Hypopharyngeal and cervical esophageal malignancies are rare upper aerodigestive tract cancers that create a treatment challenge for head and neck surgeons. Although the hypopharynx and cervical esophagus are two separate subsites, the management of carcinoma in this area is, for the most part, similar. These tumors are characterized by multicentricity, submucosal spread, and early lymphatic metastasis. Patients typically present with advanced-stage disease and severe malnutrition that requires a multidisciplinary approach to their care. This multidisciplinary team must consist of the head and neck surgeon, radiation oncologist, medical oncologist, plastic surgeon, speech pathologist, and nutritionist to help guide the treatment to not only remove the disease but to maintain as much of the patient’s baseline function from before the tumor arose.
Despite these efforts from multiple fronts, the overall prognosis of patients with these tumors is poor. Advancements in surgical technique have been expanded to transoral techniques that may minimize the morbidity from major resections. Sequential chemotherapy and innovations with radiation techniques as part of clinical trials have resulted in larynx preservation without compromising survival (
1,
2,
3). Together, these improvements result in improved function without compromising diseasefree intervals. Enhanced microsurgical techniques with a diverse range of reconstructive flaps have created many options for surgical approaches in the primary and salvage setting. Thus, the development of a sound approach to patients with these tumors must include a thorough understanding of the anatomy, pathology, and clinical signs when preparing the most appropriate treatment.
ANATOMY
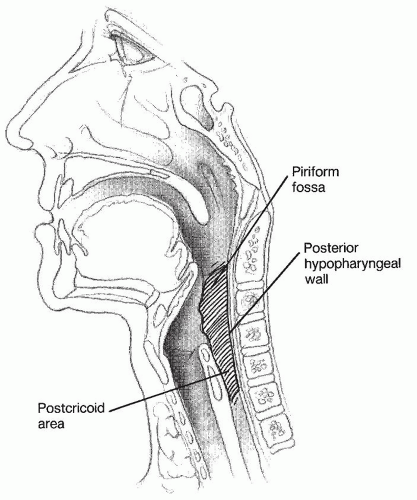
The hypopharynx begins at the level of the hyoid bone where it borders the oropharynx superiorly and funnels inferiorly to the cervical esophagus at the level of the inferior cricoid cartilage (
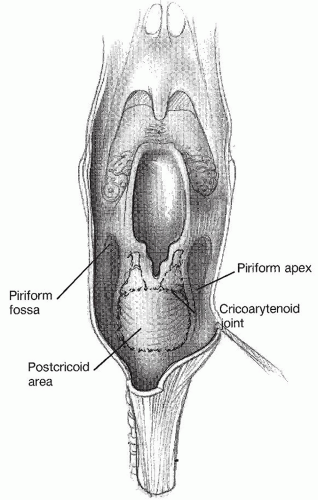
Fig. 122.1). Three subsites make up the hypopharynx: the pyriform sinuses laterally, the postcricoid area anteriorly, and the pharyngeal wall posteriorly (
Fig. 122.2). The paired pyriform sinuses lie in the form of inverted pyramids that begin at the pharyngoepiglottic folds superiorly and the apex blending into the cervical esophagus at its inferior extent. The postcricoid area represents the mucosa overlying the posterior portion of the cricoid ring. It extends from the arytenoid cartilage to the inferior border of the cricoid cartilage. The proximity of the pyriform sinuses and the postcricoid area to the larynx can lead to direct invasion of tumors of these regions into the paraglottic space and laryngeal skeleton. The posterior pharyngeal wall is the portion of the hypopharynx overlying the vertebrae. Tumors of this area can directly invade the potential retropharyngeal space, paraspinous muscles, and prevertebral fascia, making complete resection extremely difficult.
The lining of the hypopharynx is stratified squamous epithelium that overlies a submucosal loose areolar tissue layer, followed by a muscular layer made up of the posterior cricoarytenoid muscles anteriorly and the middle/inferior pharyngeal constrictors posteriorly and laterally. These structures are enclosed by the buccopharyngeal fascia. The muscular layer is notable because tumor extension from the posterior cricoid mucosa can invade into the posterior cricoarytenoid muscles causing vocal cord fixation, and the inferior constrictor muscles blend into the cricopharyngeus muscle inferiorly where the potential area weakness, Killian triangle, is located. Although this region is noted for the site of pharyngeal diverticula, tumor extension to the prevertebral space is also possible.
