FIGURE 11.1 Nasopalatine Duct Cyst, 200×.
The cyst is lined by a ciliated pseudostratified cuboidal to the low columnar epithelium (inset lower left, 400×) with underlying fibroconnective tissue and a prominent nerve (arrow). Nasopalatine duct cysts originate from embryonic remnants of the nasopalatine ducts. Although nonodontogenic, when lined by squamous epithelium, the cyst may resemble a periapical (radicular) cyst. Clinical and radiographic information combined with pathology are essential in arriving at the correct diagnosis.
FIGURE 11.2 Fibroepithelial Polyp (Fibroma), 20×.
The polyp is lined by an acanthotic, stratified squamous epithelium with a dense fibrovascular tissue stroma. Fibroepithelial polyps are common in the oral cavity and frequently occur in areas that are prone to trauma such as the tongue, buccal mucosa, and lower lip. They may be referred to as “irritation” or “bite” fibromas.
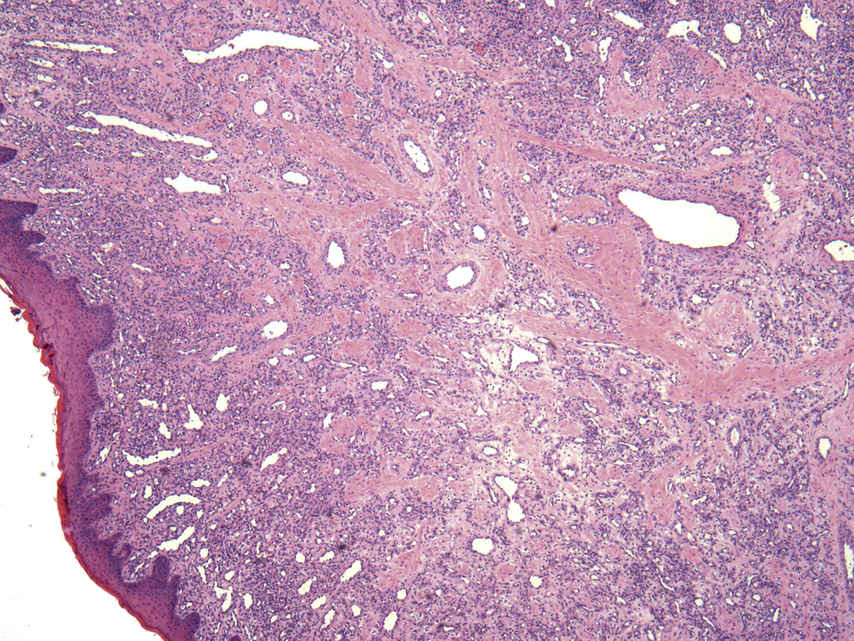
FIGURE 11.3 Pyogenic Granuloma (Lobular Capillary Hemangioma), 40×.
The image shows proliferation of small capillary-sized blood vessels in a lobular arrangement with a central small ectatic feeder vessel in each lobule and an inflammatory background. Lobular capillary hemangioma is a common lesion of the oral cavity. When pyogenic granulomas occur during pregnancy, they are commonly referred to as pregnancy epulis or granuloma gravidarum.
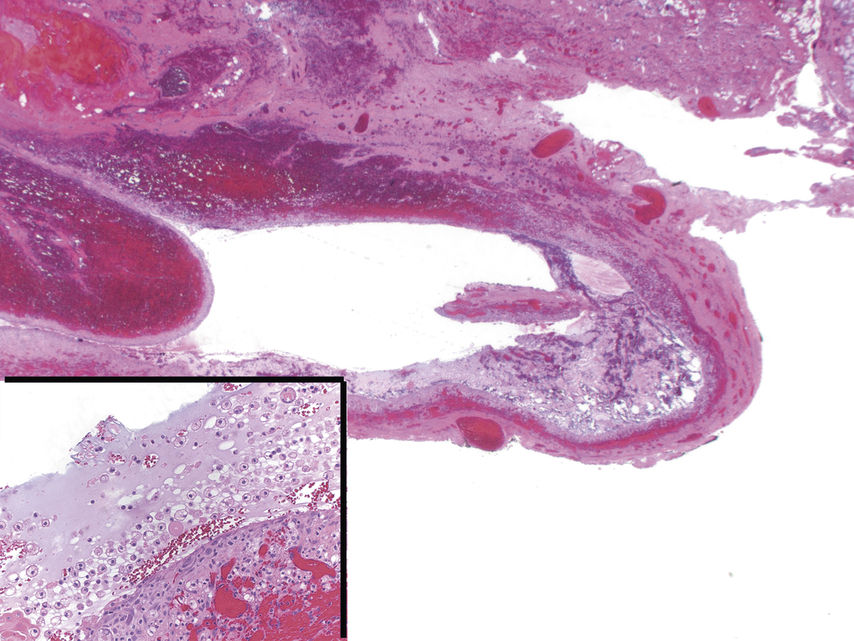
FIGURE 11.4 Mucocele of the Sublingual Gland (Ranula), 20×.
The image shows a large area of extravasated mucin. The mucin (which appears gray/pink in color) contains abundant histiocytes with mucin in their cytoplasm (muciphages) (inset lower left, 200×). The extravasated mucin is surrounded by a wall of granulation tissue and as such, mucoceles are pseudocysts (lacking an epithelial cyst lining). Mucoceles are commonly seen in the lower lip because of bite injury to the minor salivary gland ducts. Mucoceles that arise from the sublingual glands push up into the floor of the mouth, forming a noticeable smooth cystic mass, termed ranula.
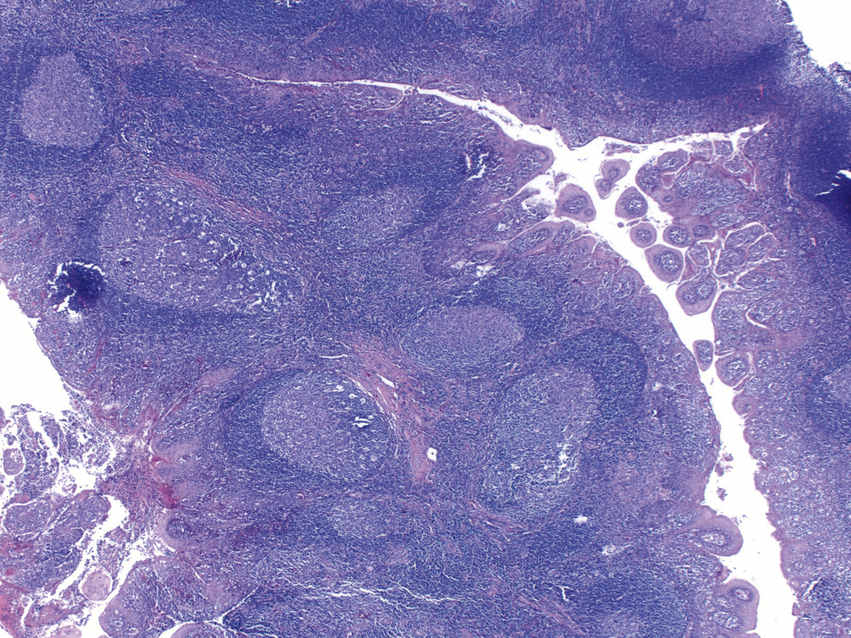
FIGURE 11.5 Reactive Lymphoid Follicular Hyperplasia, 20×.
This hyperplastic tonsil shows a lymphoid-rich stroma with prominent germinal centers, including tingible-body macrophages. When clinically worrisome, ancillary studies, such as flow cytometry, immunohistochemistry (IHC), and molecular testing for clonal gene rearrangements, are essential to exclude a neoplasia (including lymphoma).
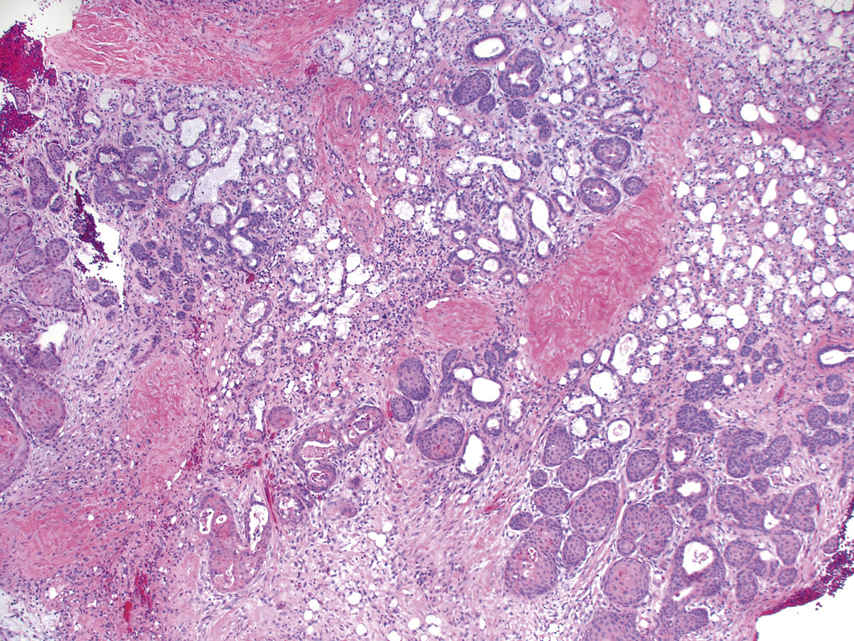
FIGURE 11.6 Necrotizing Sialometaplasia, 20×.
The image shows squamous metaplasia of the minor salivary gland ducts in a lobular arrangement. A few open ducts are present; however, many have been obliterated by the squamous metaplastic process. A moderate chronic inflammatory cell infiltrate is seen in the background with marked atrophy and destruction of the minor salivary gland acini (see also Fig. 11.7).
