Facial nerve dysfunction after resection of a vestibular schwannoma is one of the most common indications for facial nerve habilitation. This article presents an overview of common and emerging management options for facial habilitation following resection of a vestibular schwannoma. Immediate and delayed nerve repair options, as well as adjunctive surgical, medical, and physical therapies for facial nerve dysfunction, are discussed. Two algorithms are provided as guides for the assessment and treatment of facial nerve paralysis after resection of vestibular schwannoma.
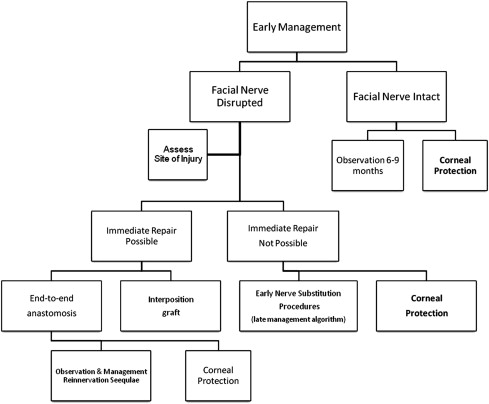
The incidence of facial nerve dysfunction after resection of vestibular schwannoma has significantly decreased with the widespread use of microsurgical techniques. Postoperative facial nerve weakness, however, is reported in 8% to 20% of patients in the immediate postoperative period. The incidence is as high as 25% of patients when delayed postoperative paralysis is considered. Facial nerve paralysis associated with resection of a vestibular schwannoma remains the most common indication for facial nerve habilitation. This overview presents common and emerging management options for facial habilitation after resection of vestibular schwannoma. Immediate and delayed nerve repair options, as well as adjunctive surgical, medical, and physical therapies for facial nerve dysfunction, are discussed. Two algorithms ( Figs. 1 and 2 ) are provided as guides for the assessment and treatment of facial nerve paralysis after resection of vestibular schwannoma.
Early postoperative assessment and management of facial paralysis
Consultation for facial paralysis after resection of a vestibular schwannoma begins with obtaining the pertinent history, which includes determination of preoperative facial nerve function and inquisition regarding the functional and anatomic findings of the facial nerve during surgery; for example, whether increased stimulation thresholds were required with intraoperative electromyographic monitoring, whether the nerve was stretched, or whether the nerve was transected. If the nerve was transected, it is important to know if an anastomotic procedure was attempted. Examination of the patient provides clinical assessment of the facial nerve function. The House-Brackmann (HB) scoring system ( Table 1 ) is the ideal facial nerve grading system for these patients. The HB score should be documented at each visit. A complete cranial nerve examination must also be performed because other cranial nerve deficits (IX–XII), although uncommon, may alter management decisions. The ophthalmic division of the trigeminal nerve and the hypoglossal nerve are particularly important in this regard.
| Grade | Description | Characteristics |
|---|---|---|
| I | Normal | Normal facial function in all areas |
| II | Mild dysfunction | Gross: slight weakness noticeable on close inspection; may have very slight synkinesis At rest: normal symmetry and tone Motion: forehead, slight to moderate movement; eye, complete closure with minimal effort; mouth, slight asymmetry |
| III | Moderate dysfunction | Gross: obvious but not disfiguring difference between the 2 sides; noticeable but not severe synkinesis At rest: normal symmetry and tone Motion: forehead, slight to moderate movement; eye, complete closure with effort; mouth, slightly weak with maximal effort |
| IV | Moderately severe dysfunction | Gross: obvious weakness and or/disfiguring At rest: normal symmetry and tone Motion: forehead, none; eye, incomplete closure; mouth, asymmetric with maximum effort |
| V | Severe dysfunction | Gross: only barely perceptible motion At rest: asymmetry Motion: forehead, none; eye, incomplete closure; mouth, slight movement |
| VI | Total paralysis | No movement |
Early management decisions are based on clinical examination results and surgical details (see Fig. 1 ). If the facial nerve is disrupted during surgery, the gold standard for repair is primary end-to-end anastomosis or interposition grafting. When a truncated proximal facial nerve does not allow for primary repair, nerve substitution procedures may be considered early in the postoperative management (see Fig. 2 ). In some cases, nerve substitution procedures may be considered during the initial hospitalization. All of these patients require corneal protection.