The vascular supply of the hypopharynx comes from the external carotid system and includes branches of the superior thyroid artery and ascending pharyngeal and lingual arteries. Venous drainage mirrors the arterial system in addition to the prevertebral venous plexus.
Sensory innervation to the hypopharynx is derived from branches of the glossopharyngeal and vagus nerves via the pharyngeal plexus and the internal branch of the superior laryngeal nerve. The latter nerve pierces the thyrohyoid membrane and joins with the vagus nerve where fibers coalesce with branches of Arnold nerve to the external auditory canal. This connection can result in the referred otalgia seen in many patients with hypopharyngeal pathology. Motor innervation to the pharyngeal constrictors comes from the pharyngeal plexus, while the posterior cricoarytenoid muscles are innervated by the recurrent laryngeal nerve.
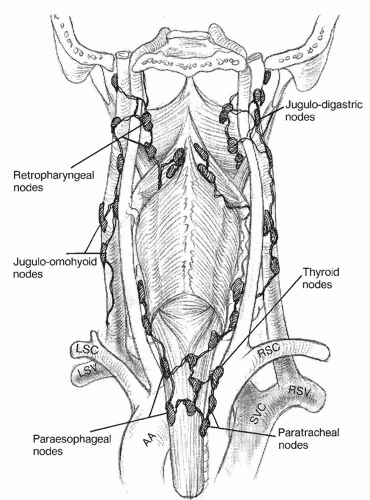
The hypopharynx has an extensive network of lymphatics. Drainage can occur laterally to the jugulodigastric nodes (
Fig. 122.3). The second drainage pathway is posterior to the retropharyngeal nodes and can extend as high as the skull base in the nodes of Rouviere. Inferiorly, nodal metastases can occur in the paratracheal and paraesophageal nodes (
4). Bilateral drainage is common, especially for lesions located in the medial pyriform and posterior pharyngeal wall.
The cervical esophagus begins at the inferior extent of the hypopharynx and extends to the thoracic inlet. It can vary in length depending on the length and angle of the neck. The lining of the cervical esophagus is stratified squamous epithelium that covers a submucosal layer that is rich in lymphatic vessels. The muscular layer contains the bilayered esophageal muscles, with an internal circular layer and external longitudinal layer. The serosa is a continuation of the buccopharyngeal fascia. Arterial supply to this region comes from the inferior thyroid circulation off the thyrocervical trunk, and venous drainage occurs via the inferior thyroid veins. Innervation is derived from the recurrent laryngeal nerve and sympathetic chain. Lymphatic drainage occurs in the paraesophageal and paratracheal nodes, which can then spread to the superior mediastinum or lateral cervical nodes.
EPIDEMIOLOGY
In the United States and Canada, two large studies of national cancer registries have shed light on the presence of hypopharynx carcinoma in the population. In a review of the National Cancer Data Base (NCDB), a hospitalbased cancer registry that covers the entire United States, hypopharyngeal carcinomas account for approximately 3% to 4% of all head and neck cancers, which account for approximately 3% of all cancers (
5,
6). Of note, this report also showed that the percentage of hypopharyngeal carcinoma cases decreased nearly 30% from 2000 to 2004 compared to the 1990 to 1994 (
5). This number has been attributed to the decrease in cigarette smoking among
Americans (
7). A previous review of the NCDB revealed that 77% of patients with hypopharynx carcinoma present with stage III or IV disease (
8). Of note, the survival in the most recent update of the NCDB showed hypopharynx carcinoma to have the worst survival of any primary head and neck mucosal surface malignancy, with a 5-year overall survival around 35% (
5).
A review of the data pooled from multiple Canadian cancer registries from 1990 to 1999 revealed that hypopharynx cancer is 5% of all head and neck cancers (
9). In Canada, this represents an incidence of approximately 0.8 new cases per 100,000 persons/year. Peak incidence was seen in the sixth and seventh decades of life with 80% of cases being male. Similar to the NCDB data, 74% of patients presented with advanced-stage disease, and survival was 35% at 5 years.
Cervical esophageal carcinoma is difficult to assess, as they are very rare and many times mislabeled as thoracic esophageal carcinoma or hypopharyngeal carcinoma as the tumors grow. They have been reported to account for between 2% and 10% of all esophageal carcinomas (
10). In 2010, esophageal carcinoma represented 5% of all digestive system cancers, estimated at 16,640 new cases with 14,500 deaths, thus explaining the paucity of data with cervical esophageal carcinoma (
6).
