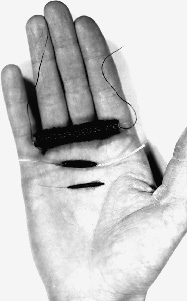
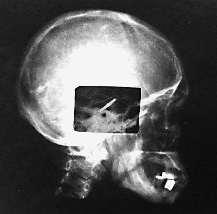
1 Marc D. Eisen The cochlear implant has created a paradigm shift in the treatment of sensorineural hearing loss. It has had great impact in the brief time it has been available. In less than four decades, the cochlear implant progressed from the first attempts to elicit hearing via direct electrical stimulation of the auditory nerve to a commercially available device that has restored varying degrees of hearing to tens of thousands of deaf patients. Several themes that pervade the implant’s history are widely applicable to the development of other neural prostheses. For one, the implant’s development was truly an interdisciplinary effort. Significant contributions came from members of fields as diverse as engineering, otology, audiology, auditory neurophysiology, psychoacoustics, and industry. The interaction among these players was not always harmonious, but the strife yielded synthesis and progress. Another theme is the courage of a few clinicians to risk their reputations and eschew scientific dogma in the hope of helping the patients that sought their care. Lastly is the theme of patients being willing to take substantial risks in serving as research subjects, sometimes without any promise of individual medical gain. This chapter does not aim to be exhaustive in mentioning each and every contributor or contribution that occurred during the implant’s early development. Rather, it discusses in depth a selection of events and characters that—with the aid of the “retrospectroscope”—exemplify these themes and demonstrate a progression of events leading toward a device that allows the patient who has lost all hearing to regain the ability to converse on the telephone, and one that allows the deaf child to develop near-normal speech production and understanding. The story of the implant’s development is parsed into several periods. The first began in 1957 and continued throughout the 1960s. This was the period of pioneering and experimentation. The second period in the 1970s was a time of feasibility study, exploring whether the implant safely stimulated the auditory pathway and elicited useful hearing. The third period led to the development of a commercially viable multielectrode cochlear prosthesis. Several discoveries made during the first half of the 20th century were not directly related to electrical stimulation of the cochlear nerve, but were influential on the early development of the cochlear implant and therefore warrant mention. These include Homer Dudley’s work on the synthesis of speech and his “vocoder,” Glenn Wever and his discovery of the cochlear microphonic, and S. S. Stevens and coworkers’ description of electrophonic hearing. Homer Dudley was a researcher at the Bell Telephone Laboratories in New York. He described and demonstrated in 1939 a real-time voice synthesizer that produced intelligible speech using circuitry designed to extract the fundamental frequency of speech, the intensity of its spectral components, and its overall power. The spectral components were extracted with a series of 10 band-pass filters covering the frequency range of speech.1 He named the synthesizer the “vocoder,” a compressed version of “coding the voice.” The operating principles of the vocoder for condensing speech into its principal components formed the basis of early speech processing schemes for multichannel cochlear implants. In 1930, Wever and Bray2 recorded and described the electrical potentials in the cochlea that faithfully reproduced the sound stimulus. This phenomenon became known as the “Wever–Bray effect.” The source of these measured potentials was initially incorrectly assumed to represent auditory nerve discharges. This theory of the origin of these potentials would be equivalent to the “telephone” theory of hearing, referring to the analog representation of the voice carried along the “cable” of the auditory nerve as it would along the wires of a telephone line. In truth, what Wever and Bray were recording was not a response of the cochlear nerve, but the cochlear microphonic produced by the outer hair cells in the cochlea. Regardless of the ultimate dismissal of the telephone theory of hearing, it inspired several of the earliest pioneers of the cochlear implant. S. S. Stevens3 and his colleagues classically described in the 1930s the mechanism by which the cochlear elements respond to electrical stimulation to produce hearing. This mechanism was coined “electrophonic hearing.” We now know that electrophonic hearing results from the mechanical oscillation of the basilar membrane in response to voltage changes. The primary tenet of their description was the requirement that the cochlea be intact. Prior to 1957, efforts to stimulate hearing electrically were performed on subjects with at least partially functioning cochleae. In these subjects, therefore, responses could be accounted for by electrophonic hearing rather than direct nerve stimulation. Furthermore, the earliest cochlear implant efforts had the burden of proving that they were directly stimulating the cochlear nerve rather than eliciting electrophonic hearing. Although numerous attempts to treat deafness with electricity have been reported over the past several centuries,4 the first reported direct stimulation of the cochlear nerve for