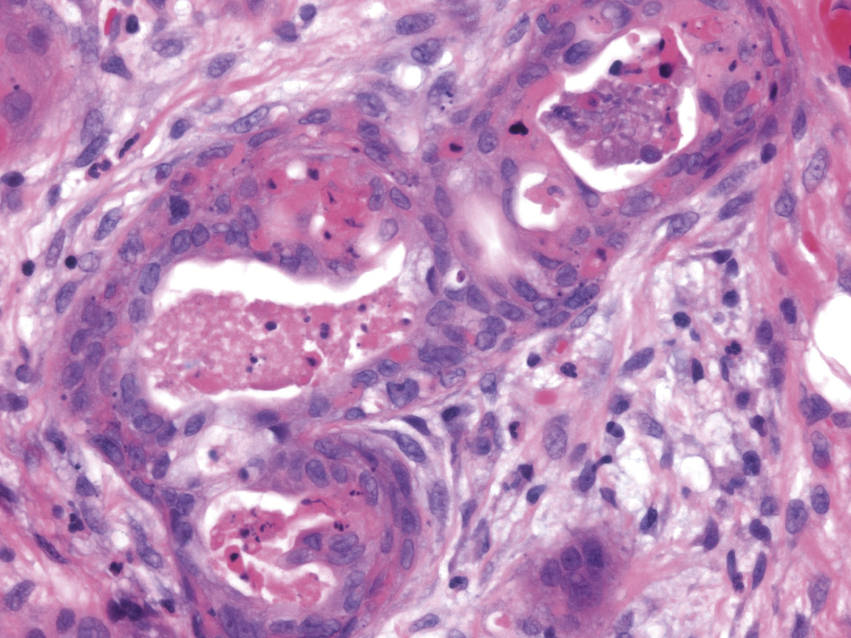
FIGURE 11.7 Necrotizing Sialometaplasia, 400×.
Necrotic debris and neutrophils are seen within the lumens of the metaplastic ducts. The metaplastic ductal cells are crowded and show nuclear hyperchromasia. The lesion may mimic a squamous cell carcinoma (SCC) in small biopsy specimens, where the lobular arrangement of the cells is difficult to appreciate. Necrotizing sialometaplasia presents as an ulcerative lesion of the hard palate, and may clinically simulate a carcinoma; however, these lesions usually lack the marked nuclear atypia and increased mitoses seen in a squamous cell carcinoma.
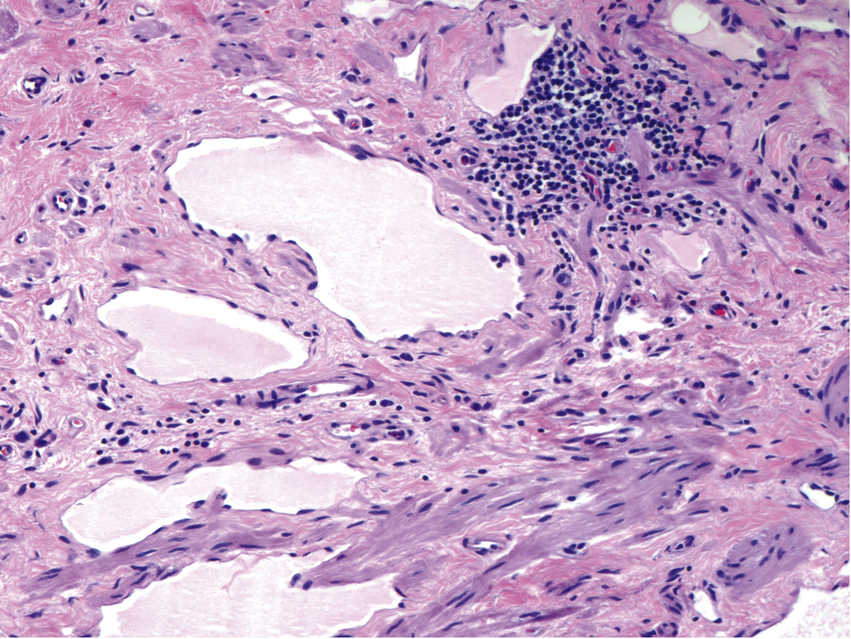
FIGURE 11.8 Lymphangioma, 200×.
The image shows large dilated lymphatic vessels lined by a flat, inconspicuous layer of bland endothelial cells. The lymphatic channels contain a pale proteinaceous material (lymph). A dense collection of lymphocytes is seen in the stroma surrounding the vessels. Lymphangiomas are generally seen in the head and neck region of newborns and are treated surgically.
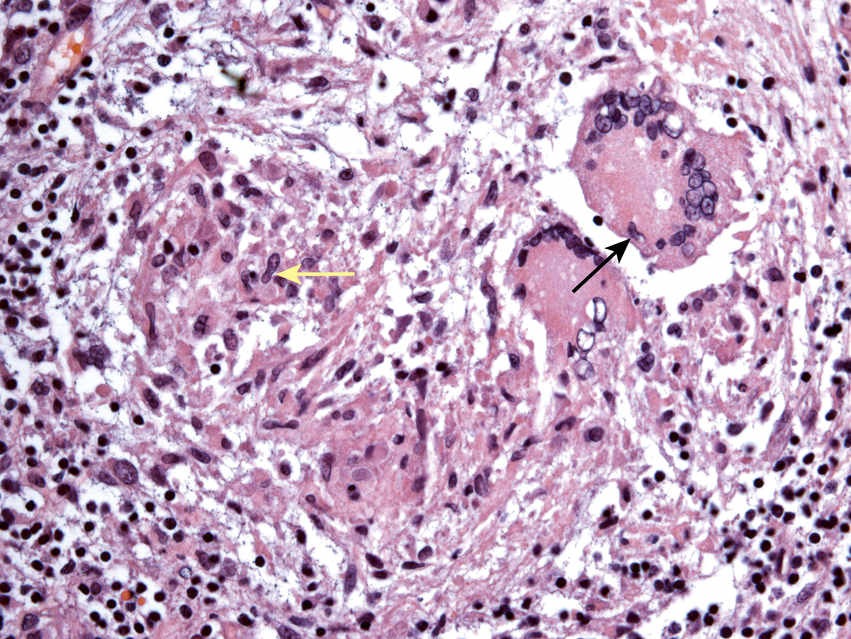
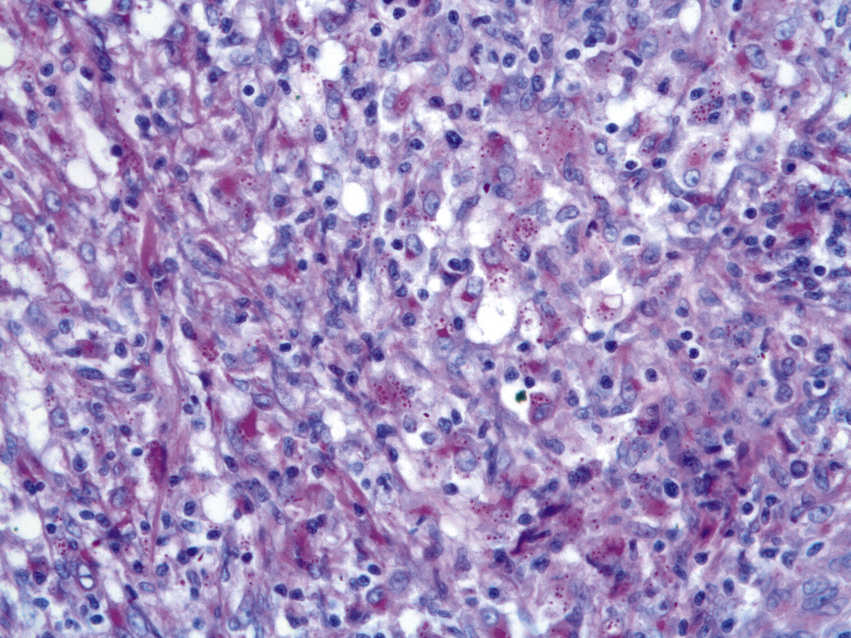
FIGURE 11.10 Granulomatous Inflammation, 200×.
The image shows a granuloma consisting of epithelioid histiocytes with elongated, curved nuclei (yellow arrow), multinucleated giant cells (black arrow), and a peripheral rim of small lymphocytes. The differential diagnosis of granulomatous inflammation is broad and includes a wide range of bacterial (eg, tuberculosis), fungal (eg, histoplasmosis), immunologic (eg, sarcoidosis, Crohn disease) and neoplastic conditions (eg, Hodgkin lymphoma).