If the facial nerve is anatomically intact at the end of surgery, the chance for regeneration is good, and provision of corneal protection is the most important treatment during the acute hospitalization and immediate future. These patients may be treated conservatively, with observation and delay of definitive treatment for 6 months to 1 year.
Immediate Facial Nerve Repair: Primary Anastomosis and Interposition Grafting
There is a disruption of the facial nerve in an estimated 2% to 10% of vestibular schwannoma resections. When this occurs, the gold standard is to repair with primary end-to-end anastomosis or interposition nerve grafting. The repair of the facial nerve follows standard microsurgical techniques, with delicate and minimal nerve handling and tensionless approximation of the epineurium. The goal of exposing many neurotubules to one another is encouraged by freshening the nerve ends to angles of 45° before anastomosis.
Primary end-to-end anastomosis of the intracranial facial nerve is possible when a proximal stump remains at the brainstem, and is much more difficult in the absence of a proximal stump because the nerve is not covered by epineurium at the brainstem and, therefore, is difficult to suture. Primary end-to-end anastomosis is advantageous to the patient because it addresses the deficit at the time of the initial surgery while possibly avoiding a second operation. In addition, primary repair avoids deficits from the use of a nerve graft for interposition. The main challenge in primary anastomosis involves obtaining a tensionless repair. Often, a tensionless repair necessitates facial nerve rerouting to provide additional length of the distal segment. The ability to perform rerouting and mobilization depends on the surgical approach used for resection and the surgeon’s experience.
Following a translabyrinthine approach:
- •
5 to 10 mm of nerve can be mobilized by drilling out the nerve from the stylomastoid foramen and following it through to the internal auditory canal.
- •
Once the nerve is mobilized and repositioned, it may be sutured to the proximal stump.
- •
Because of the technical difficulty of suturing this anastomosis, fibrin glue has been described as an alternative for fixation of the nerve at the brainstem. Advocates of fibrin glue maintain that there is decreased neural trauma using glue rather than suture.
- •
The tenuous anastomosis (with either suture or fibrin glue) is supported by packing. A variety of materials has been proposed as support, including abdominal fat, fascia, or acellular dermal matrix.
In one case series reporting rerouting with anastomosis at the cerebellopontine angle, 15 of 18 patients achieved HB grade III or IV outcomes.
When primary end-to-end anastomosis is not possible, interposition grafting is the next preferred option. Advantages for the patient are similar to end-to-end repair, except for donor nerve morbidity. Some investigators advocate the use of interposition grafting versus facial nerve rerouting because it is not as technically demanding. In addition, the blood supply of the distal facial nerve remains uninterrupted when using an interposition graft. The main disadvantage of interposition grafting is that there are 2 anastomotic sites. The sural and great auricular nerves are the most commonly used donor nerves. Like primary end-to-end repair, the intracranial anastomotic sites should be surrounded with structural support. Multiple case series using interposition grafting between the intracranial facial nerve and the extratemporal facial nerve report HB scores better than III in 45% to 75% of patients at 1 year.
Corneal Protection
Corneal protection is of paramount importance for patients with facial paralysis. The cornea is at risk secondary to paralytic lagophthalmos and lower lid laxity. The risk is increased when accompanied by a sensory deficit of the ophthalmic division of the trigeminal nerve. The inability to completely close the eye leaves the cornea exposed to foreign bodies and the risk of drying, and may lead to exposure keratitis, abrasions, ulcerations, and abscess formation. Devastating consequences to vision may occur, therefore immediate postoperative protection is essential. Conservative, nonsurgical options should be used in the immediate postoperative period until more definitive options are available. These interventions include frequent use of artificial tears, application of lubricating ophthalmic ointment, and application of an occlusive moisture chamber during sleep.
When patients are at high risk for corneal injury, early surgical intervention should be considered. High-risk patients include those who:
- •
Lack Bell phenomena
- •
Have trigeminal sensory deficits
- •
Are unable to independently perform frequent eye care
- •
Are advanced in age
- •
Have a significant contralateral vision deficit.
Tarsorrhaphy and placement of an upper eyelid weight are the common early procedures performed. In high-risk patients, the treating physician should strongly consider surgical intervention for corneal protection before the patient’s discharge from the initial hospitalization.
