Purpose
To determine the clinical outcomes after glued trans-scleral posterior chamber intraocular lens (IOL) exchange for anterior chamber (AC) IOL.
Design
Retrospective case series.
Methods
Eyes with AC IOL explantation with glued IOL implantation in a single setting at the Dr Agarwal Eye Hospital and Eye Research Centre, Chennai, India, from 2008 through 2012 were included. Data were collected from the patient records.
Main Outcome Measures
Corrected distance visual acuity (CDVA) in logarithm of the minimal angle of resolution units, intraocular pressure, AC reaction, AC depth, central corneal thickness, central macular thickness, endothelial cell density, and endothelial cell loss (percentage) were determined before and after IOL exchange.
Results
Thirty-eight eyes with mean follow-up of 24.1 ± 15.4 months (range, 8 to 60 months) were analyzed. The indications were corneal decompensation (39.4%), malpositioned AC IOL (28.9%), uveitis (15.7%), glaucoma (13.1%), and broken haptic (2.6%). There was significant improvement in the postoperative CDVA ( P = .000) and central corneal thickness ( P = .000) after AC IOL removal. CDVA better than 20/60 was obtained in 65.7% eyes. Thirty-four (86.8%) eyes showed an increase in the CDVA after IOL exchange. The mean endothelial cell loss was 3.4 ± 2.4% (range, 0.13% to 10.5%). There was significant correlation between the CDVA and the central corneal thickness ( P = .000). There was significant change in the AC depth ( P = .000), the intraocular pressure ( P = .005), and the AC inflammation ( P = .000) after IOL exchange. The preoperative macular edema in 3 eyes resolved after surgery (mean central macular thickness, 205.6 ± 7.2 μm).
Conclusions
Glued trans-scleral fixated posterior chamber IOL exchange for AC IOL can be an excellent alternative in eyes with ocular complications related to AC IOL.
For the past 2 decades, anterior chamber (AC) intraocular lenses (IOLs) have been an established method of IOL implantation in those eyes with deficient capsules either as a primary or secondary procedure. AC IOLs are preferred over sulcus sutured IOLs because they are easier to implant technically, are reasonably well tolerated, and have a low rate of postoperative IOL decentration or tilt. They also have shown lesser probability of posterior segment complications and have been used widely under emergencies of intraoperative capsular loss or dialysis. However, it has to be remembered that the AC IOLs have become one of the causes of IOL exchange and explantation in the last 2 decades. A study reported that AC IOLs comprise 66.7% of the removed lenses in a pseudophakic eye. Implant-related problems like discrepancies between the AC biometry and the IOL size may cause pseudophakodonesis in the aqueous, resulting in progressive endothelial cell loss. Infrequently, AC IOL–iris contact may lead to pigment dispersion with subsequent inflammation. Occasionally, secondary angle closure and glaucoma with corneal decompensation resulting from haptic displacement may develop. For these reasons, eyes with a shallow AC or early corneal guttata have been treated as relative contraindications for AC IOL.
IOL exchange has been indicated in situations where the existing lens has been recognized as the cause for the ocular complication. The most frequent indications for AC IOL explantation have been pseudophakic bullous keratopathy, uveitis-glaucoma-hyphema syndrome, and cystoid macular edema. Whenever an AC IOL has been explanted, a scleral suture-fixated IOL or another AC IOL has been placed. In our study, the explanted AC IOL was replaced by a sutureless glued trans-scleral fixated posterior chamber (PC) intraocular lens (IOL). The main advantage of a glued IOL is its anatomic position, which is similar to that of the normal lens with no suture-related complications. In this study, we analyzed the indications, visual outcomes, and complications after AC IOL explantation with glued PC IOL fixation. Although there have been studies of AC IOL exchange, there are no published data on the outcomes of AC IOL exchange with glued IOL
Methods
A preoperative informed consent for the surgery was obtained from all patients, and the retrospective review of patient data was approved by the Academics and Research Board of Dr Agarwal’s Eye Hospital and Eye Research Centre, Chennai, India, and the tenets of Declaration of Helsinki were followed. In this retrospective study, case files of patients who underwent glued IOL in exchange of AC IOLs from January 2008 through December 2012 in the practice of Dr Agarwal’s Eye Hospital and Eye Research Centre were included. In 5-year period, there were 82 eyes that underwent IOL exchange with glued IOL. Of 82 eyes, 34 eyes that had a PC IOL in the AC, an iris-sutured IOL, or AC IOL explantations with corneal transplantation and 10 eyes with follow-up of less than 6 months were excluded. Preoperative ocular evaluation included uncorrected distance visual acuity (UDVA), spectacle-corrected distance visual acuity (CDVA), intraocular pressure (IOP) using noncontact tonometry, slit-lamp biomicroscopy, dilated fundus examination, AC cellular reaction by optical coherence tomography (Visante, Carl Zeiss Meditec, Dublin, California, USA), central macular thickness (CMT; Cirrus OCT, Carl Zeiss Meditec), potential visual acuity (retinal acuity meter), corneal thickness (Visante OCT, Carl Zeiss Meditec), gonioscopy, AC depth (ACD), fundus photography, and clinical slit-lamp photography. Endothelial cells were evaluated by specular count (SP-2000P; Topcon, Tokyo, Japan). Endothelial cell density (ECD) and percentage loss were determined.