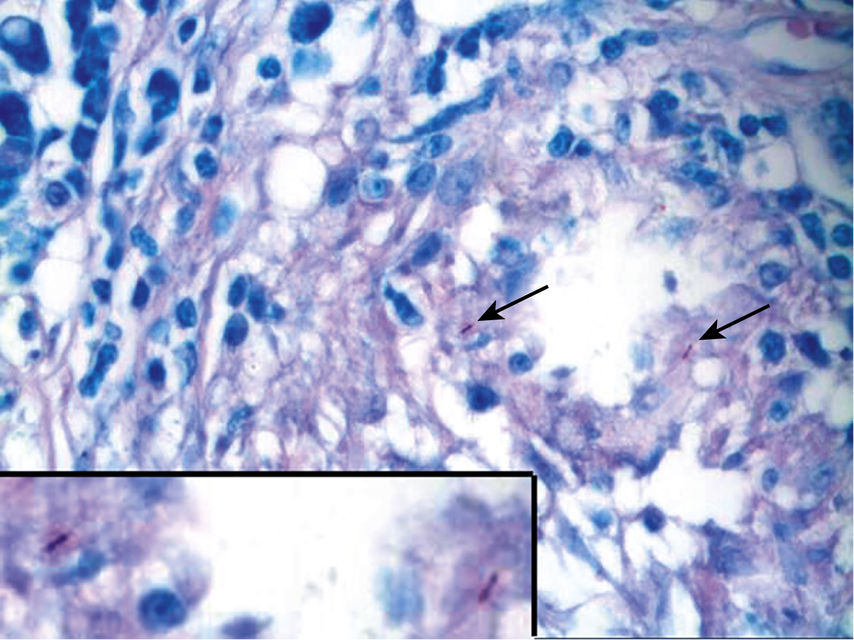
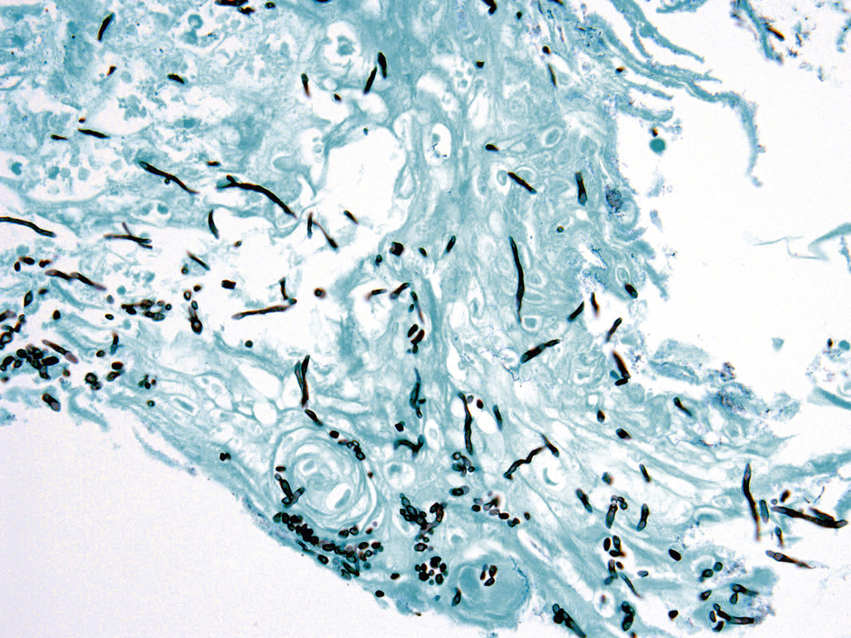
FIGURE 11.11 Tuberculosis (Ziehl-Neelsen Stain), 200×.
The image shows acid-fast bacilli (indicated by arrows in the image and magnified in the inset lower left). Oral tuberculosis usually presents as an atypical area of ulceration in the oral cavity and may clinically resemble a malignancy. It results from direct inoculation of acid-fast bacilli into the oral tissues.
Benign Tumors
• Granular cell tumor (Fig. 11.13)
• Lipoma (Fig. 11.14)
• Spindle cell lipoma (Fig. 11.15)
Epithelial Precursor Lesions
• Mild epithelial dysplasia (Fig. 11.16)
• Severe epithelial dysplasia/carcinoma in situ (Fig. 11.17)
• Verrucous hyperplasia (Fig. 11.18)
Malignant Epithelial Lesions
• Verrucous carcinoma (Fig. 11.19)
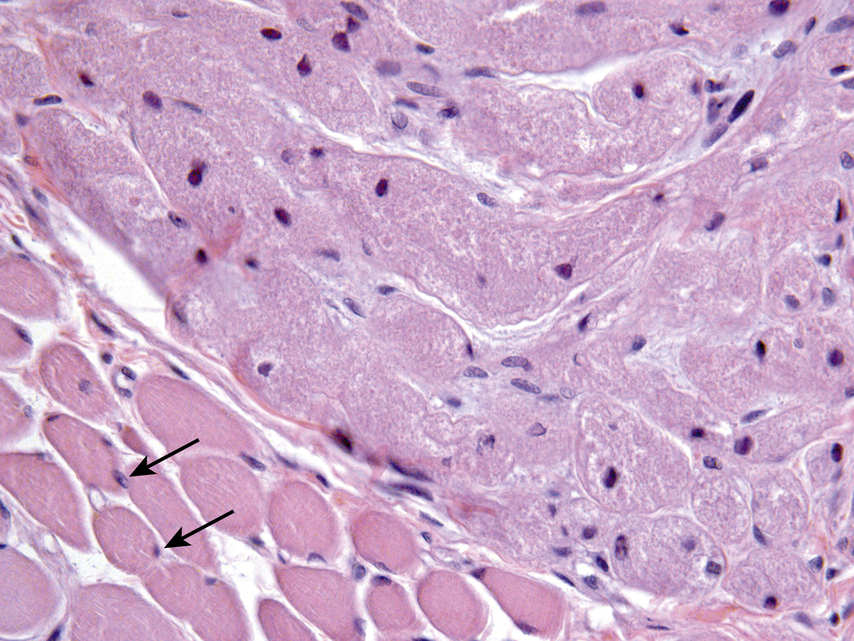
FIGURE 11.13 Granular Cell Tumor of the Tongue, 200×.
Cytologically bland large polygonal cells with abundant granular, oncocytic (pink) cytoplasm and centrally located small, dark nuclei. In this image, the granular cells abut skeletal muscle cells (arrows). Note the similarity between the two cell types, distinguished by the circumscription of the granular cell tumor with granular, pink fluffy cytoplasm and the skeletal muscle with a glassy/striated cytoplasm. The granular cells react strongly and diffusely with antibodies to S100 protein and are thought to be of Schwannian origin.
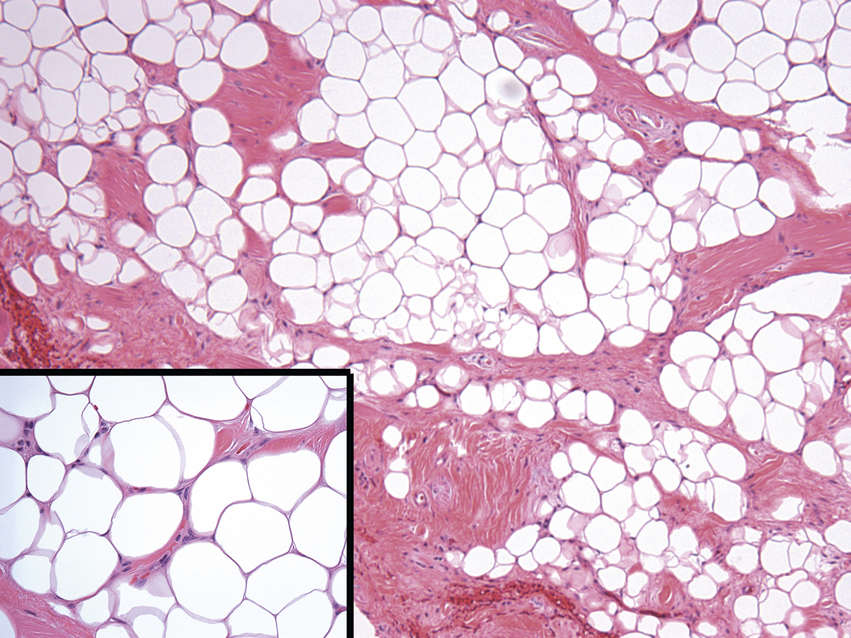
FIGURE 11.14 Lipoma, 100×.
The image shows lobules of mature adipose tissue separated by thick bands of fibrous tissue. The adipocytes are univacuolated with their slender hyperchromatic nuclei located at the periphery of the cell, often not apparent (inset lower left, 400×). Nuclear atypia or marked variation in size might raise the possibility of liposarcoma. Lipomas are usually solitary lesions in adults and commonly involve the oral cavity and the larynx; the degree of cellularity and fibrosis is location-dependent.
• Basaloid squamous cell carcinoma (Fig. 11.22)
• Adenosquamous carcinoma (Fig. 11.23)
• Nonkeratinizing squamous cell carcinoma (Fig. 11.24)
• Immunohistochemistry for p16 (Fig. 11.25)
Malignant Mesenchymal Tumors
• Kaposi sarcoma (Fig. 11.26)
Immune-Mediated Lesions
• Pemphigus vulgaris (Fig. 11.27)
• Pemphigoid (Fig. 11.28)
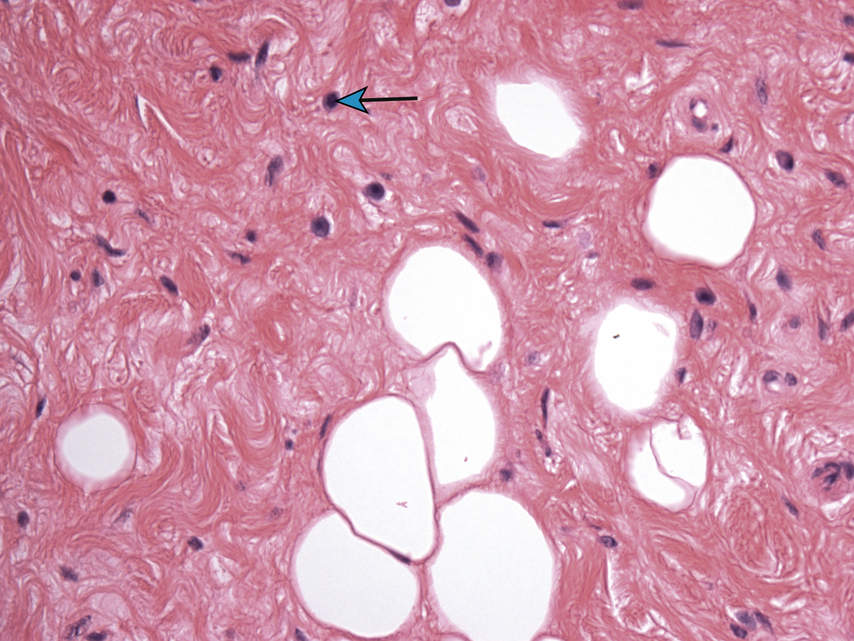
FIGURE 11.15 Spindle Cell Lipoma, 400×.
