Purpose
To describe and compare graft survival and intraocular pressure (IOP) control after penetrating keratoplasty (PK) and Descemet stripping automated endothelial keratoplasty (DSAEK) in eyes with iridocorneal endothelial (ICE) syndrome.
Design
Retrospective case series.
Methods
Multicenter study conducted at the Singapore National Eye Centre (SNEC) and Price Vision Group. Twenty-nine consecutive eyes with ICE syndrome that underwent keratoplasty between 1991 and 2011 were identified from the SNEC transplant database and Price Vision Group patient database and the following data extracted: demographics, graft failure, IOP indices, and glaucoma treatment pre- and postkeratoplasty. The main outcome measures were graft failure and need for additional IOP-lowering treatment after keratoplasty.
Results
The mean follow-up duration was 7.0 ± 4.9 years in the PK group (n = 17) and 4.0 ± 2.6 years in the DSAEK group (n = 12). After a mean of 4.1 ± 3.1 years, 37.9% of grafts (11/29) failed: 7 PK compared to 4 DSAEK ( P = .72). The graft failure rate was 50% in eyes with prekeratoplasty glaucoma surgery vs 31.6% in those without ( P = .43). Additional glaucoma treatment was required in 37.9% of eyes (11/29): 41.2% of PK eyes and 50% of DSAEK eyes ( P = .28) Eyes that had undergone glaucoma surgery before keratoplasty were less likely to require escalation of IOP-lowering therapy postkeratoplasty (9.1% vs 50%, P = .03).
Conclusions
One-third of grafts failed after keratoplasty for ICE syndrome at a mean duration of 4 years and additional IOP-lowering treatment was required in 37.9%. Both PK and DSAEK had similar outcomes with regard to graft failure and IOP control.
Iridocorneal endothelial (ICE) syndrome is typically a unilateral, nonfamilial condition, affecting mainly white women in early to middle adulthood. ICE is variably associated with corneal endothelial changes, edema, anterior chamber angle abnormalities (broad peripheral anterior synechiae attached at or anterior to the Schwalbe line), iris stromal alterations (ie, correctopia, iris atrophy, nodules, and holes), and secondary glaucoma. Chandler syndrome, essential iris atrophy, and Cogan-Reese syndrome are 3 clinical manifestations of ICE syndrome. Approximately half of all patients with ICE syndrome develop glaucoma and those with essential iris atrophy tend to develop a more severe form of glaucoma compared to those with Chandler syndrome. The glaucoma associated with ICE syndrome is often difficult to treat.
Keratoplasty is indicated if the corneal endothelial cell loss produces irreversible corneal edema causing pain or reduced visual acuity (VA) to a point where it interferes with a patient’s normal activities. There are very few publications reporting the outcomes of penetrating keratoplasty (PK) in ICE syndrome, in part owing to the relative rarity of this condition and the difficulty in obtaining a large series. In addition, those few publications did not examine graft survival in relation to pre- and postkeratoplasty glaucoma treatment. Endothelial keratoplasty (EK) techniques, which selectively replace diseased endothelium, offer many advantages compared with PK for treatment of endothelial dysfunction. Because of their superior visual rehabilitation compared to PK, EK techniques have become the gold standard for endothelial replacement and more recently have been used in patients with ICE syndrome. Several studies have demonstrated good short-term EK graft survival in patients with ICE syndrome, but it is still unclear how the long-term graft survival would compare with PK. The aim of this study was to describe the effect of keratoplasty on corneal graft survival and intraocular pressure (IOP) control in eyes with ICE syndrome, and to compare these outcomes between PK and Descemet stripping automated endothelial keratoplasty (DSAEK).