There were 3 probable clinical results expected after the IOL exchange, which included the improvement of preoperative status, worsening of preoperative status, and finally the persistence of preoperative status. Improvement in the clinical condition has been defined as a decrease in corneal edema, a decrease in AC cell count, a decrease in the IOP, and increase in CDVA of more than 2 lines, or a decrease in CMT. Worsening was defined as progressive corneal decompensation, an increase in the IOP, an increase in AC cell count, an increase in CMT, and loss of CDVA of more than 2 lines. Persistence of the clinical condition was defined as there being no change in any of the preoperative parameters. Symptomatic changes after IOL exchange were obtained through a subjective questionnaire enclosed with the case files during the follow-up period. Late postoperative complications were complications that occurred more than 1 month after glued IOL surgery. Reoperation for a complication was defined as additional surgery requiring a return to the operating room to manage a surgical complication, such as an IOL repositioning or vitrectomy. Serious complications were defined as surgical complications that produced loss of 2 lines or more of CDVA.
Surgical Technique
Preparation for glued intraocular lens
Under peribulbar anesthesia, localized peritomy at the intended site of scleral flaps was performed. Two partial-thickness limbal-based scleral flaps ( Figure 1 ) approximately 2.5 × 2.5 mm were created exactly 180 degrees diagonally (using a scleral marker) apart. An infusion cannula (23 or 20 gauge) or AC maintainer was inserted for infusion. Two straight sclerotomies with a 20-gauge needle were made approximately 1 to 1.5 mm from the limbus under the existing scleral flaps.
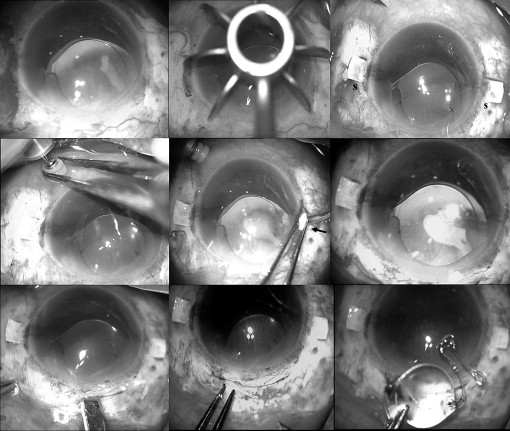
Anterior chamber intraocular lens removal
A small scleral tunnel incision was made with a 3.2-mm keratome and the incision was beveled forward. The AC was filled with an ophthalmic viscoelastic device. Anterior vitrectomy in the AC and pupillary region was performed to reduce the vitreous traction. The scleral wound then was extended to 6 mm ( Figure 1 ). The existing IOL in the AC was removed by McPherson forceps under the cushioning effect of the ophthalmic viscoelastic device to prevent intraoperative endothelial touch. Techniques used for AC IOL explantation included the synechiae release, ophthalmic viscoelastic device injection, hinging, moving the optic, rotating the remainder of the lens through the incision, and transection with Vannas scissors (Appasamy Associates, Chennai, India) if necessary.