the purpose of generating hearing appeared as recently as 1957 with the work of André Djourno and Charles Eyriès. Despite the revolutionary impact that the cochlear implant has had on all auditory disciplines, these beginnings in Paris received little attention. André Djourno (1904–1996) received degrees in both science and medicine, yet he devoted his career to science. His early endeavors were studying the electrophysiology of the frog peripheral nerve.5,6 He then ventured into more medical applications of electricity. Several of Djourno’s earlier innovations reflected his inventiveness: a device to measure the pulse continuously,7 high-frequency electrical stimulation to remove metal fragments from bones,8 and the use of electroencephalography (EEG) to study narcolepsy.9 Perhaps the most prescient development from this period was artificial respiration utilizing direct phrenic nerve stimulation.10 Although this innovation did not reach widespread clinical implementation, it demonstrated Djourno’s interest in neural prostheses. Djourno focused the next phase of his career on fabricating and testing implantable induction coils to be used for “telestimulation,” or stimulation through inductive coupling without wires. Djourno assembled these induction coils himself and called them “microbobinages,” as the coils wound with wire resembled small spools of thread (Fig. 1–1). Both the active coil and the ground electrode were implanted under the skin of an animal, and stimulation was transcutaneous (rather than percutaneous). The implantable coils were first used to stimulate the sciatic nerve and thus trigger a jump behavior in rabbits. Djourno studied numerous aspects of telestimulation, including electrode biocompatibility (he described using one of the first bioresistant resins, araldite, for example, to coat the electrodes11). He addressed the effect of stimulus frequency on muscle contraction, and he found that with higher frequency stimuli, muscles would not contract, whereas with lower frequency stimuli muscle contraction was painful. Djourno found the “right” stimulus frequency between 400 and 500 Hz. Because this frequency was within the speech range, he began to use the analog signal of his own voice as the telestimulating stimulus.12 Triggering a nerve with his voice may well have contributed to the idea of stimulating the cochlear nerve to restore hearing. Figure 1–1 Example of implantable induction coils (“microbobinages”) assembled by Djourno in his laboratory. Induction coils like the ones shown here were used in various applications, including the stimulation of the auditory pathway. The hand holding the coils gives perspective to the implant’s size. (Courtesy of John Q. Adams Center for the History of Otolaryngology–Head and Neck Surgery, Alexandria, VA.) Djourno also addressed the safety of repetitive stimulation on tissue, demonstrating that the sciatic nerve from an implanted rabbit, when examined histologically and grossly, showed no changes after 2 years of repetitive stimulation.13 Throughout this time Djourno revealed little interest in hearing. He recognized, however, the potential of using the microbobinages to stimulate the auditory system, as he and Kayser noted the possibility of “treating deafness” as a potential application in a 1954 publication.12 Charles Eyriès (1908–1996) completed his training in otolaryngology in Paris in the early 1940s. Clinically, Eyriès earned early recognition for his description of a procedure to treat ozena, or atrophic rhinitis, by placing implants underneath the nasal mucosa to decrease the caliber of the nasal passages.14 This procedure became known in the French literature as the “Eyriès operation.” Eyriès was named chief of otorhinolaryngology and head and neck surgery at L’Institut Prophylactique in 1953, which has since been renamed L’Institut Arthur Vernes. Although primarily a clinician, he had research interests in neuroanatomy and embryology of the facial nerve, and he wrote about surgical facial nerve repairs.15 Eyriès had shown little interest in hearing at this point in his career and had never worked with Djourno, although he knew of Djourno because both he and Djourno had laboratories in the medical school associated with the hospital. As the local expert on facial nerve repairs, Eyriès was asked in February 1957 to provide a consultation for a rather unfortunate patient—a 57-year-old man who suffered from large bilateral cholesteatomas. A right-sided temporal bone resection was performed 5 days prior to the consultation and an extensive left temporal bone resection was performed several years earlier. Both procedures involved ablation of the labyrinth and facial nerve sectioning. As a result, the patient was left with bilateral deafness and bilateral facial nerve paralysis. Eyriès was consulted to consider a facial nerve graft for reanimation.16 On examination, Eyriès found that the caliber of the patient’s remaining nerve was too small to support a local nerve transfer. Eyriès therefore embarked on a search for appropriate graft material. He went to the medical school seeking cadaverous material, where he met Djourno, who offered to help and suggested stimulating hearing at the same time. Although Eyriès was primarily interested in his patient’s facial reanimation, he agreed to implant an electrode into the patient at