Tarsorrhaphy
Tarsorrhaphy involves partially sewing the eyelids together and is a reversible, technically simple procedure that was the standard of care for corneal protection for many years. It remains an option for temporary protection. However, because tarsorrhaphy results in poor cosmetic outcomes and deficits in the visual field, it is reserved for patients with the highest risk of exposure keratitis, those who have failed other options, or those with corneal sensory deficits.
Placement of an upper eyelid weight
Placement of an upper eyelid weight, most commonly a gold weight, is the first-line surgical treatment for paralytic lagophthalmos. Gold weights are well tolerated by patients and can easily be removed if facial function returns. Prefabricated gold weights from 0.6 to 1.8 g are available.
- •
To choose the appropriately sized weight, a sample weight is taped to the upper eyelid, centered over the medial limbus, while the patient is in an upright position.
- •
The weight remains in place for 15 minutes to allow the patient to adapt and make size adjustments.
- •
The largest weight that produces eyelid closure with only slight lid ptosis is chosen.
- •
Placement of an upper eyelid weight can be performed under local anesthesia.
Although widely used, gold-weight placement does carry some risks:
- •
Implant extrusion is the most commonly reported undesired outcome. The literature demonstrates a wide range of implant extrusion from 1% to 45%.
- •
Gold allergy is reported in 3% to 9% of the population.
- •
Local inflammation
- •
Implant bulging
- •
Postoperative infection.
Recently, thin-profile platinum weights have been described as an alternative to gold. The platinum weights are 11% smaller than the gold counterparts because of platinum’s greater density, and allergy to platinum has not been described. Thin-profile platinum weights are implanted using the same surgical procedure as gold weights. Silver and colleagues described their experience with 100 patients who underwent placement of platinum weights; all patients had adequate closure of the eye and only 6% experienced complications, with a 2.9% incidence of extrusion and 2% incidence of local tissue thickening.
The lower eyelid may also need to be addressed to provide adequate corneal protection in patients with facial paralysis. The orbicularis oculi muscle contributes to the integrity and tension of the medial and lateral canthal ligaments. Patients with paralytic lower lid deficits often present with paralytic ectropion, punctal displacement, and epiphora. Older patients with preoperative lower lid laxity and patients who do not undergo reinnervation procedures are at the highest risk. There are several procedures to address the lower lid, including tightening procedures such as the Bick procedure, lateral tarsal strip, medial canthoplasty, and mid-lid wedge resection. Lower lid elevation procedures include augmentation of the inferior tarsus with cartilage grafts or palate mucosa.
Because multiple surgical options are available and the lower lid anatomy is complex, a thorough examination is required before planning the repair.
- •
Normally, the lower lid should sit at the height of the lower limbus or above. A snap test performed by pulling the lower lid down and then allowing it to rebound may ascertain whether excessive laxity is present. A normal lower lid rebounds in 1 to 2 seconds, and a delayed retraction signifies loss of elasticity.
- •
Another helpful examination maneuver is a lateral traction test; this simulates a lid-tightening procedure. If the lateral traction test shows excessive punctum displacement (>2 mm), a medial canthoplasty should be considered.
Most commonly, the authors prefer the Bick procedure or lateral tarsal strip in primary cases.
- •
The Bick procedure involves resecting a lateral wedge of the lower lid, developing a tongue of tarsus, and resuturing the lower lid to the lateral orbital rim ( Fig. 3 ).
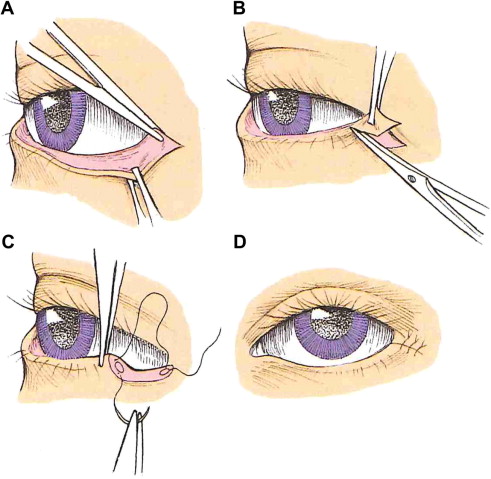
Fig. 3
Bick procedure. ( A–D ) The operative steps performed in a Bick procedure for lower lid ectropion repair.