Spindled fibroblasts, shredded collagen fibers, mast cells (arrow), myxoid change, and mature adipocytes characterize this variant of lipoma, commonly seen in the oral cavity.
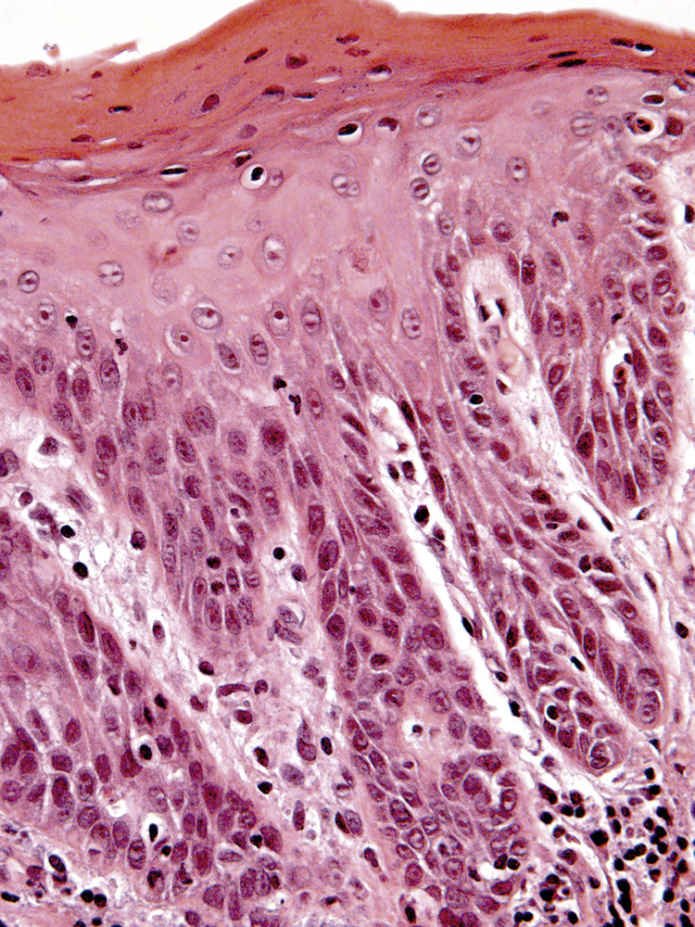
FIGURE 11.16 Mild Epithelial Dysplasia, 200×.
The epithelium shows drop-shaped rete processes and loss of nuclear polarization. Cytologically, there is nuclear hyperchromasia, and prominent nucleoli. The architectural and cytologic features are confined to the lower one-third of the epithelium.
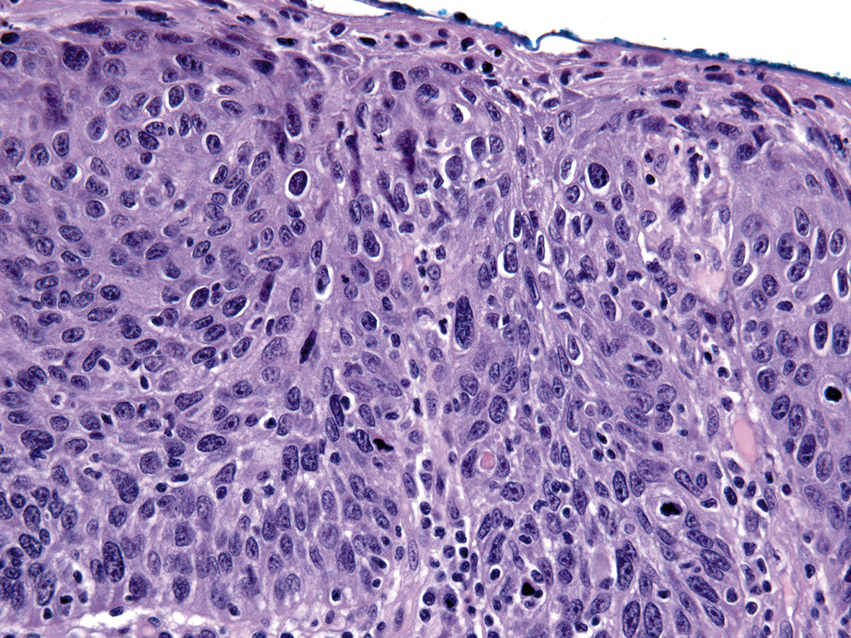
FIGURE 11.17 Severe Epithelial Dysplasia/Carcinoma in Situ, 200×.
There is full-thickness, marked nuclear atypia and pleomorphism, with suprabasilar mitoses and disordered maturation of cells.
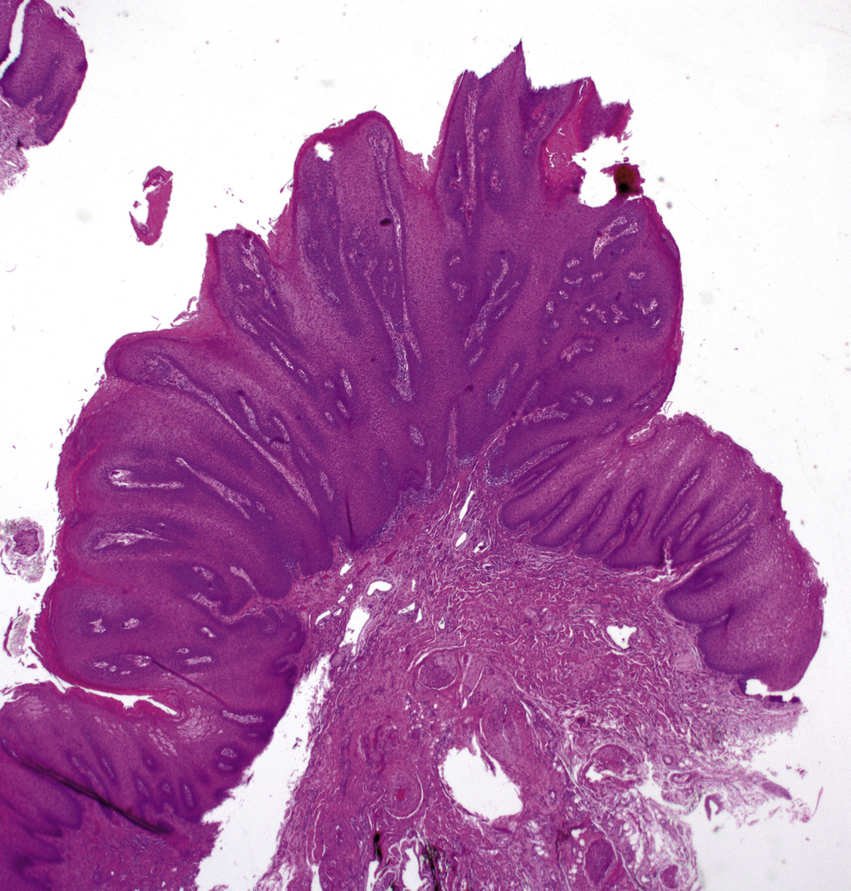
FIGURE 11.18 Verrucous Hyperplasia, 20×.
Although the rete processes appear elongated, they are relatively uniform and do not appear deeper than the surrounding normal epithelium. The lesion lacks significant cytologic atypia. Verrucous hyperplasia is seen in the oral cavity, often in the setting of proliferative verrucous leukoplakia. It has a high recurrence rate and may transform into verrucous carcinoma or invasive SCC. The word verrucous reflects the warty appearance of these lesions with pointy, spire-like architecture.
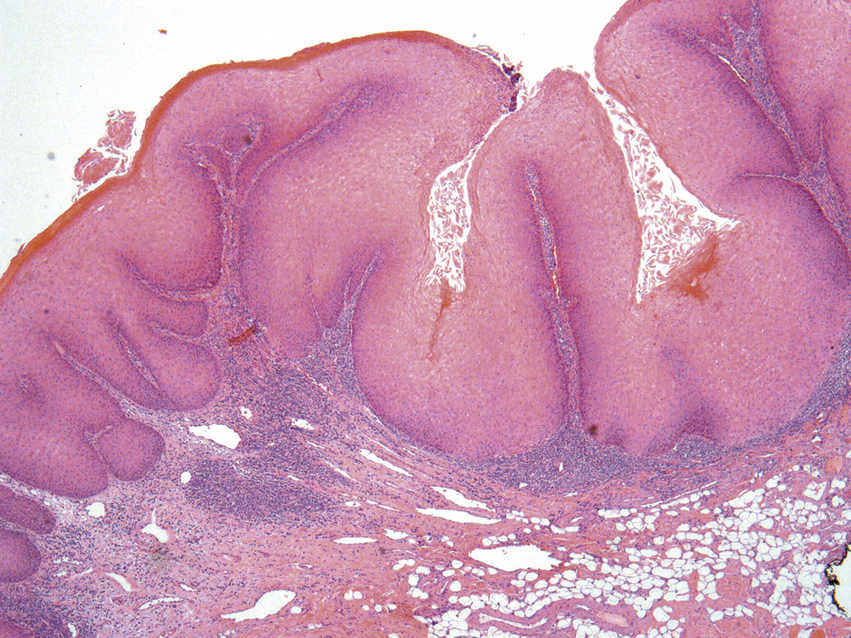
FIGURE 11.19 Verrucous Carcinoma, 20×.