the time of surgery. Eyriès’ justification for agreeing to the implantation was that the cavity was already exposed and the patient had nothing to lose in having the extra procedure.16 From Djourno’s standpoint, the patient was deaf and begging to escape from the silence that haunted him, and Djourno was fascinated by the opportunity to telestimulate the auditory system.17 The procedure took place on February 25, 1957. Eyriès performed the right-sided facial nerve graft using fetal sciatic nerve as the graft material, which purportedly proved to be successful. At the time of surgery, the proximal cochlear nerve stump was found to be significantly shredded. Djourno and Eyriès chose to seat the active electrode into the remaining stump and place the induction coil into the temporalis muscle. A postoperative lateral skull film confirmed its placement (Fig. 1–2). Some testing was done intraoperatively. The stimulus waveforms included bursts of a 100-Hz impulse signal administered 15 to 20 times per minute, low-frequency alternating current, and the analog signal of words spoken into a microphone. The patient described detecting auditory sensations. Several qualitative observations were made: the patient’s discrimination of intensity was good, frequency discrimination was poor, and no speech recognition was evident. The patient underwent an extensive postoperative rehabilitation with the implant under the guidance of the speech therapist. Over the ensuing months more complex stimuli were administered, and the patient was able to differentiate between higher frequency (described as “silk ripping”) and lower frequency (described as “burlap tearing”) stimulation. He appreciated environmental noises and several words, but could not understand speech. The publication that resulted from this work is the seminal citation for direct cochlear nerve stimulation.18 Several months later, during testing, the electrode suddenly ceased to function. Djourno and Eyriès brought the patient to the operating room to investigate. They found that a solder joint connecting the wires to the ground electrode embedded into the temporalis muscle had broken, and the implant was replaced. The second implant, however, suffered the same fate. Eyriès held Djourno responsible for the broken electrodes and refused to perform a third implantation.19 The falling out between the two men over this problem was the end of Eyriès involvement in the project. After this event, he and Djourno rarely conversed for the rest of their lives. Figure 1–2 Lateral skull film of Djourno and Eyriès’ first implant following surgery. The coil has been embedded in the temporalis muscle, whereas the electrodes were placed near the remaining stump of the cochlear nerve. (Courtesy of John Q. Adams Center for the History of Otolaryngology–Head and Neck Surgery, Alexandria, VA.) This was not quite the end of the story for Djourno, however. He went on to address several aspects of hearing applicable to electrical stimulation. For one, he examined the oscillographic representation of spoken words in an effort to give deaf patients a visual representation of speech that they could use for biofeedback when learning to speak.20 After the first implant effort, a colleague approached Djourno to enter into a business venture to develop the implant. The colleague proposed that in exchange for an exclusive arrangement on the project, he would provide Djourno with the financial and engineering support of industry.17 Djourno was always an academic idealist, and he did not believe in profiting from his discoveries. Furthermore he detested industry and would have no part in granting exclusivity. As a matter of principle, Djourno chose to do another implant with a different otolaryngologist, Roger Maspétiol.17 This second patient, deafened from streptomycin ototoxicity, was implanted with an electrode near the promontory, rather than within the temporal bone. The patient showed little enthusiasm for her device, and she was lost to follow-up only a few months after it was implanted. Djourno subsequently lost funding for further implant work.21 This signaled the end of Djourno’s participation in developing an auditory prosthesis. The legacy of the Djourno and Eyriès work was sustained despite the abrupt departure of the two men. Claude-Henri Chouard, who was a student in Eyriès’ laboratory working on the facial nerve, resumed work on the cochlear implant several years later. Chouard22 was instrumental in developing one of the first functional multichannel implants, and he credits Charles Eyriès as his major source of inspiration. Although Djourno and Eyriès’ implantation on February 25, 1957 is typically credited as the first cochlear implant, a closer evaluation of the patient’s anatomy raises the question of whether the cochlear nerve or the auditory brainstem was stimulated by the implanted electrodes, as wallerian degeneration may have destroyed the cochlear ganglion cells.23 Whether Djourno and Eyriès stimulated the cochlear nerve or the cochlear nucleus should not overshadow the significance of their work. Electrical methods to treat deafness had been described by numerous practitioners for almost 200 years prior to 1957, beginning with the classic work of Alessandro Volta in the late 18th century.4 These previous efforts, however, were either aimed at treating deafness with therapeutic electrical stimulation, or they were examples of electrophonic hearing. Dissemination of the work of Djourno and Eyriès was slow to arrive in America. This is likely attributable to the fact that their publication appeared only in the French medical literature. Additionally, of the pair, the more likely to present his work among clinicians was Eyriès, as he was an otolaryngologist. Eyriès’ demonstrated little enthusiasm for the project, however, and his interest was short lived. Djourno was a physiologist rather than a clinician, making interaction between him and American otolaryngologists less likely despite his continued interest. Word of their work reached William House in California serendipitously when his patient brought a summary of the Djourno and Eyriès work written in English to him sometime around 1959.24 The piece was optimistic regarding electrical stimulation to replace hearing, and House was inspired. William House was a dentist-turned-otologist who began working with his brother Howard House at the Otologic Medical Group in Los Angeles upon completion of his residency in 1956. Early in his career he had already made significant contributions to otology and neurotology, including the facial recess approach. He was working at the time on the middle fossa approach to the internal auditory canal in collaboration with John Doyle, a neurosurgeon who also practiced at St. Vincent’s Hospital in Los Angeles.25 House and Doyle first sought to record the cochlear nerve response to sound when the nerve was exposed during the middle fossa approach for vestibular neurectomy as a treatment for Meniere’s disease. Specifically, they sought to record the nerve output associated with tinnitus.26 They relied on Doyle’s brother, James Doyle, an electrical engineer, to address the technical challenge of recording such signals intraoperatively. The nerve output was recorded, but no tinnitus was observed. Successful recordings of sound-induced potentials from the cochlear nerve, however, inspired stimulating the nerve with similar waveforms to restore hearing. House and Doyle first attempted electrical stimulation to elicit hearing during stapes surgery by placing a needle electrode on the promontory or into the open oval window. An ear speculum inserted into the external auditory canal served as a ground lead. With square wave stimuli, patients reported hearing the stimulus without discomfort, dizziness, or facial nerve stimulation. These responses were sufficient to encourage House and Doyle to implant a patient with a hard-wire device. The first willing subject was a 40-year-old man with severe otosclerosis and deafness. Promontory stimulation of the right ear on January 5, 1961, revealed consistent responses. On January 9, therefore, a gold wire electrode was inserted under local anesthesia through a postauricular approach into the round window. The wire was brought out through the postauricular skin.27 The patient reported hearing the electrical stimuli, but he had poor loudness tolerance. Several weeks later the wire was removed. A second patient was also implanted in January 1961. The woman had deafness, tinnitus, and vertigo associated with congenital syphilis, and she was brought to the operating room for a vestibular neurectomy through the middle fossa approach. During the procedure, a single gold wire electrode was placed through the middle fossa approach into scala tympani at the basal part of the cochlea. The wire was brought out through a skin incision. The patient described hearing the square wave stimulation upon waking from anesthesia. Over the ensuing days, the current intensity required to elicit a response increased. For fear of infection or edema, the wire was removed. With the first patient’s encouraging responses, and with the hope of producing discrimination of higher frequencies, House and Doyle decided to reimplant him with a five-wire-electrode array inserted through the mastoid facial recess and round window. The electrode array was attached to a more permanent electrode induction system seated in the skull. Over a several-week testing period, the patient’s intensity requirement increased and his post-auricular skin began to swell. This device was also removed for risk of infection. Worries of biocompatibility of materials ensued. The theoretical basis for the multiple electrode design was to spread high-frequency stimuli among spatially separated electrodes. By stimulating different subpopulations of auditory nerve fibers at rates slower than their refractory period, they thought, summation among the subpopulations would purportedly yield an overall high-frequency response along the whole nerve. This implant design and its theoretical basis became the foundation for an early cochlear implant patent application submitted by James Doyle and Earle Ballantyne in 1961. The patent was not granted until 1969.28 Despite being founded on what has since been shown to be an erroneous theory of electrical stimulation, the patent was ironically prescient in its statement that a 16-channel unit would be necessary for implant patients to be able to converse on the telephone. Word of the two implanted patients reached the lay press. The brief articles were overly optimistic in their descriptions of an “artificial ear,” going so far as to announce that “surgical implantation of a transistorized device designed to restore hearing of deaf persons is scheduled within 30 days.”29 The effect of the lay press publications were calls from deaf patients to Drs. House and Doyle seeking a cure for their deafness, as well as investors seeking to cash in on emerging medical technology. Dr. House recognized the danger in such publicity, and broadcasting of the implant work became an area of considerable strife between the Doyles and House. Disagreement over how aggressive to proceed with the implant, given the initial bio-incompatibility problems, opened an irreparable rift between House and the Doyles that brought an end to their collaboration. House had a very busy otologic practice, and implant development took a low priority for several years to follow. The Doyles, on the other hand, continued to experiment, implanting numerous subjects. They collaborated with the Los Angeles otolaryngologist Frederick Turnbull, whose office was used for most of the testing. They reported their results in local30 and national31 forums, reporting optimistically that electrical stimulation could yield speech perception, but not offering systematic testing or analysis. The Doyles ceased their investigations in 1968 due to a lack of research funding.26 F. Blair Simmons had worked as a research associate in the laboratory of S. S. Stevens at Harvard as a medical student and then with Robert Galambos at the Walter Reed Institute prior to his residency in otolaryngology at Stanford University. Simmons was an assistant professor in Stanford’s Division of Otolaryngology in 1962 for less than a month before he was presented with an unexpected opportunity to stimulate the cochlear nerve intraoperatively. The patient was an 18-year-old who had developed a recurrence of a cerebellar ependymoma that manifested itself as mild hearing loss. Exploratory craniotomy under local anesthesia was planned, and the cochlear nerve would be exposed during the procedure. Prior to surgery, Simmons discussed stimulating the patient’s cochlear nerve electrically. The patient agreed to the intraoperative testing and to a preoperative auditory training session. During the awake craniotomy, the patient was asked to describe what he heard when a bipolar electrode was used to stimulate the exposed cochlear nerve with 100-μs square wave pulses. The patient described auditory sensations and was able to discriminate stimulation frequencies up to 1 kHz.32 Simmons’s first implanted device was then placed 2 years later, in 1964. This second subject was a 60-year-old man who had been unilaterally deaf for several years, and whose better hearing ear became deaf. He also suffered from retinitis pigmentosum and had associated severe sight disability. Despite being made fully aware that implantation would likely fail and very likely yield no useful hearing, the subject agreed to undergo the implantation. Local anesthesia was used, and the promontory exposed through a postauricular transmeatal approach and elevation of a tympanomeatal flap. A 2-mm cochleostomy and then subsequent 0.1-mm drill hole into the modiolus were performed. A partial mastoidectomy was also performed, and the middle ear entered anterior to the facial nerve by removing the incus. A six-electrode array was placed through the mastoid opening into the epitympanum and then through both the cochleostomy and the modiolar opening to a depth of 3 to 4 mm. The electrodes were attached to a plug that was then secured to the mastoid cortex. Psychoacoustic testing was performed both at Stanford and at Bell Laboratories in New Jersey.33 Unfortunately, the combination of disabilities made psychophysical testing very challenging. Simmons’s conclusions from these experiences were pessimistic regarding the future of implantation. He estimated the likelihood that electrical stimulation of the auditory nerve could ever provide a clinically useful means of communication to be “considerably less than 5%.”34 Human implantation at Stanford was postponed until further animal testing could prove its utility. With the advancements in pacemakers and ventriculoperitoneal shunts in the late 1960s, William House resumed an interest in cochlear implantation with more confidence in the safety and efficacy of indwelling devices (Fig. 1–3). House was working with a talented engineer named Jack Urban, a collaboration best known for several influential developments in neurotologic instruments. House and Urban aggressively pursued implanting their single-channel device in human patients. As much as any other parameter, the durability and safety of the device was on House’s mind with this group of patients. Of several patients implanted in 1969, one required having his implant removed due to tissue rejection, and another was lost to follow-up. A third patient, however, was Charles Graser, who became a long-term experimental subject. In Graser, House found that stimulation levels and results remained stable over the course of years. This gave credence to the safety of electrical stimulation.
History of the Cochlear Implant
♦ Precursors
The Vocoder
The Cochlear Microphonic
Electrophonic Hearing
♦ Pioneers (1957 to 1973): André Djourno and Charles Eyriès
♦ Early Developments in America
Los Angeles
Stanford University
House Resumes Work
Ento Key
Fastest Otolaryngology & Ophthalmology Insight Engine




