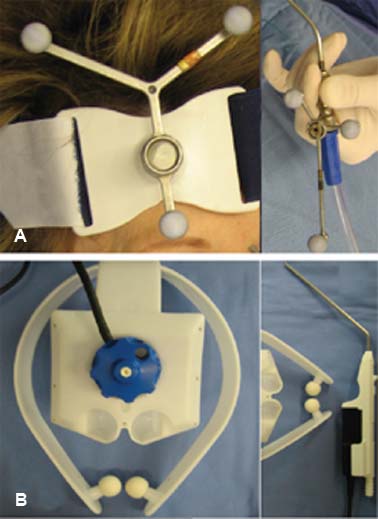
34 Core Messages • Image guidance is helpful in both planning and execution of endoscopic tumor resection. It should never replace a thorough understanding of anatomy and the patient’s disease. • Tracking of surgical instruments may be achieved either through optical or electromagnetic systems although there is no specific advantage of either with respect to surgical accuracy. • Optimal accuracy requires high resolution preoperative imaging coupled with careful intraoperative registration. Advances in imaging within the past two decades have led to the widespread use of image-guided surgery (IGS) in the management of sinonasal neoplasms. While the basic tenets of stereotactic surgery were introduced a century ago, current systems allow for detailed triplanar preoperative planning with multiple imaging modalities imaging as well as highly accurate intraoperative surgical navigation. Despite these advances, all systems may be associated with several types of errors and thus should be used only to augment sound clinical judgment and surgical expertise. While the first stereotactic devices were developed in the early 20th century, the coupling of radiography to surgical navigation did not occur until the 1940s.1 The introduction of computed tomography (CT) heralded significant improvements in image quality, eventually leading to a CT-guided frame-based system in 1976.2 With this improved surgical accuracy, the bulky and intrusive stereotactic frame provided an impetus to the development of early frameless systems in the 1980s.3 Since that time, the use of IGS has become widely accepted for both inflammatory and neoplastic sinonasal diseases and is currently endorsed by the American Academy of Otolaryngology-Head and Neck Surgery (Table 34.1).4 While all commercially available IGS systems use a unique method of tracking, they all function to monitor the position of an intraoperative localization device (ILD) within the surgical volume. Each system uses a proprietary software platform designed to construct a virtual data set from preoperative imaging. These data are subsequently correlated to the surgical volume through a process of registration.1 The ILD may be either fabricated as a surgical implement or directly attached to a preexisting instrument. Current systems use either optical or electromagnetic (EM) technology to track the ILD (Fig. 34.1).3 EM systems use a radiofrequency field to gather positional information through a receiver in the ILD. Drawbacks of this system include the potential for field distortion by ferromagnetic objects and the reliance on wires to communicate with the system.3 Some systems also require the patient to use the same headset from the preoperative imaging to the surgery, although some studies suggest that this has little effect on accuracy.5,6 In optical systems, the ILD is composed of an array of light-emitting diodes (active tracking) or reflective spheres (passive tracking), which are captured by an overhead camera and referenced to a set of markers on the headset worn by the patient. The principal drawback of this technology is that it requires an uninterrupted line of sight to the overhead camera.3 Before use, all IGS systems must be registered and calibrated. Registration involves the process of defining the relationship between fiducial position and the corresponding point in the data set volume. Calibration refers only to the confirmation of the relationship between the instrument tip and the ILD.3 Various registration paradigms include automatic, paired-point, and contour-based registration. Automatic registration uses a headset that incorporates the fiducial points in a fixed position, allowing the software platform to perform the registration automatically. In this system, changes in the headframe position result in diminished accuracy.3 In contrast, paired-point and contour-based registrations require manual mapping of the fiducial points that have been predefined in the virtual data set. In contour-based registration, this is followed by the acquisition of up to 500 points over fixed facial contours. The computer then calculates the registration by aligning these points with the virtual data set. Table 34.1 AAO-HNS Indications for Intraoperative Use of Computer-Aided Surgery
Image Guidance for Sinus Tumors
Historical Context
Operating Principles
System Setup
Revision sinus surgery |
Distorted sinus anatomy of development, postoperative, or traumatic origin |
Extensive sinonasal polyposis |
Pathology involving the frontal, posterior ethmoid, and sphenoid sinuses |
Disease abutting the skull base, orbit, optic nerve, or carotid artery |
Cerebrospinal fluid rhinorrhea or conditions where there is a skull base defect |
Benign and malignant sinonasal neoplasms |
AAO-HNS, American Academy of Otolaryngology-Head and Neck Surgery.
Preoperative Planning
One of the benefits of the IGS system is the ability to perform detailed preoperative planning with rapid scrolling through sequential high-resolution images in multiple axes. This allows the surgeon to map the location and extent of the neoplasm as well as the degree of involvement of adjacent anatomic structures.7
If the lesion involves the skull base, preoperative review aids in the selection of the optimal surgical corridor as well as in the estimation of the size of the expected defect (Fig. 34.2).1 The ability to perform three-dimensional (3D) reconstruction is particularly useful in characterizing morphologically complex lesions at the skull base. Rosahl et al8 reported that in a series of 110 patients, virtual 3D reconstruction enhanced surgical planning and assisted in targeting structures that would otherwise be hidden or obscured in the surgical field.
Advances in neuroimaging have also allowed for the incorporation of soft tissue data into the preoperative planning stage. CT alone does not allow for accurate delineation of soft tissue and neurovascular structures. This has been addressed by the advent of image-to-image registration software that allows magnetic resonance imaging (MRI) data to be coupled to CT images by using anatomic landmarks, thereby enabling the creation of a fusion image.9
The proximity of sinonasal lesions to major sinonasal or intracranial vasculature structures may also be visualized by using 3D CT angiography (3D-CTA). By using this protocol, images are captured as the contrast bolus fills the internal carotid system, thereby allowing for the simultaneous acquisition of the vascular and bony anatomy of the skull base.9 Leong et al10 performed 3D-CTA in 18 cases and found an accuracy of 2 mm or more and concluded that 3D-CTA provided an accurate assessment of the location of the internal carotid artery and its relationship to the surgical field. The incorporation of diffusion-weighted MRI and positron emission tomography scanning may also prove to aid in planning approaches to metabolically active tumors.1
Figure 34.1 Components of optical and electromagnetic tracking systems. (A) The optical system uses reflective spheres in both the headset and the instrument to monitor the intraoperative localization device. (B) In the electromagnetic systems, the headpiece transmits a radiofrequency signal that is detected by a receiver in the handpiece.