8 Histology of Pituitary Tumors
 Normal Pituitary Gland
Normal Pituitary Gland
The human pituitary gland is a small bean-shaped organ located at the base of the brain and is situated within the confines of the sella turcica. It measures 10 mm in length, 5 to 10 mm in height, and 10 to 15 mm in width and has an average weight of 600 mg. The pituitary gland on average weighs more in females and increases in weight during pregnancy.1 Considered the master endocrine organ, it functions along with the hypothalamus in the regulation of other endocrine organs. The pituitary is attached to the hypothalamus by the pituitary stalk and is composed of two anatomical and functionally distinct parts: the anterior lobe (adenohypophysis) and the posterior lobe (neurohypophysis).
The anterior lobe is embryologically derived from Rathke’s pouch and constitutes the majority of the gland (80%). It consists of three components: the pars distalis, pars intermedia, and pars tuberalis. The pars distalis constitutes the majority of the anterior lobe, and histologically five different cell types can be identified in the pars distalis: growth hormone (GH)-producing somatotrophs; prolactin (PRL)-producing lactotrophs; corticotrophs (adrenocorticotropic hormone [ACTH], pro-opiomelanocortin [POMC], melanocyte-stimulating hormone, endorphins, and lipotrophins); thyrotrophs (thyroid-stimulating hormone [TSH]); and go-nadotrophs (follicle-stimulating hormone [FSH], luteinizing hormone [LH]).
Various transcription factors control the development of the anterior pituitary. The identification of these transcription factors has enabled further classification of pituitary tumors and has provided an explanation for adenomas with multiple hormone production. Three main pathways of cell differentiation have been defined in the anterior pituitary. The expression of T-pit, which binds to corticotrophin upstream transcription-binding element (CUTE) proteins, is involved in the differentiation of ACTH-producing corticotrophs. The cytodifferentiation of gonadotrophs requires the expression of steroidogenic factor (SF)-1, GATA-2, and members of the Lhx gene family. Cells that express Pit-1 can develop into somatotrophs, lactotrophs, mammosomatotrophs, or thyrotrophs.2,3
The posterior pituitary comprises the remaining 20% of the gland and is composed of the infundibulum, pituitary stalk, and pars nervosa. It consists of pituicytes (modified glial cells) and axonal processes arising from the supraoptic and paraventricular nuclei in the hypothalamus. It is embryologically derived from an outpouching of the floor of the third ventricle. Antidiuretic hormone (vasopressin) and oxytocin are produced in the neuronal bodies of these nuclei and are stored in axon terminals in the posterior pituitary.
The pituitary has a dual circulation—a portal and an arterial system. The internal carotid artery gives rise to the superior, middle, and inferior hypophyseal arteries, which supply the gland. The infundibulum of the neurohypophysis is supplied by the superior hypophyseal artery, which forms the portal system of the pituitary. Direct blood supply to the anterior pituitary comes from the middle hypophyseal artery, whereas the pars nervosa is supplied by the inferior hypophyseal artery. Venous drainage is mainly to the internal jugular.
 Surgical Pathology Approach to the Diagnosis of Pituitary Adenoma
Surgical Pathology Approach to the Diagnosis of Pituitary Adenoma
The surgical pathologist often encounters several challenges during the process of diagnosing pituitary adenomas. In intraoperative consultations the main challenge is distinguishing between pituitary tissue and other lesions (Table 8.1) that are in the differential diagnosis for masses in the pituitary region. This is particularly important in cases where the distinction is between a tumor and an inflammatory condition, which may affect the surgeon’s decision about the continuation of the operation. The recognition of a neuroendocrine cell versus features specific for the other lesions is essential. Both smear preparations and frozen sections are helpful for providing information on cellular morphology and architecture.
The next challenge is to distinguish between normal pituitary and adenoma. On cytologic preparations, abundant cellularity usually favors adenoma. On both cytologic and histologic preparations, cytologic monomorphism, uniform cytoplasmic staining quality, occasional multinucleate or pleomorphic cells, prominent nucleoli, and mitotic figures are all indicative of adenomas. Histologically, the presence of acinar or small-cluster architecture with delicate fibrous septa and variability in the cytologic staining pattern are indicative of a normal pituitary tissue. The effacement of the acinar pattern and replacement by a sheet-like morphology lacking organized fibrous septa, the presence of papillary morphology or a sinusoidal architecture, with or without pseudo-rosette formation are indicative of a pituitary adenoma. Additional challenges facing the pathologist are cases of previously treated adenomas that may present excessive fibrous deposition and cases of pituitary apoplexy in which infarcted, hemorrhagic, nonviable tissue is predominantly seen. Occasionally, hypoxic damage in a pituitary adenoma may produce small cells with pyknotic nuclei resem bling lymphocytes, which may be confused with lymphocytic hypophysitis.
Table 8.1 Sellar Region Masses
| Tumors/masses of Pituitary Origin | Metastatic/Hematopoietic Tumors |
| Tumors of adenohypophyseal origin | Carcinoma |
| Pituitary adenoma | Lymphoma |
| Intrasellar gangliocytoma | Plasmacytoma |
| Spindle cell oncocytoma | Leukemias |
| Pituitary carcinoma | Cysts/hamartomas/malformations/vascular |
| Tumors of neurohypophyseal origin | Rathke’s cleft cyst |
| Pituicytoma (glioma) | Arachnoid cyst |
| Granular cell tumor | Epidermoid, dermoid, teratomatous cysts |
| Adenohypophyseal hyperplasia | Hamartomas |
| Primary nonpituitary tumors | Aneurysms |
| Craniopharyngioma | Cavernous hemangioma |
| Germ cell tumors | Arteriovenous malformation |
| Meningioma | Inflammatory conditions |
| Chordoma | Lymphocytic hypophysitis |
| Gliomas | Langerhans cell histiocytosis |
| Schwannoma | Sarcoidosis |
| Paraganglioma | Other granulomatous conditions |
| Tumors of mesenchymal origin | Mucocele |
| Sarcomas (osteo-, chondro-, leiomyo-, rhabdomyo-, fibro-, etc.) | Pyogenic/infectious abscess |
| Solitary fibrous tumors/hemangiopericytoma (HPC) | |
| Lipoma | |
| Chondroma | |
| Giant cell tumor of bone |
In permanent sections the diagnosis is usually made by routine hematoxylin and eosin (H&E) staining, assisted by immunohistochemistry of the antibodies against the six hormones (PRL, GH, ACTH, TSH, LH, and FSH) in addition to synaptophysin (with or without chromogranin). The immunohistochemical staining may be useful for both diagnostic purposes (for example, distinguishing between a pituitary adenoma and a metastatic carcinoma, or between nonneoplastic pituitary, normal, or hyperplastic versus adenoma), and for providing useful information to the endocrinologist (for example, distinguishing between PRL-secreting adenoma versus PRL elevation in a nonfunctional adenoma due to compression of the stalk). The addition of synaptophysin with or without chromogranin might be useful for the diagnosis of adenomas that do not stain with any hormonal product (about one fourth to one third of all adenomas). Occasionally, reticulin staining can be helpful in distinguishing between adenomas and normal pituitary.
 Primary Tumors of Adenohypophysial Cells
Primary Tumors of Adenohypophysial Cells
Most pituitary tumors that arise in the anterior pituitary are benign (pituitary adenomas). Pituitary carcinomas represent a small proportion of pituitary lesions.
Pituitary Adenomas
The majority of pituitary tumors are pituitary adenomas, which constitute approximately 10 to 25% of all intracranial tumors.4,5 Although most of these tumors arise in the sella turcica, some may be ectopic.6 It has been reported that up to 27% are incidental findings (incidentalomas) found at the time of autopsy or during radiologic studies for other reasons.5,7 These incidental tumors are usually hormonally inactive or slow growing without mass effect.
There are no currently reliable markers to predict behavior in adenomas. Proliferation markers such as Ki-67/Mib-1, proliferating cell nuclear antigen (PCNA), and DNA topoisomerase IIα have been suggested as useful parameters.8–10 Growth factors and receptor expression,11–13 in addition to oncogene and tumor suppressor gene alterations,14,15 have been evaluated as potential prognostic markers.2,16 DNA ploidy does not predict tumor prognosis or recurrence.17
Pituitary adenomas are most commonly found in adults, with the peak occurrence in the fourth to sixth decade. They are uncommon in children, representing approximately 2 to 6% of all intracranial tumors.18–20 The majority of pituitary adenomas are functioning in children, with nonfunctioning tumors being rare. Some authors have found that tumors in children are larger, more aggressive, and more invasive.19
Although the majority of lesions occur in isolation (sporadic), a small percentage is associated with multiple endocrine neoplasia (MEN) type 1. Multiple adenomas have been reported in less than 1% of patients at autopsy and are very rare in surgical specimens.21
The pathogenesis and development of these tumors is not well understood, although several factors have been implicated.3 They lack mutations in well-known tumor suppressor genes and oncogenes.22
Several classification systems are used for pituitary adenomas. Functional, anatomical, and histologic classifications have been employed. In the functional classification system, tumors are classified according to their in vivo hormonal activity and are either functioning or nonfunctioning (silent [no clinical symptoms] or hormone negative [immunohistochemical and ultrastructurally negative for hormone]). Anatomical or radiographic classification differentiates tumors according to their size and degree of local invasion. Morphologic classification uses a combination of conventional cytoplasmic staining, immunohistochemical detection of tissue antigens, and ultrastructural evaluation. Immunohistochemical detection of hormone content with the addition of immunohistochemical detection of other markers of cell differentiation such as transcription factors and cytokeratins can also be useful in classifying pituitary adenomas. Histologic (acidophil, basophil, chromophobe) and ultrastructural classifications are not commonly used. The most effective classification scheme is clinicopathologic and combines both morphologic and clinical features of the tumors.