ETIOLOGY
Tobacco smoking and alcohol are well-established risk factors for upper aerodigestive tract tumors. In a recent study reviewing smoking in Europeans, the association with active tobacco smoking was strongest for cancer risk of hypopharynx compared to the oral cavity, oropharynx, and esophagus (
11). Alcohol intake appears to be more common in patients with hypopharyngeal cancer when compared with laryngeal cancer and is considered an independent risk factor in the development of hypopharyngeal carcinoma when controlled for smoking (
12). Daily consumption of alcohol has been found to increase the risk of hypopharyngeal carcinoma by 2.2 (
13). The mechanism of carcinogenesis by alcohol is unclear. Many believe it has a direct carcinogenic effect on the hypopharyngeal mucosa, potentiates tobacco, or is related to the poor nutritional status in alcoholics that contributes to the development of hypopharyngeal carcinoma (
14). The use of smokeless tobacco products has also been implicated as a cause of hypopharynx cancer increasing the risk by 4.6 in a study from India (
13).
Another causative factor for hypopharyngeal carcinoma that is specifically associated with the postcricoid mucosa is Plummer-Vinson syndrome. This syndrome is characterized by dysphagia, iron deficiency anemia, and hypopharyngeal webs. Chronic irritation of the webs is thought to be the causative factor in the progression to carcinoma.
PATHOLOGY
Squamous cell carcinoma is the most common histology seen in the hypopharyngeal and cervical esophageal cancer, representing over 95% of cases (
9). Three variants are commonly seen in the hypopharynx. The first is basaloid squamous cell carcinoma, which has a more aggressive course. Lymphoepithelial carcinomas are similar in nature to nasopharyngeal carcinoma. Adenosquamous carcinoma is the third variant with a similar aggressive course to basaloid squamous cell carcinoma. The remaining 5% of histologies consist of adenocarcinomas, thought to arise from ectopic gastric mucosa, lymphomas, and sarcomas (
8).
Patterns of Spread
A distinguishing feature of hypopharyngeal and cervical esophageal carcinomas is submucosal extension of the tumors. An understanding of the patterns of spread of tumors begins with the site of origin. Most hypopharynx cancers are located in the pyriform sinuses, followed by the posterior pharyngeal wall, and then the postcricoid mucosa (
Table 122.2) (
23). Submucosal extension is more common in the inferior hypopharynx and cervical esophagus, thought to be caused by the rich submucosal lymphatic network seen at the pharyngoesophageal junction. Satellite lesions are also a characteristic of these tumors; however, it is difficult to determine if these tumors are separate primary tumors or metastatic lesions. The presence of submucosal extension, especially in the inferior hypopharynx and cervical esophagus, must be taken into account when assessing margins during surgical resection. It has generally been recommended that 3-cm margins be taken in the cervical esophagus and inferior hypopharynx, 2 cm laterally and 1.5 cm along the superior margin.
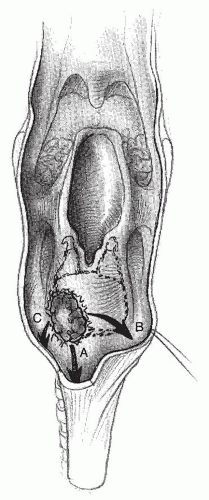
Tumors of the pyriform fossae can spread laterally to the thyroid cartilage and soft tissues of the neck. Medial extension of these tumors will involve the larynx and the paraglottic space (
Fig. 122.4). Postcricoid tumors tend to grow circumferentially. Inferior extension via submucosal spread involves the cervical esophagus. Anterior extension spreads to the cricoarytenoid joint and posterior cricoarytenoid muscles causing vocal cord immobility (
Fig. 122.5). Advanced cases of hypopharyngeal carcinoma can present with direct invasion into the thyroid gland (
24).