Methods
Consecutive eyes with ICE syndrome that underwent keratoplasty (either PK or DSAEK) between 1991 and 2011 were identified from the Singapore National Eye Centre corneal transplant database and Price Vision Group (Indianapolis, Indiana, USA) patient database. Case records were retrospectively reviewed and the following data extracted: demographics (age at time of keratoplasty, sex, and race); type of keratoplasty (DSAEK or PK); duration of follow-up; prekeratoplasty best-corrected visual acuity (BCVA), IOP indices, gonioscopic findings, glaucoma interventions and treatment, additional intraoperative procedures at the time of keratoplasty; and postkeratoplasty IOPs at 1, 3, 6, and 12 months, IOP indices (mean IOP, mean IOP change, and IOP range), IOP-lowering treatment, and graft failures. Approval for the conduct of this study and for informed consent was given by the Singhealth Centralised Institutional Review Board and the study adhered to the tenets of the Declaration of Helsinki. The diagnosis of ICE was based on the presence of at least 2 of the following 3 principal diagnostic criteria : unilateral corneal edema caused by an abnormality of the corneal endothelium or a typical unilateral hammered-silver appearance of the posterior cornea; iris atrophy with corectopia, ectropion uveae, holes, or nodules in the same eye as the corneal changes; and broad peripheral anterior synechiae (PAS) or other iridocorneal adhesions at or anterior to the Schwalbe line. Based on the nature of the iris abnormalities, patients were further classified into the 3 subcategories of ICE syndrome: Chandler syndrome if iris atrophy was minimal or absent; essential iris atrophy if there was marked iris atrophy, with or without holes; and Cogan-Reese syndrome if iris nodules were present.
Prior to initiation of DSAEK at Singapore National Eye Centre in 2006, all cases underwent PK surgery. A PK was defined as an allograft procedure involving full-thickness replacement of corneal tissue. A standard technique using a Hanna vacuum trephine system (Moria SA, Antony, France) was performed. The recipient cornea was excised with the Hanna trephine, after which a 0.25- to 0.50-mm oversized donor cornea was punched out and sutured onto the recipient with 10-0 nylon. Intraoperative astigmatic control was achieved using a microscope-mounted keratoscope. A bandage contact lens was placed at the end of the surgery, and subconjunctival steroids and antibiotics were injected. DSAEK was performed as previously described. Briefly, the donor graft was prepared using an automated lamellar therapeutic keratoplasty microkeratome (Moria USA, Doylestown, Pennsylvania, USA). Trephination was performed using a standard Hanna punch trephine system with standard trephine sizes between 7.75 mm and 9 mm, at 0.25-mm increments. A 5-mm scleral tunnel incision was made in all cases. Stripping of the Descemet membrane and endothelium was performed with a reverse Sinskey hook under air followed by insertion of the donor graft by the taco-folded technique, the Sheets glide-insertion technique, or the Endoglide technique. An air bubble was injected into the anterior chamber for tamponade and to enhance donor graft adhesion, while drainage of interface fluid was achieved through 4 paracentral vents. The air bubble was partially evacuated before the end of the procedure to leave an air bubble approximating the size of the graft. Additional intraoperative procedures at time of keratoplasty included lens extraction by manual expression or phacoemulsification, synechiolysis, and vitrectomy. Postoperatively, the steroid regimen was similar in the DSAEK and PK group. Patients were given a combination of topical steroids (prednisolone acetate 1% or dexamethasone phosphate 0.1%), initially at 3-hour intervals, with topical antibiotics (tobramycin, levofloxacin, or moxifloxacin). Topical steroids were tapered gradually, reducing to 4 times daily at 1 month, 3 times daily at 5 months, twice daily at 9 months, and once daily by the end of 12 months. Patients with steroid-related IOP increase were given lower-strength steroids (ie, prednisolone mild 0.12%) and tapered similarly.
IOP measurements were not corrected for corneal thickness. The mean postoperative IOP for each eye was calculated by averaging the IOP measurements at different postoperative times. Change in IOP before and after keratoplasty was calculated by subtracting the preoperative IOP from the postoperative average IOP for each eye, and the mean of all the differences was calculated for all included eyes. The mean highest IOP after keratoplasty was calculated by dividing the sum of all the highest IOPs of each eye after keratoplasty by the number of eyes. Similarly, the mean lowest IOP after keratoplasty was calculated by dividing the sum of all the lowest IOPs of each eye after keratoplasty by the number of eyes. IOP range after keratoplasty was the difference between the minimum and maximum IOP recorded after keratoplasty, and the mean range of IOP after keratoplasty was calculated by dividing the sum of differences between maximum and minimum IOP of each eye after keratoplasty by the number of eyes. All IOP parameters and analyses were based on IOP measured at 1, 3, 6, and 12 months postkeratoplasty.