( From Wackym PA, Rhee JS. Facial paralysis. In: Snow JB, Wackym PA, editors. Ballenger’s otorhinolaryngology. Shelton (CT): People’s Medical Publishing House; 2009. p. 408. Copyright © 2009 BC Decker Inc; with permission.)
- •
The lateral tarsal strip procedure involves performing an inferior lateral cantholysis to free the lower lid. The inferior lateral tarsus is exposed and sutured to the lateral orbital rim.
- •
Vagefi and Anderson described a modification to the standard lateral tarsal strip that includes the addition of a mini-tarsorrhaphy whereby the lower and upper tarsus are denuded and sutured together to create a sharp canthal angle and prevent upper eyelid overhang. This mini-tarsorrhaphy modification is not reversible and should not be performed in patients who may have nerve recovery.
Despite the careful preoperative examination and surgical execution, a significant percentage of patients have recurrence of lower lid laxity and require reoperation.
If a patient is not at high risk for corneal injury, periocular procedures may be performed electively. The decision to perform periocular reanimation procedures is dictated by:
- •
Patient symptoms
- •
Adherence to eye-lubricating regimens
- •
Patient-specific anatomic issues
- •
Timetable of anticipated facial nerve recovery.
Periocular procedures are often performed as an adjunct during a nerve substitution operation, dynamic muscle transposition, or static procedures.
Delayed postoperative assessment and management
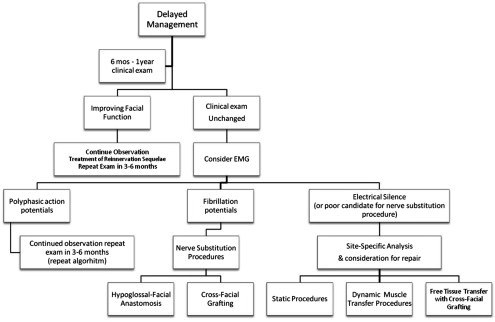
In patients who have complete facial paralysis in the presence of an intact facial nerve at the conclusion of resection, observation for 6 months to 1 year is recommended. This time allows opportunity for the regeneration of facial nerve at the expected 1 mm per day. Physical examination findings with or without facial electromyography (EMG) will dictate management decisions at the time of follow-up (see Fig. 2 ). If any degree of facial movement is detected, continued observation is recommended because improved function is likely anticipated.
If there is no detectable facial muscle tone, facial EMG can be a helpful diagnostic tool. There are 3 outcomes of the EMG:
- 1.
Polyphasic action potentials
- 2.
Fibrillation
- 3.
Electrical silence.
If the EMG shows polyphasic action potentials, neural regeneration is anticipated and nerve substitution procedures are not indicated at this time. The patient may continue to be observed with repeat physical examination in 3 months.
If the EMG demonstrates fibrillation potentials, motor end plates are intact, but there is no evidence of neural regeneration, and nerve substitution procedures should be considered.
When a patient presents with late facial paralysis (usually >2 years), EMG may demonstrate electrical silence, and this indicates nonviable motor end plates. When this occurs, nerve substitution procedures should not be performed and free innervated muscle transfer, dynamic muscle transpositions, or static procedures should be considered.
Although the algorithms (see Figs. 1 and 2 ) presented are meant to guide management decisions, treatment of facial paralysis is complex, and patients may require a combination of procedures to obtain optimal outcomes. A review of nerve substitution procedures, dynamic muscle transpositions, and static procedures follows. Adjunctive therapies for further optimization and management of these patients, including the use of botulinum toxin injections and physical therapy, are also discussed.
Nerve Substitution Procedures: Hypoglossal-Facial Anastomosis and Cross-Facial Grafting
When primary repair of the facial nerve is not possible, nerve substitution operations offer the patient the best outcome to restore meaningful movement and tone. The most commonly used nerve substitution procedures are hypoglossal-facial anastomosis (XII–VII) or cross-facial (VII–VII) grafting. Prerequisites for nerve substitution procedures include the presence of an intact proximal donor nerve, an intact distal facial nerve, and viable motor end plates as determined by EMG. Preoperative counseling includes a clear discussion of realistic surgical expectations. These operations do not restore mimetic voluntary facial movement, but do have the potential to restore tone and symmetry at rest as well as providing some voluntary facial movement with rehabilitation.