Lymph node metastases are common in hypopharyngeal carcinoma with 64% to 90% of patients presenting with nodal disease and bilateral disease seen in 8% to 16% of cases (
9,
25,
26). Regional metastasis occurs in levels II to IV, with rare involvement of levels I and V (
25,
27). Invasion of the pyriform apex (20%), postcricoid mucosa (57%), and subglottis is associated with metastasis in the paratracheal and paraesophageal nodes (
4,
28,
29). Mediastinal nodal involvement is noted in 73% to 80% of T4 hypopharynx cancers and 33% to 62% of cervical esophageal carcinomas of all stages (
30,
31). Forty-three percent of cervical esophageal carcinomas are noted to have paratracheal and paraesophageal nodal disease (
29). Retropharyngeal nodal disease is seen in 20% to 50% of patients with hypopharyngeal and cervical esophageal cancers (
32,
33,
34). The significant percentage of nodal metastasis in hypopharyngeal and cervical esophageal percentage
requires treatment of the nodal basins that are specific to each subsite of these regions.
Staging
The seventh edition of the American Joint Committee on Cancer Staging Manual has similar TNM staging compared to the sixth edition manual. This staging system facilitates the study of prognostic variables as related to the volume of local, regional, and distant disease. T4 tumors are divided into T4a (resectable; advanced local disease) and T4b (low likelihood of resection free margins; very advanced local disease). This has led to stratification of stage IV disease into IVA (moderately advanced local/regional disease), IVB (very advanced local/regional disease), and IVC (distant metastatic disease). The TNM staging for the hypopharynx is as follows:
TX: Primary tumor cannot be assessed.
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1: Tumor limited to one subsite and ≤2 cm in greatest dimension
T2: Tumor invades more than one subsite of the hypopharynx or adjacent site, or measures greater than 2 cm but not ≤4 cm in greatest dimension without larynx fixation.
T3: Tumor greater than 4 cm in greatest dimension or with fixation of the hemilarynx
T4a: Tumor invades thyroid/cricoids cartilage, hyoid bone, thyroid gland, esophagus, or central compartment soft tissue.
T4b: Tumor invades prevertebral fascia, encases carotid artery, or involves mediastinal structure.
NX: Regional lymph nodes cannot be assessed.
N0: No regional lymph node metastasis
N1: Metastasis in a single ipsilateral lymph node less than 3 cm in greatest dimension
N2a: Metastasis in a single ipsilateral lymph node ≥3 cm but less than 6 cm in greatest dimension
N2b: Metastasis in multiple ipsilateral lymph nodes, all less than 6 cm in greatest dimension
N2c: Metastasis in bilateral lymph nodes, all less than 6 cm in greatest dimension
N3: Metastasis in lymph node is ≥6 cm in greatest dimension
MX: Distant metastasis cannot be assessed.
M0: No distant metastasis
M1: Distant metastasis
The TNM staging system for the cervical esophagus is different from the hypopharynx. T stage is based on tumor depth of invasion, and N stage is based on the number of nodes present. The staging system has been updated since the sixth edition and is as follows:
TX: Primary tumor cannot be assessed.
T0: No evidence of primary tumor
Tis: Carcinoma in situ
T1: Tumor invades lamina propria or submucosa.
T2: Tumor invades muscularis propria.
T3: Tumor invades adventitia.
T4: Tumor invades adjacent structures (hypopharynx, thyroid gland, carotid sheath).
NX: Regional lymph nodes cannot be assessed.
N0: No regional nodal metastasis
N1: Regional lymph node metastasis 1 to 2 nodes
N2: Regional lymph node metastasis in 3 to 6
N3: Regional lymph node metastasis in greater than 7 nodes
MX: Distant metastasis cannot be assessed
M0: No distant metastasis
M1: Distant metastasis
Molecular Staging
Hypopharyngeal and cervical esophageal cancers have distinct clinical behavior compared to other head and neck cancers; however, no specific molecular markers have paralleled this behavior. One study found an association between the stem cell markers, Oct4 and Sox2, and hypopharyngeal cancer progression, but the authors in this study did not compare their results to other head and neck sites (
35). The recent interest in human papillomavirus (HPV) in head and neck carcinoma has not translated to hypopharyngeal carcinoma. A study from M.D. Anderson Cancer Center revealed the presence of HPV correlated with poor prognosis in hypopharyngeal carcinoma (
36). Another study from Belgium identified no difference in survival between HPV-positive and HPV-negative hypopharyngeal carcinomas despite the presence of HPV in 74% of patients (
37). This may represent the molecular heterogeneity within hypopharyngeal carcinoma that makes HPV status irrelevant, but future study is warranted.