The main outcome measures were graft failure and the need for additional IOP-lowering treatment after keratoplasty. Corneal graft failure was defined as irreversible edema and loss of graft clarity in a graft that was previously documented to be thin and clear. Additional IOP-lowering treatment after keratoplasty was defined as the need for additional topical or systemic IOP-lowering medication; the performance of glaucoma filtration surgery or bleb needling with adjunctive antimetabolites, for example, 5-fluorouracil, to rescue an existing failing bleb; or glaucoma laser procedures aimed at lowering IOP after keratoplasty, or a combination thereof. Additional IOP-lowering treatment was instituted when the intraocular pressure was deemed too high for the degree of glaucomatous damage. Statistical analysis was performed using SPSS software version 19.0 (SPSS, Inc, Chicago, Illinois, USA). Kaplan-Meier survival analysis was used to estimate the risk of graft failure, and survival curves were compared between our Singapore and US sites with the log-rank test. Analysis was performed using the χ 2 or t test as appropriate. A linear mixed-effects model multivariate analysis was used to analyze the multivariate relationship of IOP with graft failure, type of keratoplasty, age, sex, and prior glaucoma operations. Statistical significance was defined as P < .05.
Results
A total of 29 eyes with ICE syndrome underwent keratoplasty between 1991 and 2011 and were followed up for 5.8 ± 4.3 (mean ± standard deviation) years (range 0.04–15.3 years). Table 1 shows the baseline characteristics of these eyes. The mean follow-up duration was 7.0 ± 4.9 years in the PK group (n = 17) and 4.0 ± 2.6 years in the DSAEK group (n = 12). This difference was not statistically significant ( P = .070). All eyes were included for analysis. Seventeen (58.6%) were female, 17 (58.6%) were Chinese, and 5 (17%) were white. The mean age was 55.3 ± 10.2 years at time of keratoplasty. Prior to keratoplasty, these eyes had a mean IOP of 14.7 ± 3.9 mm Hg, were on a median of 1.0 topical IOP-lowering medications (range 0–3), and had 2.0 ± 0.9 quadrants of angle closure on gonioscopy. Sixteen (94.1%) in the PK group and 6 (50%) in the DSAEK group had a BCVA of 6/60 or worse. Seventeen eyes (58.6%) underwent PK and 12 (41.4%) underwent DSAEK. Sixteen had additional surgical procedures at time of keratoplasty. These were cataract extraction (n = 14), either by manual expression (n = 7) or phacoemulsification (n = 7); synechiolysis (n = 4); and pupilloplasty (n = 1).
| All (n = 29) | SNEC (n = 23) | Price (n = 6) | P Value a | |
|---|---|---|---|---|
| Age at time of keratoplasty, y | 55.3 ± 10.17 | 55.7 ± 10.85 | 53.7 ± 7.55 | .669 |
| Duration of follow-up, y | 5.24 ± 4.35 | 5.91 ± 4.53 | 2.65 ± 2.30 | .101 |
| Prekeratoplasty IOP, mm Hg | 14.7 ± 3.95 | 15.0 ± 3.93 | 13.2 ± 4.09 | .365 |
| Number of prekeratoplasty glaucoma medications | 0.86 ± 1.03 | 0.83 ± 1.07 | 1.00 ± 0.89 | .719 |
| Female sex, n (%) | 17 (58.6) | 14 (60.9) | 3 (50) | .669 |
| Prekeratoplasty glaucoma surgery, n (%) | 10 (34.5) | 7 (30.4) | 3 (50.0) | .633 |
| Type of keratoplasty, n (%) | ||||
| PK | 17 (58.6) | 17 (73.9) | 0 (0.0) | .002 |
| DSAEK | 12 (41.4) | 6 (26.1) | 6 (100) | |
a For comparison between centers, using χ 2 tests for categorical variables and 2-sample t tests for continuous variables.