Large and broad rete processes push deeply into the submucosa in broad, pushing nests. The folded and thickened epithelium shows marked surface keratinization. A dense chronic inflammatory response may be seen at the advancing front of the lesion, and there is no epithelial atypia. Small and superficial biopsies can create diagnostic difficulties because the base of the lesion may not be visualized.
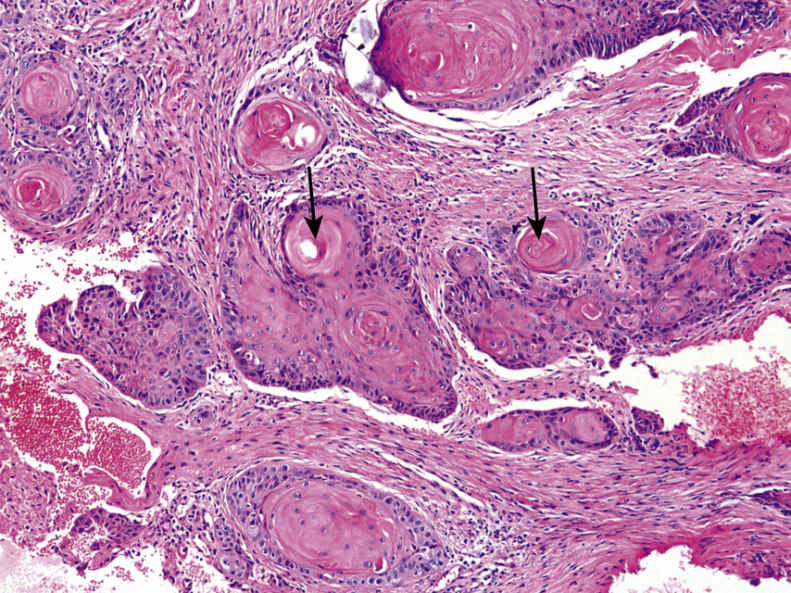
FIGURE 11.20 Keratinizing Squamous Cell Carcinoma, 200×.
The image shows a well-differentiated SCC with abundant intralesional keratinization, forming keratin pearls (arrows).
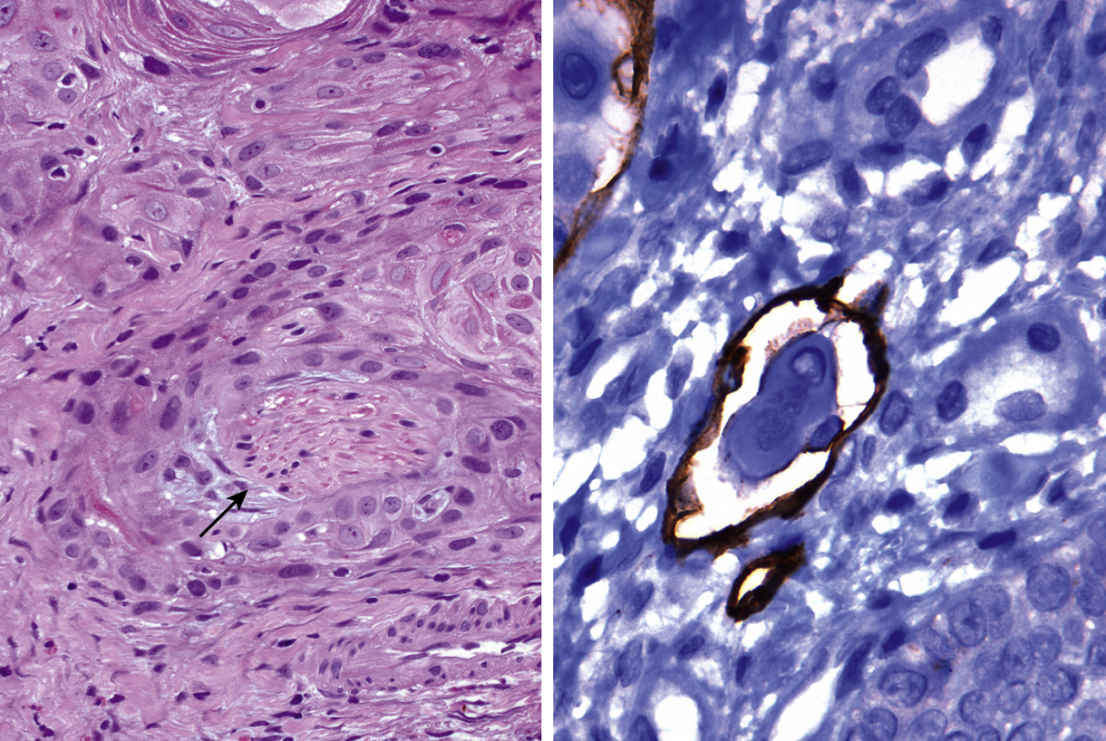
FIGURE 11.21 Keratinizing Squamous Cell Carcinoma with Perineural and Lymphovascular Invasion, 400×.
Malignant squamous cells surround a peripheral nerve (arrow, left image). Malignant squamous cells in a lymphatic vessel (right image, D2-40 IHC). Perineural invasion and lymphovascular invasion are frequently observed in SCCs.
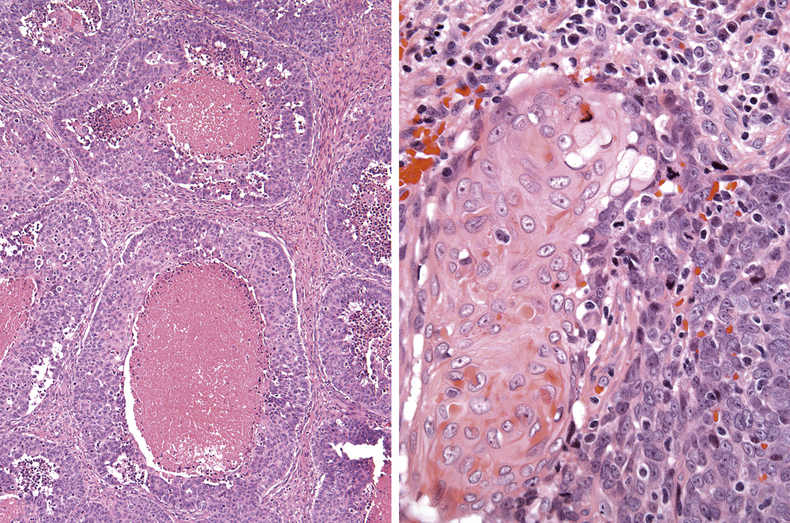
FIGURE 11.22 Basaloid Squamous Cell Carcinoma.
Islands of atypical basaloid cells (the term basaloid means the cells show a high nuclear to cytoplasmic ratio with small dense hyperchromatic nuclei, similar to basal epithelial cells) with central areas of necrosis (comedo-type necrosis) (left image, 100×). Most basaloid squamous cell carcinomas (BSCCs) show focal areas of high-grade surface dysplasia or keratinizing SCC (right image, 400×). Correctly diagnosing a BSCC can be challenging in small biopsies. The differential diagnosis includes other basaloid lesions such as adenoid cystic carcinoma (ADCC), salivary duct carcinoma, and nonkeratinizing, HPV-related SCC.
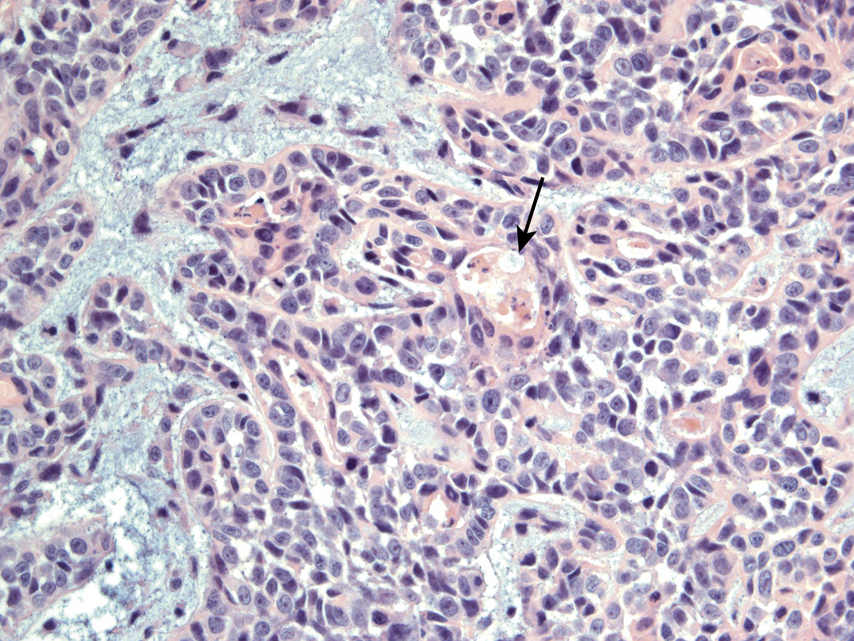
FIGURE 11.23 Adenosquamous Carcinoma, 200×.
The image shows an SCC with areas of true glandular differentiation (arrow). Adenosquamous carcinomas often behave aggressively.
