shown poor graft survival rates after PK, with 10-year survival rates as low as 50% to 60%. This poor survival rate is explained chiefly by episodes of immunologic graft rejection of the donor endothelium and by the continual attrition of endothelial cells leading to graft failure. In the Singapore Corneal Transplant Study, more than half of all PK graft failures were attributable to these 2 causes.
Advances in surgical instrumentation and technique have led to a revival of interest in anterior lamellar keratoplasty, in which just the diseased anterior layers of the cornea are replaced, while leaving the healthy recipient endothelial cell layers intact. Conventional anterior lamellar keratoplasty surgery involves manual dissection of the stromal layers and can lead to an irregular stromal bed and subsequent interface haze, reducing postoperative visual acuity (VA). Through a variety of techniques, deep anterior lamellar keratoplasty (DALK) has made it possible to remove the entire corneal stroma down to the Descemet membrane (DM) with greater safety and consistency. The preferred technique in use at the Singapore National Eye Centre is a modified Anwar technique (DALKa), in which air is injected forcibly in the stromal bed to separate DM from deep corneal stroma. Because DALKa eliminates the graft–host stromal interface, concerns of suboptimal visual outcomes resulting from surface irregularities and scarring are minimized.
DALK may have several advantages over PK for the treatment of keratoconus. It leaves the recipient endothelium intact, eliminating the possibility of donor endothelial layer-mediated rejection of the transplant. For the same reason, patients undergoing DALK typically require fewer months of topical steroids compared with PK patients, and therefore are less susceptible to complications from corticosteroid-associated intraocular pressure elevation, decreased wound strength leading to globe rupture, and reduced local ocular surface immunity. Furthermore, the endothelial cell loss, and thus potential for graft failure, that accompanies DALK consistently has been shown to be lower than that for PK at all time points 1 to 5 years after surgery. Although long-term graft survival data still are lacking, at least 1 study of 1144 eyes predicted higher 20-year graft survival rates for DALK compared with PK (63.2% and 40.6%, respectively). Lastly, because a healthy donor endothelium is not required, lower quality, less expensive grafts are suitable for use in DALK.
DALK, however, is a more complex technique that requires an operative time approximately twice as long as that of PK. Intraoperative complications specific to the DALKa technique such as DM microperforation or macroperforation, or failure to expose DM, also may necessitate conversion of DALKa to a manual dissection technique of DALK (DALKm) or PK, which are likely to lead to comparatively worse VA outcomes. Thus, although evidence is accumulating from a clinical point of view that the DALK technique may result in improved outcomes compared with PK, whether DALK is cost effective relative to PK remains unclear.
This study performed a cost-utility analysis of DALK compared with PK for the treatment of keratoconus using a cohort of patients seen at the Singapore National Eye Centre. To date, no study has quantified and compared the relative benefits and costs of these 2 techniques. The model presented easily can be adapted to other settings, allowing clinicians and policy makers to make informed decisions concerning the optimal allocation of finite healthcare resources.
Methods
Overview
An incremental cost-utility analysis from a health system perspective comparing DALK to PK was performed using retrospective 1-year VA and complications data on patients treated for keratoconus seen at the Singapore National Eye Centre. The diagnosis of keratoconus, indications for surgery, and surgical technique have been previously described. In short, the diagnosis of keratoconus was made clinically from history, slit-lamp examination, keratometry, and refraction. The indication for grafting was poor functional vision and intolerance of other methods for optimal optical correction such as spectacles or rigid gas permeable contact lenses. A total of 196 patients underwent corneal grafts between January 1991 and January 2009 at the Singapore National Eye Centre. Of those, 30 cases with incomplete follow-up, and 18 cases of automated lamellar therapeutic keratoplasty were excluded. Automated lamellar cases differ from DALK technically, and were excluded because of the possibility of confounding the comparison between pre-Descemetic and Descemetic groups. The remaining 148 cases were used in the analysis and comprised 102 PK and 46 DALK cases.
Visual Acuity Data
VA was assessed using Snellen VA charts for all patients before and 1 year after corneal transplantation. Both preoperative and postoperative VA results refer to best spectacle-corrected visual acuity. Best spectacle-corrected visual acuity results were converted to logarithm of the minimal angle of resolution (logMAR) equivalent units for analysis. All VA results are reported in logMAR units.
Decision Model
A decision tree ( Figure 1 ) was constructed using TreeAge Pro Suite 2009 (TreeAge Software, Williamstown, Massachusetts, USA) to compare 1-year postoperative VA outcomes associated with PK and DALK. The root of the decision tree is a decision node with 2 branches corresponding to PK and DALK.