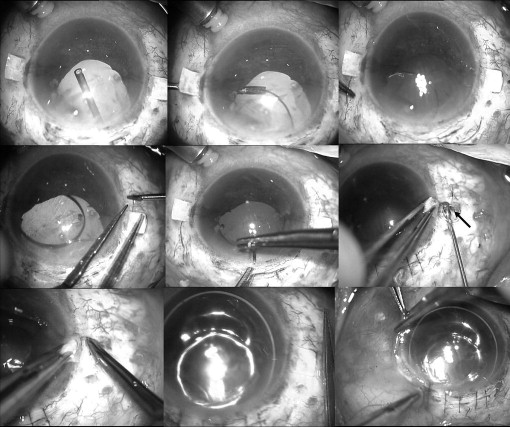
Glued trans-scleral intraocular lens fixation
The PC IOL held in a McPherson forceps was passed through the scleral wound, and the glued IOL forceps (Microsurgical Technology, MST Redmond, Washington, USA or Epsilon Eye Care Pvt. Ltd., Mumbai, India) was passed through the sclerotomy. The tip of the leading haptic was grasped and externalized by the glued IOL forceps. The haptic was held by an assistant. The second haptic then was flexed into the AC and pulled through the opposite sclerotomy by the glued IOL forceps using the handshake technique ( Figure 2 ). When both haptics were externalized under the flaps, they were tucked into the limbus parallel to the intralamellar scleral tunnels made with a 26-gauge needle at the point of haptics externalization. Reconstituted fibrin glue (Tisseel [fibrin sealant], Baxter Healthcare Corporation, Westlake Village, California, USA) then was injected under the scleral flaps and local pressure was applied for 10 seconds. The scleral wound was closed with 10-0 monofilament nylon suture. The conjunctiva was closed with fibrin glue in all eyes ( Figure 2 ). After surgery, topical 0.3% ofloxacin and 1% prednisolone acetate eye drops were prescribed 4 times daily for 4 weeks.
Statistical Analysis
Data were entered in a Microsoft Excel Sheet (Microsoft Corp, Redmond, Washington, USA) and were analyzed using SPSS software version 16.1 (SPSS, Inc, Chicago, Illinois, USA). Continuous variables were expressed as means ± standard deviations, and categorical variables were expressed as individual counts. The Snellen CDVA was converted into logarithm of the minimal angle of resolution units for analysis. Visual acuity of hand movements and light perception were assigned arbitrarily a logarithm of the minimal angle of resolution unit. The statistical tests used were the Wilcoxon signed-rank test, the Mann–Whitney U test, and the Kruskal-Wallis test. Spearman correlation was used for correlation evaluation. Differences were considered statistically significant when the P value was less than .05.
Results
A total of 38 eyes of 38 patients fulfilled our inclusion and exclusion criteria. There were 20 men and 18 women in the study group, with mean age of 59.1 ± 13.3 years (range, 32 to 89 years). The mean follow-up period was 24.1 ± 15.4 months (range, 8 to 60 months) from the date of IOL exchange surgery. The indications for IOL exchange were corneal decompensation in 15 patients (39.4%), malpositioned AC IOL in 11 patients (28.9%), chronic uveitis in 6 patients (15.7%), glaucoma in 5 patients (13.1%), and a broken haptic in the AC in 1 patient (2.6%). Of the 11 malpositioned AC IOLs, 1 (9%) eye had haptic–endothelial touch and 2 (18.1%) eyes had subconjunctival extruding haptics. More than 2 indications were present in 3 (7.9%) eyes and 2 indications were present in 14 (36.8%) eyes ( Table 1 ). All 38 eyes underwent AC IOL removal followed by glued PC IOL (AR40e [Sensar]; Abbott Medical Optics Pvt Ltd, Santa Ana, California, USA) implantation. Of the 38 AC IOLs explanted, there were 36 (94.7%) open-loop flexible IOLs and 2 (5.2%) closed-loop IOLs. There was intraoperative hyphema in 1 (2.6%) eye and uveal incarceration in 1 (2.6%) eye, and a haptic was broken while explantation in 2 (5.2%) eyes. There was no clinical IOL decentration in the immediate or late postoperative period after IOL exchange. Reoperation was not required in any of the patients in the study group.