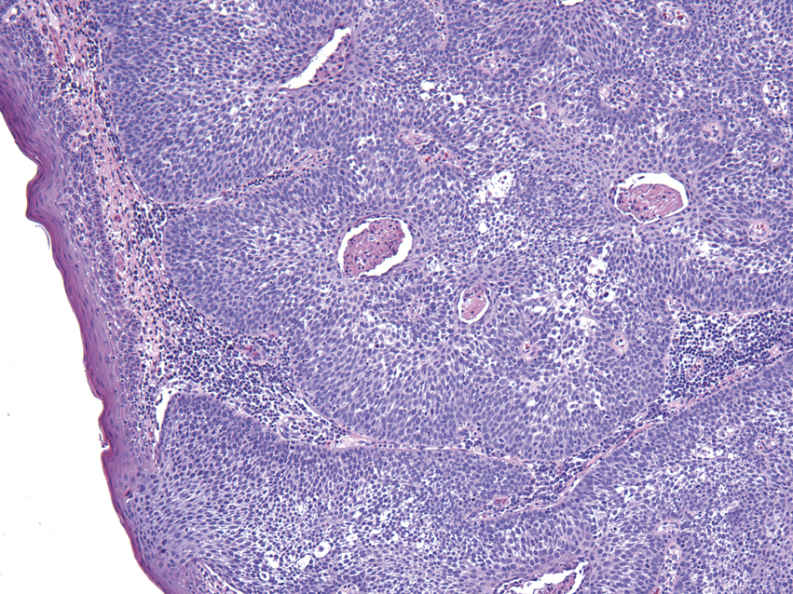
FIGURE 11.24 Nonkeratinizing Oropharyngeal Squamous Cell Carcinoma, 100×.
Nests of nonkeratinizing squamous cells with basaloid features, pushing borders, and intraluminal necrosis. Most oropharyngeal cases with this appearance are positive for high-risk (HR) HPV subtypes. They are most frequently found in the base of tongue and palatine tonsils, and careful attention may be needed to distinguish these malignant nests from the normal component lymphoid stroma. Although lacking the typical infiltrative features seen in other carcinomas, these nests of cells are considered to be invasive by nature and likely to metastasize via lymphatic channels within the tonsil if not already metastatic at the time of diagnosis.
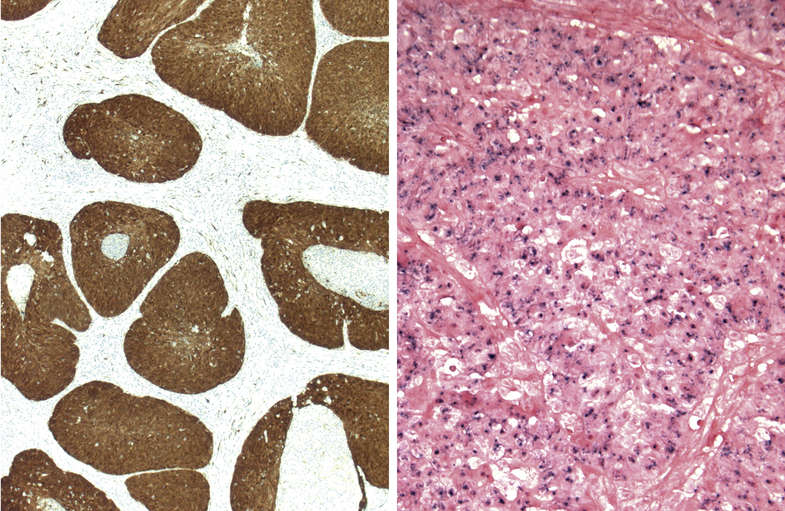
FIGURE 11.25 Human Papillomavirus-Related Nonkeratinizing Oropharyngeal Squamous Cell Carcinoma.
P16 IHC and in situ hybridization (ISH) for HR-HPV. Strong cytoplasmic and nuclear staining for P16 (image left, 200×). P16 IHC is a sensitive but nonspecific screening test for HR-HPV. More than 70% of the tumor cells should be positive for this antibody. P16-positive tumors are further evaluated for the presence of HR-HPV by polymerase chain reaction or ISH. The image on the right shows punctate positivity for HR-HPV (ISH, 400×).
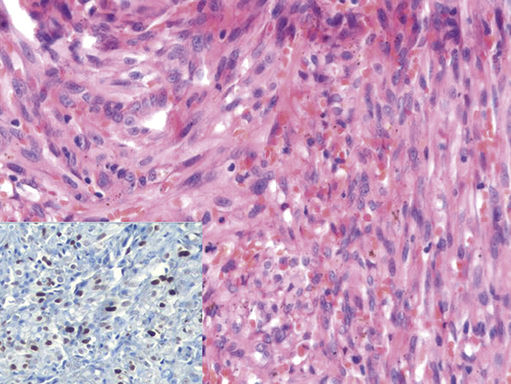
FIGURE 11.26 Kaposi Sarcoma, 400.
Moderately pleomorphic spindled cells surround slit-like vascular channels with extravasation of red blood cells. Nuclear reactivity with antibodies to human herpesvirus 8 is seen in most Kaposi sarcomas (inset lower left, 200×).
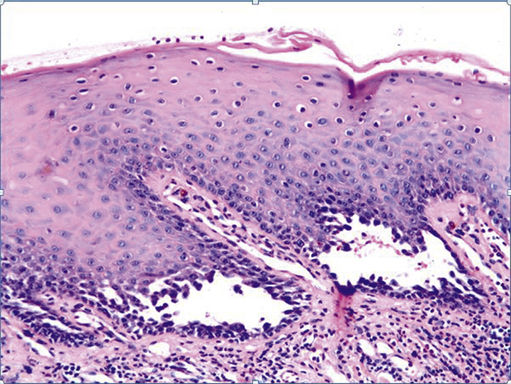
FIGURE 11.27 Pemphigus Vulgaris, 100×.
The image shows intraepithelial vesicles. The basal layer is intact. Pemphigus vulgaris is an autoimmune vesiculobullous disease. The IgG antibodies directed against the interepithelial desmosomes cause squamous cells to lose cohesion (acantholysis).
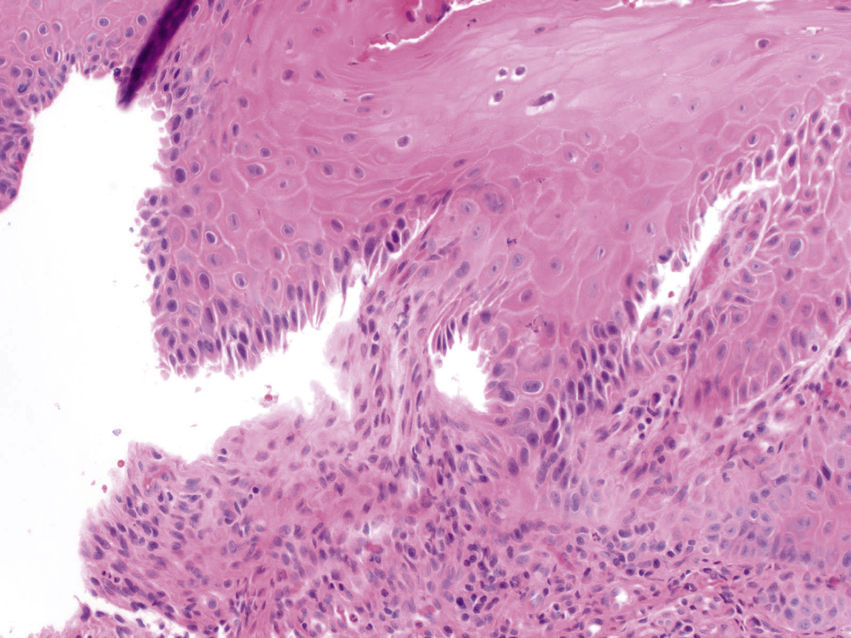
FIGURE 11.28 Pemphigoid, 200×.
A sub-basilar cleft is observed. Pemphigoid is an autoimmune vesiculobullous disease with antibodies directed against basement membrane antigens that connect the epithelial cells to the basement membrane.
• Lichen planus (Fig. 11.29)
• Sjögren syndrome (Fig. 11.30)
Lesions of the Osseous Jaw
• Osteomyelitis (Fig. 11.31)
• Osteoradionecrosis (Fig. 11.32)
• Central giant cell granuloma (Fig. 11.33)
• Osteoma (Fig. 11.34)
• Osteosarcoma (Fig. 11.35)
• Fibrous dysplasia (Fig. 11.36)
• Ossifying fibroma (Fig. 11.37)
Selected Odontogenic Cysts and Tumors
Odontogenic Cysts
• Periapical (radicular) cyst (Fig. 11.38)
• Dentigerous cyst (Fig. 11.39)
Benign Epithelial Odontogenic Tumors
• Solid/multicystic ameloblastoma (Fig. 11.40)
• Calcifying epithelial odontogenic tumor (Fig. 11.41)
• Adenomatoid odontogenic tumor (Fig. 11.42)
• Keratocystic odontogenic tumor (Fig. 11.43)
Benign Mixed Epithelial Tumors
• Odontoma (Fig. 11.44)
Benign Mesenchymal Odontogenic Tumors
• Odontogenic myxoma (Fig. 11.45)
Larynx and Hypopharynx
Non-Neoplastic Lesions
• Contact ulcer of the larynx (Fig. 11.46)
• Vocal cord polyp (Fig. 11.47)
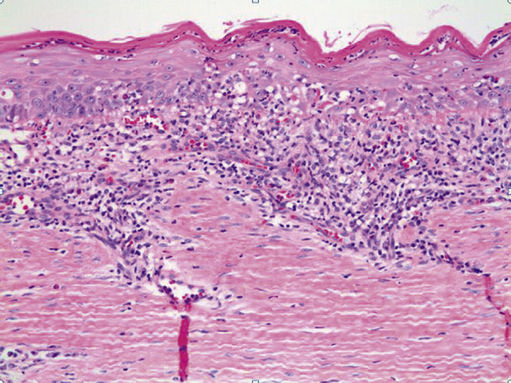
FIGURE 11.29 Lichen Planus, 100×.