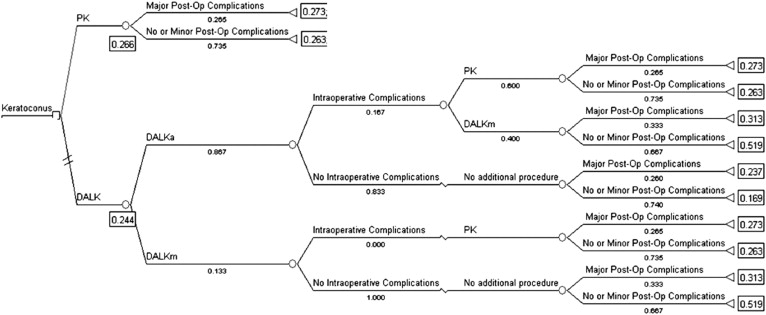
The PK branch leads to a chance node that reflects the probability of major postoperative complications. The DALK branch first features a chance node reflecting the likelihood that the operated-on eye is suitable for DALKa (as opposed to DALKm). Because DALKa is associated with better VA outcomes, it is always preferred to DALKm, except in cases where the cornea is judged to be at a high preoperative risk for intraoperative perforation (i.e., in cases of previous hydrops or deep posterior stromal scars). Chance nodes from each branch then reflect the probability of an intraoperative complication (most commonly resulting from DM macroperforation) necessitating conversion of the operative technique. For DALKa, this could be conversion to either DALKm or PK. Intraoperative complications during DALKm could necessitate conversion to PK.
The terminal nodes of the decision model reflect the least squares mean postoperative VA associated with each operative technique after controlling for differences in preoperative VA, stratified by the presence or absence of major postoperative complications. Major postoperative complications were defined as those, based on clinical experience, thought to affect postoperative VA over the long term. Major complications included wound dehiscence, suture abscess, endophthalmitis, microbial keratitis, herpes simplex virus activation, recurrence of keratoconus, glaucoma, cataract formation, graft failure, and graft rejection. The expected VA for each operative procedure then was calculated by multiplying the VA outcomes at the terminal nodes by the probability of terminating at that node.
Conversion of Visual Acuity to Utilities
Quality of life has been shown to be directly proportional to Snellen VA in the better-seeing eye. This correlation is independent of age, race, gender, socioeconomic status, and length of time of diseases. It also has been shown that VA itself, rather than the cause of the vision loss, is most correlated with ocular utilities. Because the better-seeing eye is not necessarily the eye being operated on, for the purposes of this study, VA in the contralateral eye before PK was assumed to be no better than that in the eye being operated on. VA results were converted into utility scores using a third-order polynomial equation developed by Lansingh and Carter in a regression analysis between utility values and VA ( y = −0.0479 x 3 + 0.191 x 2 − 0.4233 x + 0.9128, where y is the utility value and x is the VA in logMAR units). The regression analysis in turn used utility and VA data correlations derived by Brown using a time trade-off method.
Complications
The 1-year incidences of postoperative complications for the patient cohort were obtained from the Singapore Corneal Transplant Study. These complications include wound dehiscence, infection (suture abscess, endophthalmitis, and microbial keratitis), glaucoma, astigmatism, cataract formation, allograft failure, and allograft rejection.
Although the brief timeline analyzed in the study precluded the direct observation of corneal graft failures, these are serious complications that may affect the utility gains associated with each procedure significantly. Baseline graft failure rates for each procedure were assigned based on estimates from the only long-term comparative study of graft survival between PK and DALK. Using that study as a guide, baseline graft failure rates for PK at 5, 10, 15, and 20 postoperative years were set at 9%, 10%, 30%, and 60%, respectively. Failure rates for DALK at the same time points were set at 3%, 5%, 10%, and 37%, respectively. For simplicity, graft failures for each period were assumed to occur halfway through the period. Failed grafts were not replaced in the analysis.
Medical Costs
Medical costs associated with the primary cornea surgeries and any subsequent treatments for complications were compiled from the Singapore National Eye Centre and the Singapore Eye Bank. Costs were taken as the private, unsubsidized rates charged by the National Eye Centre and Eye Bank, because these most fully represent the true costs of the interventions from a health system perspective. The 2 exceptions concern surgeon fees and facilities fees. Although the National Eye Centre charges equivalent fees for the 2 procedures, DALK on average takes twice as long to complete compared with PK (2 hours vs 1). Because a corneal surgeon can treat 2 patients with PK in the time it takes to treat 1 patient with DALK, the quality-adjusted life-year (QALY) gains under DALK are in effect more expensive than the QALY gains under PK. To account for this opportunity cost, the surgeon and facilities fees for DALK and PK were weighted by an adjustment factor so that the DALK charges were twice that for PK, while maintaining the average of both equal to the National Eye Centre charges. When multiple evidence-based treatment options were available for a particular complication, a weighted average of the treatment costs was taken based on their frequencies of use. The costs of treating complications associated with each primary procedure (PK vs DALK) similarly were weight averaged by their incidences and were added to the primary procedure costs to create the total expected cost for each procedure. All final costs are reported in Singaporean dollars unless otherwise noted and are rounded to the nearest 10s. Because the costs analyzed were accrued in the first year, no discounting of costs was performed.
Cost-Utility Analysis
We report the incremental cost per QALY gained, also known as the incremental cost-utility ratio for each procedure over a 20-year period. To account for the decline in VA (and thus utility gain) resulting from graft failures, postoperative VA was assumed to regress in a linear fashion over the lifetime of the failed grafts, from the initial postoperative value to the average preoperative VA of the patient cohort at the time of failure. For those grafts that did not fail, VA was assumed to be maintained at postoperative year 1 levels for 20 years. Total QALY gains for PK and DALK were then taken as the weighted average of the utility gains over the range of possible outcomes, with weights based on the 5-, 10-, 15-, and 20-year graft failure rates for each procedure. Utility gains were discounted at an annual 3% per year to account for the time value of outcomes, as recommended by the Panel on Cost Effectiveness in Health and Medicine. Incremental cost-utility ratios were calculated for PK and DALK by dividing the difference in costs over the difference in QALYs gained between each procedure and its less costly alternative. For PK, the less costly alternative for severe keratoconus was to do nothing because by definition, those with keratoconus severe enough to require corneal grafting are intolerant of less aggressive procedures such as spectacles or corneal rings. For DALK, the less costly alternative was PK.
One-Way Sensitivity Analyses
Because operative time for DALK and PK was responsible for most of the difference in initial costs between the 2 procedures, sensitivity analyses were conducted to examine its impact on the cost-efficacy of DALK relative to PK. The 20-year graft failure rate for DALK also was varied to examine the extent to which DALK could remain a cost-effective treatment for keratoconus at higher long-term graft failure rates. For the purposes of the analysis, the 5-, 10-, and 15-year graft failure rates were held constant, on the assumption that the clinical data behind those predicted rates are more robust. Lastly, the intraoperative complication rate during DALKa surgery was varied to assess the magnitude of its contribution to the relative cost-effectiveness of DALK.
Results
One hundred forty-eight corneal grafts, representing 128 individuals, were included in the study. Baseline demographic data are detailed in Table 1 . The 148 grafts consisted of 102 PK procedures and 46 DALK procedures (25 DALKa and 21 DALKm), with mean ages at operation of 26.7 and 29.3 years, respectively. The mean ages ( P = .14) and ethnic ( P = .33) and gender ( P = .36) distributions between the 2 groups were not significantly different. The mean preoperative VA of eyes treated with PK (1.53 logMAR units) was significantly worse than those treated with DALK (1.13 logMAR units; P < .001).
| Primary Keratoplasty Type | ||||
|---|---|---|---|---|
| Total (n = 148) | PK (n = 102) | DALK (n = 46) | P Value | |
| Mean age (SD), yrs | 27.3 | 26.7 (10.9) | 29.3 (9.2) | .14 a |
| Women, no. (%) | 50 (33.8) | 32 (31.4) | 18 (39.1) | .36 b |
| Race, no. (%) | .33 b | |||
| Chinese | 51 (34.5) | 37 (36.3) | 14 (30.4) | |
| Malay | 29 (19.6) | 17 (16.7) | 12 (26.1) | |
| Indian | 57 (38.5) | 42 (41.2) | 15 (32.6) | |
| Other | 11 (7.4) | 6 (5.9) | 5 (10.9) | |
| Preoperative visual acuity (logMAR) | 1.4 | 1.53 | 1.13 | < .001 a |
Comparative Effectiveness
The decision tree used in the analysis is shown in Figure 1 . The various event incidences, seen as percentages underneath each branch, are derived from the incidences observed in the patient cohort. The DALK procedure resulted in better 1-year VA outcomes than PK did, with expected VAs of 0.24 and 0.27 logMAR, respectively, although the difference was not statistically significant ( P = .901). These VAs correlate with utility values of 0.81 for PK and 0.82 for DALK ( Table 2 ).
| PK | DALK | |
|---|---|---|
| Preoperative utility | 0.56 | 0.56 |
| Postoperative utility (at 1 year) | 0.81 | 0.82 |
| Utility gain (at 1 year) | 0.25 | 0.26 |
| QALYs gained over 20 years a | 2.0 | 2.8 |
The mean preoperative VA across the study population was 1.41, which corresponds to a utility of 0.56. The first postoperative year utility gains for PK and DALK compared with no surgical treatment therefore are 0.25 and 0.26, respectively. Because these utility gains are based on VA values that are not statistically different, the possibility that the 2 procedures result in equivalent utility gains in the first year cannot be ruled out. Summing the discounted expected annual utility gains over 20 years after taking graft failures into account, however, results in different total expected utilities, with 2.0 and 2.8 QALYs for PK and DALK, respectively ( Table 2 ).
Costs
Total costs for each of the procedures are detailed in Table 3 . The cost for PK and DALK are $6830 and $9440, respectively, with most of the difference stemming from higher costs because of longer operative time for DALK. Complication rates and the costs of treating those complications are detailed in Table 4 . The incidence weighted cost of treating complications associated with PK is $670, whereas the cost associated with DALK is $480. Adding the costs of the primary surgeries to the complications costs yields the total costs associated with each procedure. For PK, the total cost is $7500; for DALK, the total cost is $9920.