Graft Failures
Eleven grafts (37.9%) failed, at a mean of 3.8 ± 3.0 years after keratoplasty (range 0.9–9.1 years). Seven of the 11 graft failures (63.6%) were PK grafts and 4 (36.4%) were DSAEK grafts. A total of 41.2% (7/17) of PK grafts failed compared to 33.3% (4/12) of DSAEK grafts ( P = .720). On Kaplan-Meier survival analysis, there was no significant difference in probability of cumulative survival between PK grafts and DSAEK grafts. The estimated mean survival of PK grafts was 9.0 years (95% confidence interval [CI] 5.7–12.2) compared to 4.5 years (95% CI 3.9–5.0) for DSAEK grafts in our Singapore site and 4.7 years (95% CI 2.0–7.4) for DSAEK grafts in our US site (log-rank P = .380 for PK vs DSAEK survival curves, and log-rank P = .259 for SNEC DSAEK vs Price DSAEK survival curves, Figure ). Graft rejection preceded graft failure in 2 cases (both PK grafts), while endothelial decompensation was the cause of graft failure in the other 9 cases. There were no differences in ICE syndrome subtypes between PK and DSAEK groups ( P = .393).
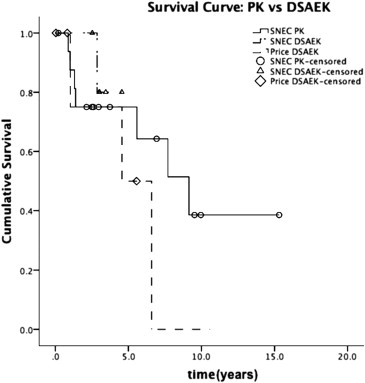
Compared to eyes without graft failure, those with graft failure were followed up for a longer duration (7.8 ± 4.4 years vs 4.4 ± 3.9 years, P = .042), had lower prekeratoplasty IOP (12.6 ± 3.6 mm Hg vs 15.8 ± 3.7 mm Hg, P = .041), had lower mean postkeratoplasty IOP (15.5 ± 3.1 mm Hg vs 18.7 ± 3.8 mm Hg, P = .032), and required fewer postkeratoplasty IOP medications (0.6 ± 1.0 vs 1.5 ± 1.1, P = .029) ( Table 2 ).
| Risk Factor | Graft Failures (n = 11) | Non-Graft Failures (n = 18) | P Value a | Estimated Difference and 95% Confidence Interval of Difference b |
|---|---|---|---|---|
| Age at time of keratoplasty, y | 54.7 ± 10.22 | 55.7 ± 10.40 | .776 | −9.47<−1.00<7.11 |
| Duration of follow-up, y | 7.8 ± 4.4 | 4.5 ± 3.8 | .042 | 0.1<3.3<6.6 |
| Prekeratoplasty IOP, mm Hg | 12.6 ± 3.6 | 15.8 ± 3.7 | .041 | −6.2<−3.2<−0.2 |
| Number of quadrants closed on gonioscopy | 2.1 ± 0.7 | 2.0 ± 1.0 | .297 | −0.6<0.1<0.8 |
| Number of prekeratoplasty glaucoma medications | 0.6 ± 1.0 | 1.0 ± 1.0 | .891 | −1.2<−0.4<0.4 |
| Mean IOP postkeratoplasty, mm Hg | 15.5 ± 3.1 | 18.7 ± 3.8 | .032 | −6.1<−3.3<−0.4 |
| Maximum IOP postkeratoplasty, mm Hg | 19.9 ± 5.5 | 24.0 ± 6.8 | .113 | −9.1<−4.1<0.93 |
| Minimum IOP postkeratoplasty, mm Hg | 12.0 ± 3.1 | 14.6 ± 3.4 | .050 | −5.2<−2.6<0.04 |
| Number of IOP medications postkeratoplasty | 0.6 ± 1.0 | 1.5 ± 1.1 | .029 | −1.8<−0.9<−0.1 |
| Ethnicity, n (%) | ||||
| Chinese | 7 (63.6) | 10 (55.6) | .723 | |
| Non-Chinese | 4 (36.4) | 8 (44.4) | ||
| Female sex (%) | 6 (54.5) | 11 (61.1) | 1.000 | |
| Prekeratoplasty BCVA, n (%) | ||||
| 6/60 or worse | 8 (72.7) | 14 (77.8) | 1.000 | |
| Better than 6/60 | 3 (27.3) | 4 (22.2) | ||
| Prekeratoplasty glaucoma surgery, n (%) | 5 (45.5) | 5 (27.8) | .432 | |
| Type of keratoplasty, n (%) | ||||
| PK | 7 (63.6) | 10 (55.6) | .720 | |
| DSAEK | 4 (36.4) | 8 (44.4) | ||
| Received other procedures during keratoplasty (%) | 7 (63.6) | 9 (50) | .698 | |
| Received additional glaucoma treatment postkeratoplasty (%) | 2 (18.2) | 9 (50) | .131 | |
| Received glaucoma operation postkeratoplasty (%) | 1 (9.1) | 4 (22.2) | .622 | |
a For comparison between individuals with and without graft failure, using χ 2 tests for categorical variables and 2-sample t tests for continuous variables.