The XII-VII substitution operations produce the most consistent results, with approximately 90% of patients demonstrating improved tone and symmetry. Three main techniques exist for XII-VII substitution procedures:
- 1.
Complete hypoglossal-facial nerve anastomosis
- 2.
Partial hypoglossal-facial nerve anastomosis
- 3.
Jump or interposition grafting between the hypoglossal and facial nerves.
Complete hypoglossal-facial nerve anastomosis
- •
The most proximal aspect of the facial nerve is identified at the stylomastoid foramen, and the nerve is dissected away from the parotid up to the main trunk at the pes anserinus.
- •
The hypoglossal nerve is divided distal to the descendens hypoglossi takeoff and then mobilized medial to the digastric muscle.
- •
A tensionless anastomosis is performed to the proximal facial nerve ( Fig. 4 ).
Fig. 4
Hypoglossal-facial substitution procedure. The hypoglossal nerve is mobilized medial to the digastric muscle and sutured to the proximal facial nerve.
( From Wackym PA, Rhee JS. Facial paralysis. In: Snow JB, Wackym PA, editors. Ballenger’s otorhinolaryngology. Shelton (CT): People’s Medical Publishing House; 2009. p. 408. Copyright © 2009 BC Decker Inc; with permission.)
The main disadvantage to end-to-end anastomosis is the sacrifice of the ipsilateral hypoglossal nerve, which leads to hemitongue atrophy. The findings of a recent meta-analysis of hypoglossal-facial anastomosis demonstrated a resulting difficulty with speech in 10% to 60% of patients and swallowing in 20% to 46% of patients. If a patient has concomitant lower cranial nerve deficiencies (IX and X), postoperative hypoglossal dysfunction may exacerbate the deficits and lead to increased difficulty with speech and swallowing.
Partial hypoglossal-facial nerve anastomosis and jump graft
Because of the postoperative hypoglossal deficits, alternatives such as partial end-to-side hypoglossal-facial anastomosis and interposition jump grafts have been developed. Although these techniques can also be associated with some degree of tongue atrophy, clinically significant negative outcomes on speech and swallowing have not been described. In these techniques, the hypoglossal nerve is not fully transected at the site of the anastomosis; rather, a small wedge that is no more than one-half of the diameter of the nerve is used. The facial nerve or interposition graft is sutured via an end-to-side anastomosis to the hypoglossal nerve.
Partial hypoglossal-facial nerve anastomosis
The partial hypoglossal-facial anastomosis is an attractive technique because only 1 anastomosis is performed and the power source, the hypoglossal nerve, is directly connected to the facial nerve. The primary difficulty with end-to-side hypoglossal-facial anastomosis is the mobilization of the nerves to approximate one another, and often the facial nerve requires mobilization. The intratemporal segment of the facial nerve may be mobilized from the stylomastoid foramen through the mastoid segment with the goal of mobilizing enough proximal facial nerve to suture it to the hypoglossal nerve proximal to the takeoff of the descendens hypoglossi. A modification that has been described is to combine a parotid release procedure with the intratemporal mobilization to provide an additional 3 to 5 mm of facial nerve. The end-to-side hypoglossal-facial anastomosis has produced satisfactory facial function without hemitongue atrophy.
Jump graft
The interposition jump graft is an attractive alternative because, similar to the partial end-to-side anastomosis, it also avoids difficulties in speech and swallowing. The procedure is also performed without retrieval of the facial nerve from the mastoid. Options for nerve graft include the great auricular nerve and sural nerve. Typically the great auricular nerve can be harvested from the same incision, and is the preferred graft. The graft is anastomosed end-to-side to the hypoglossal nerve and end-to-end to the proximal facial nerve ( Fig. 5 ). Disadvantages of this technique include 2 anastomotic sites and donor nerve morbidity (although minimal). Outcomes are similar to hypoglossal-facial anastomosis, with greater than 80% HB grade III. A representative successful case of a partial XII-VII jump graft is shown in Fig. 6 .

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