| Status | No. (%) |
|---|---|
| One complication (n = 21) | |
| Corneal decompensation | 13 (34.2) |
| Luxated IOL | 3 (7.8) |
| Uveitis | 3 (7.8) |
| Glaucoma | 2 (5.2) |
| Two complications (n = 14) | |
| Luxated IOL and glaucoma | 2 (5.2%) |
| Luxated IOL and corneal edema | 6 (15.7) |
| Luxated IOL and uveitis | 1 (2.6) |
| Uveitis and CME | 1 (2.6) |
| Uveitis and corneal edema | 2 (5.2) |
| Glaucoma and corneal decompensation | 1 (2.6) |
| Glaucoma and uveitis | 1 (2.6) |
| More than 2 complications (n = 3) | |
| Luxated IOL, pigment dispersion, uveitis | 1 (2.6) |
| Broken haptic, uveitis, CME | 1 (2.6) |
| Luxated IOL, glaucoma, CME, corneal decompensation | 1 (2.6) |
Visual Acuity
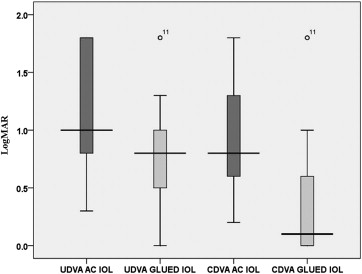
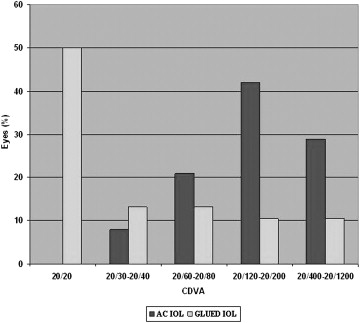
The mean preoperative UDVA and postoperative UDVA were 1.2 ± 0.5 and 0.8 ± 0.4, respectively. There was a significant difference in the preoperative and postoperative UDVA ( P = .000, Wilcoxon test) and CDVA ( P = .000, Wilcoxon test; Figure 3 ). Overall postoperative visual outcomes are presented in Figure 3 . After surgery, 20/20 CDVA was obtained in 19 (50%) eyes ( Figure 4 ). The improvement in CDVA was the result of the resolution of the corneal edema. CDVA better than 20/60 was possible in 25 (65.7%) eyes after IOL exchange. Thirty-four (89.4%) of 38 eyes showed improvement in CDVA. There was no change in the CDVA before and after surgery in 4 eyes. The cause for nonimprovement of CDVA was corneal edema (n = 3) and glaucomatous optic disc neuropathy (n = 1). There was no loss of CDVA in any of the eyes. Fourteen (36.8%) of 38 eyes had an ocular comorbidity that limited visual outcomes like resolving cystoid macular edema, epiretinal membrane, macular degeneration, and corneal opacity, and their potential visual acuity was worse than 20/20. Severe loss of vision was not seen in any eyes.
Cornea and Endothelium
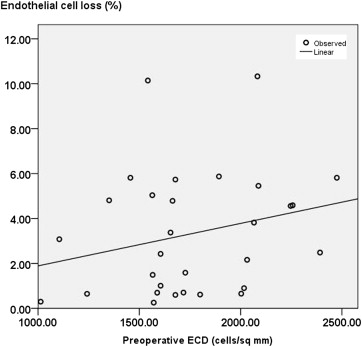
Corneal edema was noted in 20 eyes before surgery. All 20 eyes had CCT of 700 μm or more. There was a significant decrease ( P = .000, Wilcoxon test) in the CCT after AC IOL removal ( Table 2 ). There was significant change in the CCT from before surgery to the last follow-up ( P = .000, Kruskal-Wallis test). However, persistent corneal edema was seen in 3 eyes (mean CCT, 670 ± 10.7 μm) after IOL exchange. Two of 3 eyes with persistent corneal decompensation required optical penetrating keratoplasty. There was a significant difference ( P = .000, Wilcoxon test) between the ECD (in cells per square millimeter; Table 2 ) before and after IOL exchange. There were 5 eyes with preoperative ECD of less than 1500 cells/mm 2 (range, 1013 to 1456 cells/mm 2 ). All eyes with low ECD showed good visual outcome (mean CDVA, 0.58 ± 0.2). There was no loss of CDVA in any of the eyes with preoperative low ECD. Overall, there was no correlation between the ECD before IOL exchange and the postoperative endothelial cell loss at the last follow-up ( Figure 5 ). There was also no correlation between the preoperative ECD and postoperative CDVA ( P = .942) and corneal thickness ( P = .538). There was significant correlation between the CDVA and the CCT ( Figure 6 ).