A dense band-like lymphocytic infiltrate is seen at the epithelial-connective tissue interface, with hydropic (vacuolar) degeneration of epithelial basal cells. The histologic features of lichen planus can be mimicked by a number of conditions, which include lichenoid contact reactions, lichenoid drug eruptions, chronic graft vs. host disease, and lupus erythematosus. Careful clinicopathologic correlation is essential in arriving at the correct diagnosis.
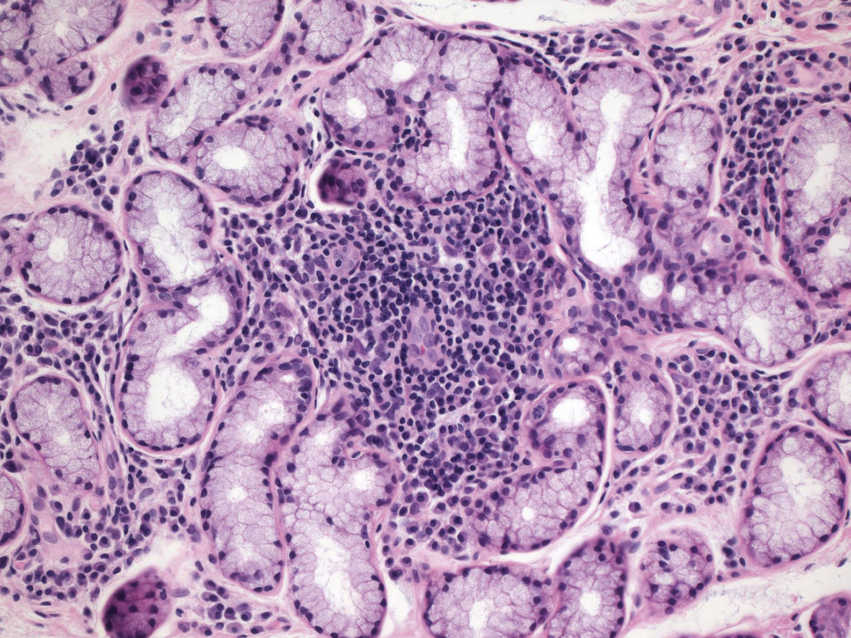
FIGURE 11.30 Sjögren Syndrome, Lip Biopsy, 400×.
The image shows a dense lymphoid infiltrate within the parenchyma of the minor salivary gland. More than 50 lymphocytes are seen in one focus. Sjögren syndrome is a systemic autoimmune disease. Patients may present with SICCA syndrome, including dry eyes and dry mouth. The diagnosis is a clinical diagnosis that may be supported by pathologic findings, generally by biopsy of non–bite line minor salivary tissue.
Benign Epithelial Tumors
• Laryngeal papilloma (Fig. 11.48)
• In situ hybridization for human papillomavirus (HPV) (Fig. 11.49)
Malignant Epithelial Tumors
• Squamous cell carcinoma of the larynx with thyroid cartilage invasion (Fig. 11.50)
• Papillary squamous cell carcinoma (Fig. 11.51)
• Spindle cell (sarcomatoid) carcinoma (Fig. 11.52)
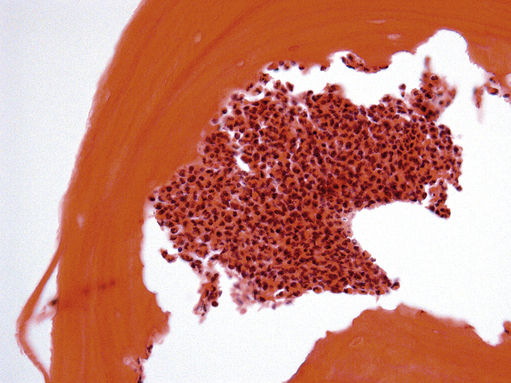
FIGURE 11.31 Osteomyelitis, 400×.
The marrow space contains a dense collection of neutrophils. The surrounding nonvital bone shows loss of osteocytes from the lacunae and peripheral “moth-eaten” areas of resorption (an attempt by the osteoclasts to remove the dead bone). Osteomyelitis commonly involves the mandible, and is associated with trauma or spread of infection from an odontogenic focus.
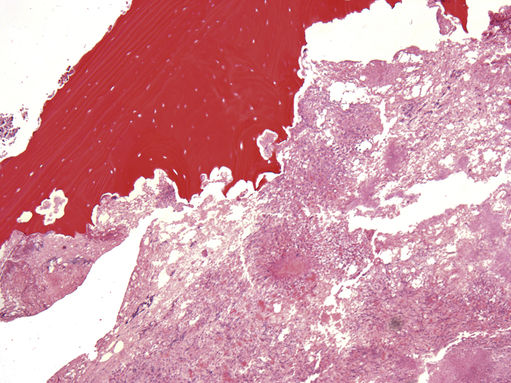
FIGURE 11.32 Osteoradionecrosis, 40×.
Necrotic bone with loss of osteocytes from the lacunae and peripheral ragged areas of resorption. The dead bone is surrounded by basophilic bacterial colonies consistent with Actinomyces spp. The diagnosis of osteoradionecrosis requires history of prior irradiation because the histologic picture is identical to osteomyelitis of bacterial origin.
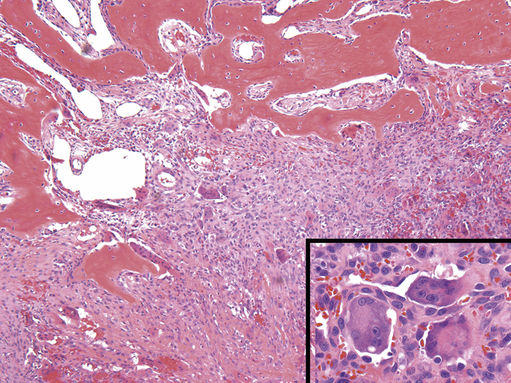
FIGURE 11.33 Central Giant Cell Lesion (Giant Cell Reparative Granuloma), 40×.
Plump cytologically bland spindled cells, osteoclast-like multinucleated giant cells, and extravasated red blood cells (inset lower right, 400×). Reactive bone is seen at the advancing front of the lesion. These are commonly seen in young adults and frequently involve the mandible. Before a diagnosis of central giant cell lesion is rendered, other entities with identical histology should be excluded. These include the brown tumor of hyperparathyroidism, cherubism, aneurysmal bone cyst, and peripheral giant cell granuloma.
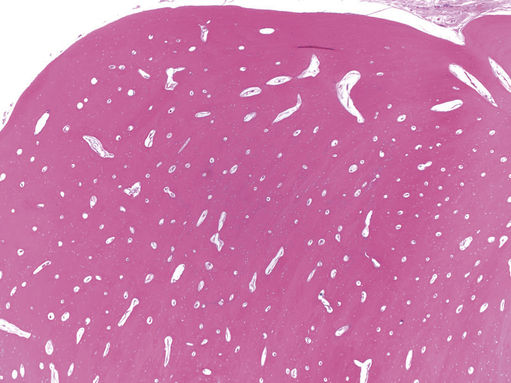
FIGURE 11.34 Osteoma, 20×.
The lesion is well circumscribed and consists of dense, mature cortical bone. Osteomas are histologically classified into compact (cortical) and spongy (trabecular) types. Multiple osteomas are seen in the setting of Gardner syndrome, an autosomal-dominant disorder characterized by gastrointestinal (GI) polyps and skin and soft tissue tumors. The GI polyps have a 100% risk of undergoing malignant transformation.
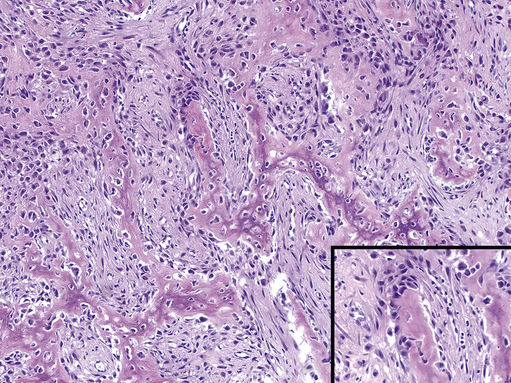
FIGURE 11.35 Osteosarcoma, Osteoblastic Type, 100×.
Atypical osteoblasts with lace-like areas of osteoid (malignant bone) deposition. A few malignant osteoblasts are entrapped within the osteoid (inset lower right, 400×). Osteosarcoma is the most common primary malignancy of bone. In the head and neck, osteosarcomas commonly arise in the mandible, and may present as rapidly enlarging painful lesions with loose teeth. Tumors may show extensive fibroblastic or chondroblastic differentiation.
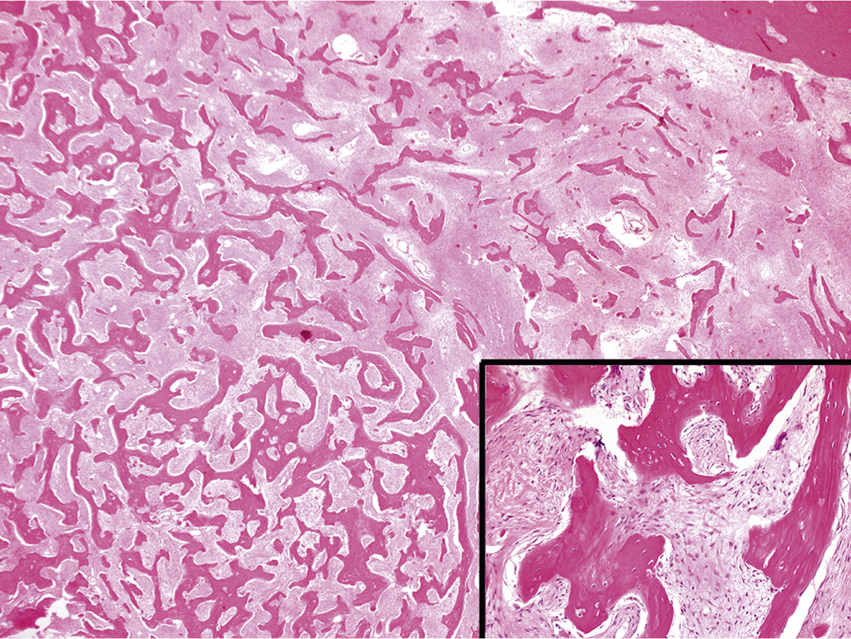
FIGURE 11.36 Fibrous Dysplasia, 20×.