Functioning Somatotroph Adenomas
Functioning somatotroph adenomas are derived from somatotrophs, which produce GH. They represent 10 to 15% of all adenomas.1
Pathology
Grossly, these tumors are located in the lateral wing of the pituitary. Microadenomas tend to be well demarcated, whereas macroadenomas, which represent the majority of GH-producing adenomas, commonly show suprasellar or lateral extension.3
Densely Granulated Somatotroph Adenomas
These tumors consist of acidophilic cells arranged in a trabecular, sinusoidal, or diffuse growth pattern (Fig. 8.1). They show strong diffuse reactivity for GH and nuclear staining for Pit-1. Perinuclear positivity for low molecular weight cytokeratin is seen, whereas there is variable staining for the α-subunit of glycoproteins. Ultrastructurally, the cells resemble normal somatotrophs. They have rough endoplasmic reticulum arranged in arrays and well-formed Golgi complexes.
Numerous dense secretory granules measuring 150 to 600 nm are present.
Sparsely Granulated Somatotroph Adenomas
These tumors are composed of chromophobic cells in a solid growth pattern. The cells show prominent nucleoli and nuclear pleomorphism. Positivity for low molecular weight cytokeratin in a juxtanuclear pattern highlights the characteristic fibrous body seen in these adenomas. There is weak immunoreactivity for GH. Nuclear reactivity for Pit-1 is seen, whereas there is occasional immunoreactivity for the α-subunit of glycoproteins. Ultrastructurally, the nuclei are eccentric and pleomorphic. The rough endoplasmic reticulum can be poor to well developed. Juxtanuclear whorls of intermediate filaments representing the fibrous body are seen. Secretory granules are sparse and measure 100 to 250 nm.
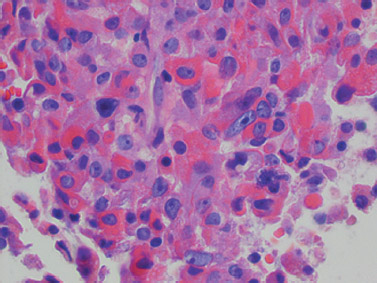
Fig. 8.1 Acidophilic, densely granulated, somatotroph adenoma with nuclear pleomorphism (×400).
Mammosomatotroph Adenomas
These tumors are composed of uniform cells with chromophobic appearance, with or without slight acidophilia, arranged in a solid or diffuse pattern. These tumor cells produce both GH and PRL and this tumor is the most common cause of gigantism.1 The tumor cells are strongly reactive for GH with variable reactivity for α-subunit and PRL.1 Perinuclear positivity for low molecular weight cytokeratin is seen. There is strong Pit-1 nuclear positivity and rare positivity for the β-subunit of TSH. Ultrastructurally, the tumor cells resemble densely granulated somatotrophs. Secretory granules in these tumors can measure up to 1000 nm and have mottled cores. Misplaced exocytosis, which is a classic feature seen in prolactinomas, is identified in these adenomas.
In a small number of these tumors, mature somatotrophs and lactotrophs are identified with GH and PRL immunostaining localized to each individual cell type. These tumors have been referred to as mixed somatotroph-lactotroph adenomas.
Functioning Lactotroph Adenomas
These adenomas produce PRL and are derived from the lactotroph cells of the adenohypophysis.
Pathology
Most arise in the lateral or posterior pituitary. Macroadenomas tend to be more aggressive lesions and may invade into the dura and nasal sinuses.
Sparsely Granulated (Chromophobe) Lactotroph Adenomas
These are the most common variant of lactotroph adenomas. They are composed of chromophobic or slightly acidophilic large cells with indistinct cell borders and irregular nuclei with prominent nucleoli. The tumor cells can show a papillary, trabecular, or solid growth pattern. Calcifications in the form of psammoma bodies may be occasionally seen. Less frequently amyloid may be identified.
There is strong staining for PRL in the Golgi region (globular juxtanuclear pattern), and the cells are periodic acid-Schiff (PAS) negative. Ultrastructurally, there is prominent rough endoplasmic reticulum arranged in parallel arrays or concentric whorls (Nebenkern formations), large Golgi complex with immature granules, and sparse electron-dense granules measuring 150 to 300 nm. Misplaced lateral cell wall exocytosis is characteristic.
Densely Granulated Lactotroph Adenomas
These are the less common variant of the lactotroph adenoma, which is composed of acidophil cells. Unlike the Golgi staining seen in sparsely granulated tumors, PRL staining in these tumors is diffuse and cytoplasmic. Ultrastructurally, there are abundant rough endoplasmic reticulum, numerous secretory granules measuring up to 700 nm, and misplaced exocytosis.
Acidophil Stem Cell Adenomas
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