b Estimated difference given by graft failures minus nongraft failures.
Ten patients had prekeratoplasty glaucoma surgery. Of these, 4 underwent trabeculectomy, 4 had implantation of glaucoma drainage devices (GDD), and 2 underwent glaucoma drainage device surgery after failed trabeculectomy. Graft failure occurred in 3 out of 4 eyes with GDD, 2 out of 2 eyes that underwent both trabeculectomy and GDD, and none of the eyes that underwent trabeculectomy alone. Graft failure was more common in eyes that had prekeratoplasty GDD surgery than in eyes that had trabeculectomy alone (83.3% vs 0%, P = .048).
Additional Glaucoma Treatment Postkeratoplasty
In our series, 16 of 29 eyes (55.2%) had received IOP-lowering therapy prior to keratoplasty: 6 (37.5%) of these were on topical medications alone, while 10 (62.5%) had prior glaucoma surgery. The mean postkeratoplasty IOP was 17.4 ± 3.8 mm Hg. The maximum, minimum, and range of postkeratoplasty IOP were 22.4 ± 6.5 mm Hg, 13.6 ± 3.5 mm Hg, and 8.7 ± 5.7 mm Hg, respectively. Of these 16 eyes with glaucoma prior to keratoplasty, 5 (31.3%) required additional IOP-lowering therapy postkeratoplasty. Of these 5 eyes, 4 were on topical medications alone and 1 had previous trabeculectomy with mitomycin C prior to keratoplasty. Of the 4 eyes that were medically controlled prior to keratoplasty, 2 required additional topical medications, 1 had a trabeculectomy with mitomycin C, and 1 had Ahmed tube implantation. Repeat GDD surgery was performed for the eye that had previous trabeculectomy. Overall, 11 of 29 eyes (37.9%) required additional topical IOP-lowering therapy after keratoplasty: 7 (41.2%) in the PK group and 4 (33.3%) in the DSAEK group. Of these 11 eyes, 4 (36.4%) required glaucoma surgery: 2 (11.8%) in the PK group and 2 (16.6%) in the DSAEK group. In each group, 1 had trabeculectomy and 1 had GDD. One patient had trabeculectomy with 5-fluorouracil prior to PK and subsequently underwent GDD surgery post-PK.
We assessed factors that may affect the need for additional glaucoma treatment postkeratoplasty ( Table 3 ). Eyes that underwent prekeratoplasty glaucoma surgery were less likely to require additional IOP-lowering therapy postkeratoplasty (9.1% vs 50%, P = .034) than those that did not. Grafts in these eyes with prekeratoplasty glaucoma surgery tended to fail more frequently (50% vs 32.6%, P = .432), although the difference did not reach statistical significance. There was no significant difference between GDD and trabeculectomy in the need for additional IOP therapy postkeratoplasty (0% vs 25%, P = .400). There were 14 patients with Chandler syndrome, 9 with essential iris atrophy, 1 with Cogan-Reese syndrome, and 5 with unknown ICE syndrome subtype. There was no significant difference in the need for additional IOP therapy postkeratoplasty between eyes with Chandler syndrome and eyes with essential iris atrophy or Cogan-Reese syndrome (30% vs 42.9%, P = .678).