Fibrous stroma and irregular trabeculae of bone that are said to resemble Chinese characters. The bony trabeculae lack osteoblastic rimming (inset lower right, 400×). Fibrous dysplasia is a developmental fibro-osseous lesion with mutations involving the GNAS-1 gene. It can be monostotic (involving a single bone only) or polyostotic. The polyostotic form can be associated with endocrine disorders, most commonly precocious puberty.
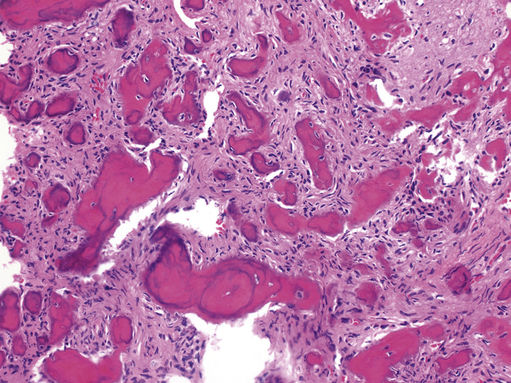
FIGURE 11.37 Ossifying Fibroma, 100×.
The image shows dense fibrous tissue, short trabeculae of bone, and numerous rounded psammomatoid (calcified psammoma body-like) deposits. Ossifying fibroma is a neoplastic fibro-osseous lesion. All fibro-osseous lesions (fibrous dysplasia, osseous dysplasia, and ossifying fibroma) share similar histology and require clinical and radiologic correlation.
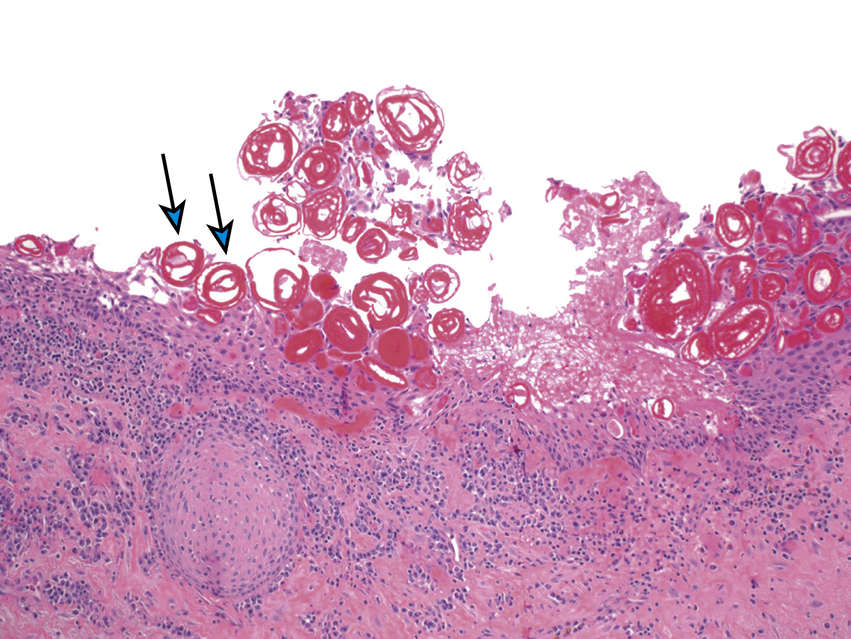
FIGURE 11.38 Periapical (Radicular) Cyst, 100×.
The cyst is lined by stratified squamous epithelium with Rushton (hyaline) bodies (eosinophilic curved glassy structures, arrows). A dense chronic inflammatory cell infiltrate is seen immediately beneath the cyst lining. The periapical cyst is an inflammatory odontogenic cyst, seen at the apex of a non-vital tooth.
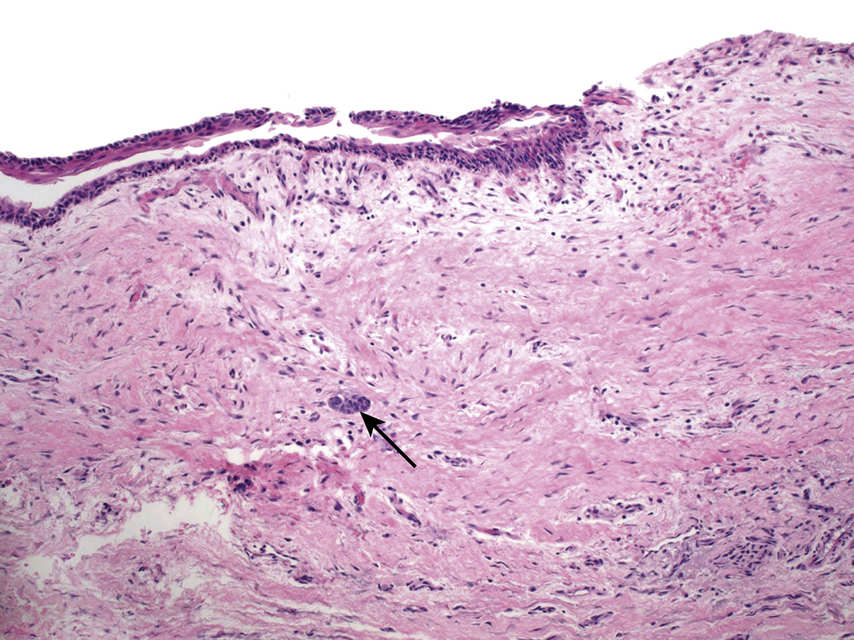
FIGURE 11.39 Dentigerous Cyst, 100×.
The cyst is lined by a thin epithelium that resembles the reduced enamel epithelium of a developing tooth. Small islands of odontogenic epithelium (arrow) are sometimes seen in the connective tissue wall of the cyst. A dentigerous cyst is a developmental odontogenic cyst and is associated with the crown of an unerupted tooth.
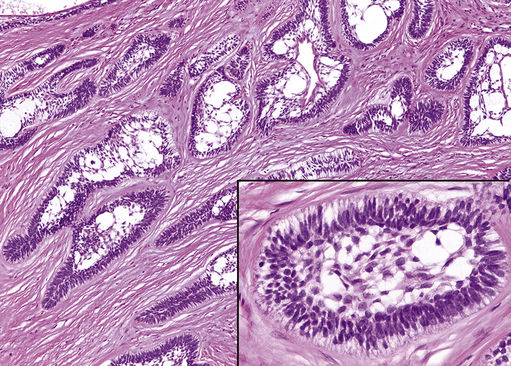
FIGURE 11.40 Solid/Multicystic Ameloblastoma, 100×.
The tumor shows a follicular growth pattern. The follicles are bordered by ameloblast-like cells with reverse nuclear polarity (inset lower right, 400×). The center-most areas often demonstrate cystic degeneration and are characterized by loosely connected cells, stellate reticulum-like cells. The stellate cells may display squamous (acanthomatous), granular, or basal cell differentiation.
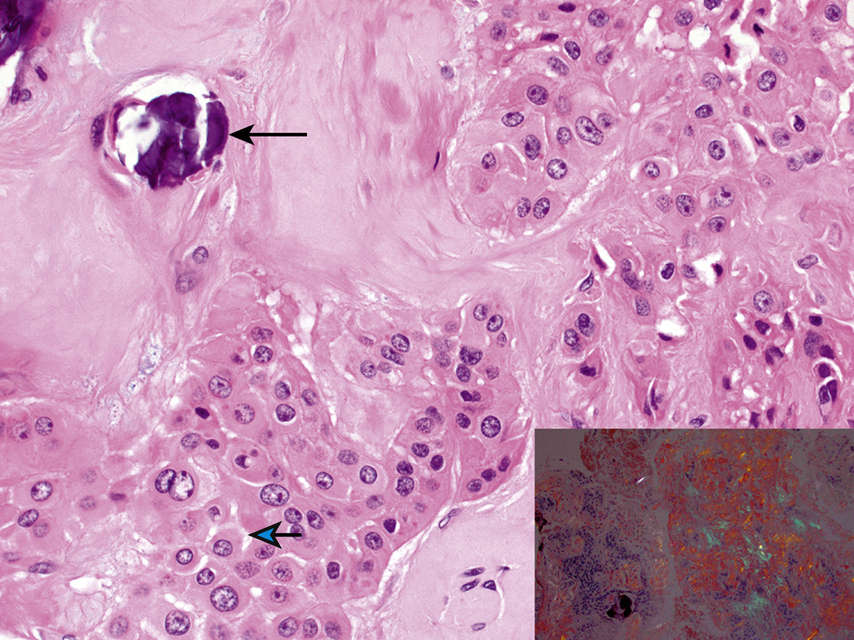
FIGURE 11.41 Calcifying Epithelial Odontogenic Tumor, 200×.
Rounded squamoid (squamous-like) cells with dense eosinophilic cytoplasm, hyperchromatic, moderately pleomorphic nuclei, and prominent nucleoli. Intercellular bridges are clearly visible (arrow). The cells surround globules of pale acellular eosinophilic material (amyloid) with a focus of calcification (arrow). The amyloid demonstrates an apple-green birefringence when viewed under polarized light (inset lower right, 40×).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree