Glaucoma Surgery
George Spaeth
L. Jay Katz
Jonathan Myers
Lorenzo J. Pecora
Ronald L. Fellman
There are three different aspects of glaucoma surgery. All of them are absolutely essential for the surgery to be successful. The first aspect deals with the evaluating the person, deciding whether or not surgery is necessary, and selecting the appropriate procedure. The second is the technique used intraoperatively. The third deals with the postoperative care. This chapter, then, has as its logical organization (1) a discussion of the indications for surgery, (2) detailed descriptions of surgical techniques, and (3) evaluation and treatment of complications.
If surgery for glaucoma was 100% safe, it would be used in every patient. It was not infrequent many years ago to proceed with surgery as soon as a definite diagnosis of glaucoma had been made, because medicinal treatments for glaucoma were not readily available and not particularly effective. Surgery was also used often because the diagnosis of glaucoma was largely limited to those with advanced disease, the type of glaucoma that often results in blindness without a highly effective intervention. However, every glaucoma procedure has complications. These complications range from troublesome to fatal not just to the eye but to the patient. Consequently, surgery for glaucoma should only be undertaken with proper attention to the risk/benefit ratio. These are given for four different procedures in Table 1.
Table 1. Risks and Benefits in Glaucoma Surgery | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
SURGERY INDICATIONS AND PROCEDURE SELECTION
EVALUATION OF THE PERSON
Preoperative assessment of the patient with glaucoma is a time-consuming but important process (Table 2). The surgeon must be prepared to deal with the frequent intraoperative and postoperative complications that are a routine part of performing surgery in patients with glaucoma. Among the most important aspects of the preoperative preparation is a discussion with the patient that leads to a realistic understanding of the advantages and disadvantages of surgery and of the anticipated benefits and problems.
Table 2. Glaucoma Surgery | |
|---|---|
|
The time spent in the operating room is a small fraction of the time spent in performing glaucoma surgery. For every hour the surgeon spends operating, 10 or 20 hours are spent in preoperative preparation and postoperative management. Even if surgical technique is perfect, if the preoperative preparation is inadequate or the postoperative management is inappropriate, the result often is unsatisfactory.
This chapter emphasizes the importance of preoperative evaluation and postoperative management. This emphasis does not suggest that technique is not important. Every aspect of the 3- to 6-month period of the surgical episode, which includes time in the operating room, constitutes an essential building block in successful glaucoma surgery.1 This point is often overlooked by those not deeply experienced in the surgical care of patients with glaucoma.
INDICATIONS FOR SURGERY
Glaucoma comes in many forms and varieties. In this chapter, glaucoma means a condition in which the ocular tissues of the eye become damaged at least partially because of intraocular pressure (IOP) higher than the eye can tolerate. An essential part of this definition, then, is that the IOP plays a role in the development of the damage. The reader should note, however, that the word elevated is not included in this definition. The IOP of the healthy eye is actually high, that is, sufficiently elevated so that it has the potential for causing damage to tissues. Indeed, rather than wonder at the occurrence of glaucoma in people with average levels of IOP, it is more appropriate to wonder why it is that everybody does not develop glaucomatous optic nerve damage at a usual level of IOP. The structure of the eye is truly remarkable to allow it to carry a pressure of 15 mm Hg and have tender, minuscule fibers passing through flexible channels in the lamina cribrosa without those neurons becoming damaged routinely. Thus, glaucoma is ocular tissue damage at least partially resulting from IOP. In the acute glaucomas this damage occurs to the cornea, iris, and lens and, given a sufficient duration of time, to the retinal ganglion cells as well. In the chronic forms of glaucoma, damage usually is within the optic nerve itself, with secondary damage to the retinal ganglion cells or, in some cases, primarily in the ganglion cells themselves. The trabecular meshwork also becomes unhealthy by mechanisms that are at least partially unrelated to those responsible for the optic nerve damage.
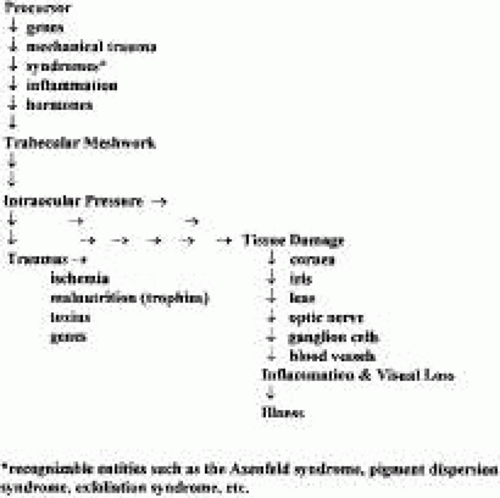
A schema of the pathogenesis of tissue damage in the glaucomas is shown in Figure 1. The pathway to tissue damage involves direct and indirect damage from the pressure within the eye, combined with a variety of traumas that directly or indirectly damage the tissues. The IOP is largely a result of how the trabecular meshwork is functioning. Reduction of IOP is at the heart of preventing damage or allowing damaged tissues to repair themselves. The modulation of other factors, such as vasospasm, anemia, excessively low blood pressure, and neurotoxins, is appropriate, but, to date, little evidence demonstrates actual benefit from such methods of treatment. The tissues of the eye can also be damaged by the treatments themselves.
Patients with glaucoma lose vision or have a diminishment of their quality of life for at least four different reasons. First, patients can lose vision or develop pain because of the damage to the tissues that the glaucomatous process causes. Second, patients can be less healthy as a consequence of the treatments applied to prevent the glaucomatous process from causing damage. The importance of this in the well-being of patients is too often forgotten or ignored. One of the most critical aspects of caring for patients with glaucoma is to “fit the punishment to the crime,” so that the treatment does not cause more disability than the disease itself. This is discussed in detail later.
Third, patients with glaucoma may become impaired because of the mandatory death of a certain number of retinal ganglion cells each year. The rate of this mandatory cell death has been suggested to be around 3000 to 5000 cells per year. This level may not be of consequence in a person with a full complement of around 1,250,000 ganglion cells. However, in a patient with advanced glaucomatous nerve damage, who may have only 50,000 ganglion cells left, a loss of 5000 cells per year would be associated with noticeable and devastating visual deterioration. However, it would be prudent to add a caveat: It is probably unwise to base therapy on the assumption that a patient with glaucoma is going to get worse because of mandatory loss of cells related to aging. First, it is not known whether this principle applies to all cells. It is possible that a proportion of retinal ganglion cells are preprogrammed to live longer than others; it is not rare to see patients with far-advanced glaucoma with a residual central island field (less than 5 degrees) to maintain that island of vision for 10 of 15 years. This seems contrary to the theory that, when 50,000 or so ganglion cells remain, eventual blindness is a certainty. Furthermore, because lowering IOP presumably will not affect the rate of preprogrammed cell death, it is not logical to perform surgery to try to prevent this cell death.
Fourth, patients with glaucoma can lose vision or develop pain because they have an illness in addition to the glaucoma: a pituitary tumor, juxtapapillary chorioretinitis, macular degeneration, and so forth. Visual field loss mimicking glaucoma can be caused by a prominent nose, myopic peripapillary changes, congenital pits of the optic disc, anterior ischemic optic neuropathy, a hypotensive episode, optic neuritis, compressed lesions of the optic nerve or chiasm, occipital lobe lesion, and juxtapapillary choroiditis. Cupping and pallor of the optic nerve that mimics glaucoma can be a normal variation or a consequence of anterior ischemic optic neuropathy, myopic changes, a congenital coloboma, a compressed lesion of the optic nerve or chiasm, syphilis, or optic atrophy-associated choroidal or retinal disease. In these situations, however, the degree of pallor usually far exceeds the amount of neuroretinal rim loss. Thus, wherever pallor is greater than the amount of expected tissue loss, the probability of a nonglaucomatous cause for visual loss must be considered. An exception is the optic atrophy associated with giant cell arteritis (temporal arteritis). This condition often causes severe cupping that is difficult to distinguish from that caused by glaucoma.
When a patient with glaucoma continues to get worse despite IOPs that appear to be in a satisfactory range, and especially if the optic nerve does not show continued glaucomatous nerve damage, the possibility of some other cause for the deterioration must always be considered and appropriate studies undertaken.
The only proven method of preventing or reversing glaucomatous damage is to lower IOP. It has been generally assumed that it is the absolute lowering of IOP that is beneficial. However, stabilizing IOP may also be important.2 Some surgical procedures, such as a guarded filtration procedure, not only can lower IOP but also can decrease the variability of IOP. This stability may be as important or even more important than pressure-lowering in controlling the glaucomatous process. Further study is needed.
Some suggest that other methods may prove to be neuroprotective, and some physicians already employ agents such as a gingko biloba to try to help the nerves resist the damaging effects of IOP. In addition, studies of a variety of potentially neuroprotective agents are under way, but these studies are preliminary. In contrast, there is no question that lowering IOP can be of benefit to many patients with glaucoma. The question is not whether lowering IOP helps but rather whom will it help and how much does the IOP need to be lowered to benefit.
Before any treatment can be initiated in a patient with glaucoma, there must be an evaluation of the patient’s likelihood to suffer from consequences of glaucoma. In patients with narrow anterior chamber angles, this is fairly easy to determine. In patients with a chronic glaucoma, this determination is far more difficult. However, it still must be done.
Glaucoma surgery rarely is restorative or curative. Usually, it substitutes one problem for another. The new problem is intended to be of lesser significance than the old. With the possible exception of iridectomy, glaucoma surgery leaves the patient damaged. Nevertheless, advising surgery is often an appropriate act for the surgeon and choosing surgery often is a wise decision for the patient. With proper technique and patient selection, the problems caused by glaucoma surgery can be minimized and usually can be kept to an acceptable level. Some ophthalmologists now are more aggressive about advising surgery than their colleagues were recently; a few surgeons are even returning to the opinion, held by many in the more distant past, that the proper time to perform surgery is when the diagnosis of glaucoma is made.3,4,5,6,7,8,9,10,11 We believe that the indications and techniques of surgery should be individualized, and in this chapter we include proposals from many schools of thought.1
Surgeons and patients must remember that, although changes in surgical techniques may not be as frequent or as rapid as changes in the clothing fashions, there also are fashions in surgery. Especially in a consumer-oriented society that stresses novelty and rewards the new rather than the proven, surgeons must consider the long-term value of the procedures that they are contemplating and place them in historical perspective. Most patients with glaucoma are affected for the remainder of their lives. Thus, “quick-fix” procedures are of little help. If the glaucoma is a chronic condition (the usual situation), a procedure is of value only if it is either long lasting or safe and easily repeated. Surgeons and patients who are considering which operation to choose should evaluate with skepticism those procedures that have not been subjected to critical long-term study (in this context, long term means years).
Finally, as with the intensity and time expended with the surgical episode, there is a common misunderstanding about the goal of glaucoma treatment among physicians (including most ophthalmologists), patients, and third-party payers and administrators: Because glaucoma is a disease of pressure, the ultimate goal of glaucoma surgery is to lower pressure.
The source of this misunderstanding is not mysterious; the teaching for the past 100 years has been that glaucoma is caused by elevated IOP. However, glaucoma is not a disease of elevated IOP. Only during the last 15 years have the concerns of some prescient ophthalmologists been confirmed (Glaukom ohne Hochdruck und Hochdruck ohne Glaukom—glaucoma without high pressure and high pressure without glaucoma).12,13,14
Approximately 95% of patients with elevated IOP (greater than 21 mm Hg) and perhaps 90% of patients with more significantly elevated IOP (greater than 24 mm Hg) will never get glaucoma.15,16 In addition, one third to one half of patients with glaucoma do not appear to have elevated IOP.14,17,18,19
Indications for Surgery Designed to Prevent an Attack of Primary Angle-Closure Glaucoma
In the past, surgery for narrow anterior chamber angles involved opening the eye and performing a peripheral iridectomy. In recent years it has been possible to make a functional hole through the iris with a neodymium: yttrium-aluminum-garnet (Nd:YAG) laser. This has changed the indications for surgery in patients with narrow anterior chamber angles. It has never been possible to predict with 100% accuracy who is going to proceed to develop an attack of angle-closure glaucoma. Even patients with peripheral anterior synechiae, in whom there is a proof that the angle has closed in the past, do not always proceed to full-fledged angle-closure attacks. Nevertheless, the safety of Nd:YAG laser iridotomy, balanced against the serious damage caused by an attack of primary angle-closure glaucoma, makes the therapeutic decision easy. Specifically, a Nd:YAG peripheral iridotomy should be performed in any patient in whom the ophthalmologist considers that the anterior chamber angle is capable of occlusion and in whom there are not compelling features suggesting that performing an iridotomy is inappropriate. Characteristics of an occludable anterior chamber angle are shown in Table 3.
Table 3. Characteristics of an Occludable Anterior Chamber Angle | |
|---|---|
|
Patients who are hyperopic, are older adults, and have developed cataracts are especially likely to have occludable angles. Mechanisms and predispositions to angle-closure glaucoma are listed in Table 4. Not all types of angle-closure respond favorably to iridectomy or iridotomy. Only those with a pupillary-block type of mechanism will be helped (all those glaucomas secondary to the mechanisms listed in Table 4. A and B, with the exception of 3 C, and in some cases 3 E). Aspects of the history, such as seeing halos around lights in the dark, are suggestive of attacks of angle-closure (Table 5). Signs of the angle-closure glaucomas are listed in Table 6.
Table 4. The Angle-Closure Glaucomas | |
|---|---|
|
Table 5. Symptoms of Primary Angle-Closure Glaucoma* | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||
Table 6. Signs of Primary Angle-Closure Glaucoma* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
A diagnosis of an occludable anterior chamber angle demands gonioscopy. Space does not permit an extensive discussion of gonioscopy here. However, the omission of this from the chapter should not be taken as a sign that the gonioscopic technique is not essential to the diagnosis of angle-closure glaucoma, as well as to the care of the glaucoma patient, including postoperative care. Historical information and slit-lamp and biomicroscopic examination are not adequate to determine whether or not the patient has an occludable anterior chamber angle, except in those rare circumstances in which adhesions extend anterior to Schwalbe’s line. In addition, there are a variety of methods by which the angle closes, and by no means are all of them appropriate to treat with an iridectomy or iridotomy. The secondary angle-closures listed in Table 4 (categories C to G) are more likely to be hurt than helped by such surgery. Thus, gonioscopy is mandatory.
Complications associated with laser iridotomy are shown in Table 7. The pressure spike that may occur following the surgery can almost always to be prevented by pretreatment with pilocarpine and an alpha agonist such as brimonidine or apraclonidine. The ghost image is not always preventable. Patients should always be cautioned that they may see a crescent of light or a ghost image in the inferior visual field following an iridotomy. It should be explained to them that this occurs because light comes through the new hole, that it is harmless, and that it will not in any way interfere with their vision or their function. Bleeding at the time of the iridotomy can be prevented in most patients by careful attention to surgical technique. Patients taking aspirin or an anticoagulant should in most cases have their iridotomy performed with an argon laser or have their iris cauterized with an argon laser before completing the iridotomy with an Nd:YAG laser.
Table 7. Postoperative Problems Associated with Iridotomy | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Indications for Surgery in Patients with the Chronic Types of Glaucoma
The purpose of all types of treatment for all conditions that can impair a person’s health, or have made a person sick, is the same. The purpose is torestore or enhance the person’s health, the person’s sense of wholeness. Glaucoma impairs health by causing pain or loss of vision. It also impairs health as a result of the treatment given to prevent or relieve the pain and loss of vision. These principles also apply to surgical treatment for glaucoma. The amount of lowering of IOP is often thought of as a measurement of the success of glaucoma surgery. This is certainly understandable, because it is the lowering of IOP that is probably largely responsible for the beneficial effect of glaucoma surgery. Consequently, considering how much IOP lowering one is likely to get from a particular type of procedure is appropriate. We will, then, consider this aspect of glaucoma surgery, which relates primarily to the treatment of the chronic forms of glaucoma, but lowering IOP has its down side as well. The proper approach to a patient with glaucoma is not to try to lower the IOP as much as possible. Every millimeter of IOP lowered costs the patient something in side effects and complications, both short and long term. These are considered in detail in the final section of this chapter. However, the avoidance of complications is so central to the entire thinking about the management of patients with glaucoma that their existence must be raised here and must constantly be kept in mind by the surgeon planning the proper approach to a patient with glaucoma.
With the acute types of glaucoma the need for treatment is usually obvious. However, in the chronic forms, damage occurs so slowly, often over a period of 5 to 10 or more years, that the patient and the physician alike may underestimate the damage that is occurring or that will occur in the future. The principle mentioned before applies especially to the chronic glaucomas. Specifically, the physician caring for a patient with glaucoma must make a serious, educated, and effective effort to determine the type of effect that the affected person’s glaucoma will have on that person. Simply having glaucoma is not a justification for treatment. Simply having severe glaucoma is not a justification for treatment. Simply having progressive glaucoma is not a justification for treatment. Treatment is only appropriate if the glaucoma is likely to cause an impairment in the person’s health. Consequently, this determination is absolutely central to the planning of the management of a patient with glaucoma. The initial part of the discussion regarding the indications for glaucoma will, then, deal with a practical way of estimating the likelihood that the person with glaucoma will be harmed by that glaucoma if no intervention is taken.
Every intervention introduces some harm and is obviously also intended to produce some benefit. The challenge in managing patients with glaucoma is to balance the presumed benefit of treatment against the risks of the treatment and the risks attendant to no treatment.
DISCERNING THE RISK OF NO TREATMENT
The two most helpful indicators of the likelihood that a person will be damaged in the future or will continue to be damaged from chronic glaucoma are (1) the nature of the optic disc and (2) the presence of characteristics known to be associated with deterioration of visual function in patients with glaucoma. The chronic glaucomas cause their damage by damaging the optic nerve. Attention to the optic nerve, then, is primary. In addition, the presence of optic nerve damage demonstrates that the patient in question has a condition that has caused and will probably continue to cause further damage. Because most chronic glaucomas are not spontaneously cured, evidence that the person has already developed damage is convincing documentation that the patient will develop more damage.
Signs of optic nerve damage caused by glaucoma are shown in Table 8. The reader should note that a large cup/disc ratio is not listed as a definite sign of glaucoma.
Table 8. Signs of Optic Nerve Damage due to Glaucoma* | ||
|---|---|---|
|
RIM NOT CUP
Measuring the cup/disc ratio tells the examiner little about the actual health of the optic nerve. In contrast, concentrating on the area where the rim is thinnest is a more reliable way of determining whether the optic nerve has been damaged. The examiner should concentrate on the inferotemporal and the supertemporal portions of the optic nerve, because those are where damage is most likely to occur.
SPECIFIC FINDINGS
Specifically, one should look for an acquired pit of the optic nerve, or a notch, or the presence of an irregular hemorrhage crossing the outer edge of the rim. The width of the rim should be measured in rim/disc ratio, where 0.5 indicates the thickest rim possible, and 0.0 means no rim is present.
MEASURING THE SIZE OF THE DISC
The size of the optic disc should be evaluated. A relatively simple method of doing this is to use a 66-diopter (D) lens to visualize the optic disc. The height of the optic nerve head is determined by decreasing the height of the biomicroscopic slit beam projected on the nerve head; when the top and bottom of the beam coincide exactly with the top and bottom of the optic disc, the height of the optic nerve is read directly from the reticule on the Haag-Streit slit lamp. The correction factors for other lenses are shown in Table 9.
Table 9. Lenses and Correction Factors Used in Measuring the Size of the Optic Disc | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||
EARLY VERSUS LATE CHANGES
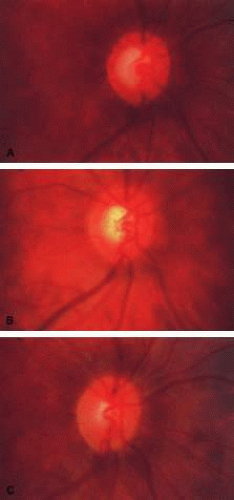
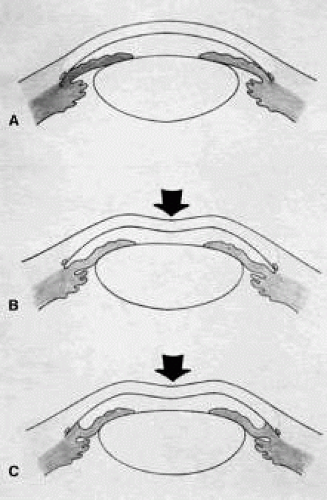
In the later stages of the disease, one can be fairly sure about whether the optic nerve is glaucomatous or not. By later stages of the disease is meant a situation in which there is absence of rim tissue in one area of the optic nerve, especially supertemporally or inferotemporally. However, in the early stages of glaucoma it is not possible on the basis of a single examination to determine whether the optic nerve is damaged. For example, a patient with a central cup and a rim/disc ratio of approximately 0.3 in all areas could have an optic nerve that has improved, remained the same, or gotten worse (Fig. 2). Consequently, in the early stages of the disease it is impossible to determine whether an optic nerve head is healthy or diseased on the basis of a single ophthalmoscopic or image analysis examination. On the other hand, it is possible with a high degree of reliability to look at such an optic nerve and determine whether or not a visual field defect is present. Normal-sized discs (around 1.75 mm) with rims thicker than 0.1 rim/disc ratios rarely have visual field loss. Small discs, that is, discs that are smaller than 1.5 mm in diameter, will rarely have visual field defects when the rim/disc ratio is greater than 0.2. Obviously, if no visual field defect is present, it is unlikely that the patient will have any disability as a result of any change that has already occurred in the optic nerve. Thus, even though it is not possible to look at an optic nerve without an acquired pit of the optic nerve, a notch, or complete loss of the neuroretinal rim all the way to the outer edge of the rim and determine with certainty whether or not the disc is damaged, it is possible to determine whether such a disc will be associated with any functional loss of vision.
DISC CHANGES USUALLY PRECEDE FIELD CHANGES
The next critical piece of information in deciding on whether treatment is necessary is to recall that the early visual field defects that occur in glaucoma are asymptomatic. Patients can develop an extensive loss of nasal field in one eye and yet be able to function entirely normally. That fact is often mentioned as one of the characteristics that makes glaucoma especially dangerous. Indeed, the lack of symptomatology in the early stages of chronic glaucomas is the reason why patients often have advanced damage when first seen. However, the lack of symptomatology in early glaucoma also means that it is not essential to prevent a patient with early glaucoma from developing further, early disc damage or even from developing visual field loss. One should remember that the goal of therapy is the health of the patient, that all treatments have some side effect, and that slight deteriorations of the optic nerve and visual field in the early stages of glaucoma do not cause any symptomatology. Thus, it becomes difficult to justify surgery in the early stages of glaucoma until enough deterioration has occurred so that loss of visual function as a result of glaucoma is relatively certain if the deterioration continues.
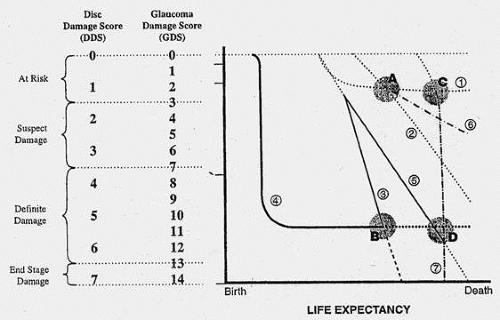
It is helpful to plot the patient’s changes in the stages of glaucoma on a graph (Fig. 3).
For rational treatment of glaucoma, the patient’s life expectancy must be considered. Surgery is not justified on a patient with early, or even moderately advanced glaucoma who is getting worse slowly, whose life expectancy is 6 to 12 months. However, there is little justification for not doing a glaucoma procedure on a patient with the same amount of damage, getting worse at the same rate, who has a life expectancy of 40 years. Although it is not possible to determine life expectancy with complete precision, it is possible to get a good idea. Factors that enter into that determination are shown in Table 10.
Table 10. Factors Permitting a Meaningful Estimation of Life Expectancy | |
|---|---|
|
The first indication for surgery, then, is documented deterioration of the optic disc or visual field resulting from glaucoma occurring at a rate that will cause a decrease in the patient’s functional ability before the time of his or her death despite maximum tolerated medicinal therapy.
Maximum tolerated medicinal therapy means the greatest amount of medicinal treatment that is well tolerated by the patient. In some people this may mean no medicinal therapy whatsoever. For example, in a patient who finds it difficult or impossible to use medicines and in whom an argon laser trabeculoplasty has not worked, surgery is appropriate, if it is clear that the person will suffer visual functional loss if an intervention is not successful. In contrast, in an extremely ill patient who is tolerating glaucoma medications well, the conjoint use of three different classes of eye drops together with an oral carbonic anhydrase inhibitor may constitute maximum tolerated medicinal therapy. Surgery may be the most appropriate first treatment in some people, whereas it may be appropriate in others only after extensive trials of medication have failed. The most important word in this qualifying phrase is tolerated.
VISUAL FIELDS MOST HELPFUL IN GLAUCOMA IN ITS MODERATE STAGES
Once the optic nerve has developed significant damage it may be difficult or even impossible to observe further change in the optic nerve despite a deterioration in the patient’s functional ability. Consequently, in the later stages of the disease visual function is the primary indicator for the adequacy of care. Thus, increasing symptomatology (i.e., deterioration of function) is the major indication for an increase in the vigor of therapy (Table 14). One way to demonstrate such deterioration of function is serial visual field examinations. These examinations must be done properly to make sure that the fields are accurate representations of what is happening to the patient. Automated perimetric machines can be helpful, but they must be used properly by an appropriately trained technician who is doing his or her job well, and the results of the visual field examinations must be interpreted thoughtfully and knowledgeably. Just because a visual field printout shows that the field has gotten worse does not mean that the patient is getting worse. Just because a visual field is the same does not mean that the patient is not getting worse. Deterioration of the field may be caused by improper refraction, fatigue, improper positioning in the field testing instrument, progressive cataract, decreasing pupil size, and so forth. The field can be deteriorating despite an absence of change on the perimetric result whenever the previous field contains areas registered as 0 unless a larger test object was used at the time of the subsequent examination. A 0 does not indicate that the patient is absolutely blind in a particular area but only that the patient failed to see a particular test object at a particular brightness in that area. 0s do not necessarily indicate absolute visual field defects. A larger or brighter text object perhaps could have been seen. Careful testing of the field and thoughtful, knowledgeable interpretation of the results are helpful in the moderate stages of glaucoma damage. This is especially true because substantial change can occur and yet not be noted by the patient when visual field defects are still in their early to moderate stages.
Table 14. Anticipated Effect of Different Levels of Vigor of Therapy on Intraocular Pressure | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||
HISTORY MOST IMPORTANT IN LATE STAGES OF GLAUCOMA
Once visual field loss has become marked, or is close to fixation, the patient’s own sense of how well he or she is seeing and functioning is often the most sensitive indicator of whether the patient is remaining stable or getting worse (or improving). Thus, meticulous questioning of the patient regarding these issues is essential. It is helpful to ask the patient specific questions such as, “Is there anything that you can’t do now that you were able to do in the past?” If the answer is a “yes,” then the timing and severity need to be further defined by asking questions such as, “What can you not do now that you could do a month ago?” “What can you not do now that you could do a year ago?” “What can you not do now that you could do five years ago?” These questions are designed to provide the physician with a specific indication of how rapidly and how seriously the patient’s visual function has deteriorated. As with deterioration of the optic nerve, it is not the deterioration that triggers the surgery. Surgery is appropriate when the deterioration is occurring at a rate that will interfere with a patient’s visual function. Thus, use of the glaucoma graph (see Fig. 3) is helpful. It is essential to know the patient’s life expectancy and the rate at which the glaucoma is worsening, as well as the stage of the glaucoma, to reach a rational decision regarding the appropriateness of treatment.
A second indication for surgery in a patient with glaucoma is a deterioration of visual function resulting from glaucoma despite maximal medical tolerated therapy occurring at a rate that will interfere with the patient’s health.
RISK FACTORS FOR PROGRESSIVE GLAUCOMA DAMAGE
Probably the most significant factor that will determine whether a person will develop visual loss from glaucoma is the genetic make-up of the person. Presently, however, it is not possible to predict with accuracy whether a person will develop functional loss from glaucoma on the basis of genetic analyses that are available. It is known that certain types of glaucoma are familial, and genetic abnormalities in some of these types of glaucoma have been identified. However, even in those conditions it has not yet been determined that all people possessing the genetic abnormality will develop functional loss from glaucoma. Possession of such a gene, however, certainly puts the person at risk and should initiate a program of meticulous observation. Patients with a family history of visual loss from glaucoma also are at risk and should be meticulously observed. It should be noted that this is not a family history of glaucoma but rather a family history of visual loss from glaucoma. This history increases the likelihood (perhaps ten-fold greater than the risk for those who do not have such a history) that the affected person will develop visual loss from glaucoma.
The level of IOP provides useful information regarding the likelihood that a patient will eventually develop damage. Pressures below 40 mm Hg are of no use in determining whether a patient actually has glaucoma. That is, pressures below 40 mm Hg do not indicate whether a patient has developed any damage related to IOP. However, the higher the IOP the greater is the likelihood that the person will eventually develop damage. In addition, the higher the IOP, the more rapidly is the damage likely to develop. The chance that a person with an IOP of 50 mm Hg will develop glaucoma damage within months is so great that initiation of therapy is almost always justified, that is, if the eye has useful vision to preserve! The chance that a person with an IOP above 30 mm Hg will develop damage is far less, and other factors, such as a patient’s longevity, must be considered before deciding on appropriate therapy. IOPs below 30 mm Hg are rarely a justification for initiating surgical treatment. Rather, the cause for initiating treatment is an IOP that has been associated with the development of further damage, whether that IOP is 15 mm Hg or 30 mm Hg. Except at levels of IOP above 30 mm Hg, then, it is not the IOP per se that initiates the surgery, but rather the knowledge that a particular level of IOP has already been documented to cause continuing damage.
Other factors that increase the likelihood that a patient with glaucoma will eventually get worse are race and considerations relating to the nutrition of the optic nerve. Black people are at greater risk for developing visual loss from glaucoma, as are those who have conditions that will lead to decreased perfusion or nutrition of the optic nerve. Such factors, however, are not adequate justification for surgery. They are only factors that increase the likelihood of a person getting worse and, therefore, are factored into the therapeutic decision. A rough plan for using the amount of optic disc damage and the intensity of risk factors as a guide for therapy is shown in Table 11A and B.
Table 11a. Stages of Optic Nerve Damage and Correction Factors for Measuring the Optic Nerve | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 11b. Management by Disc and Risk | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The last risk factor that must be considered when evaluating the indications for surgery is the person’s ability to manage his or her life. Although there are major biologic differences between the various types of glaucoma and how glaucomas manifest in different people, with proper care glaucoma should rarely be a cause for loss of visual function. Although the doctor plays an important role, how the patient acts is even more essential; the patient chooses the doctor, the patient chooses whether or not to tell the doctor certain types of information, the patient decides whether the doctor is doing a good job, and the patient chooses how to use recommended therapy. Evaluation by the physician of the patient’s life management skills is an essential part of the diagnostic evaluation of every patient. Patients may be located in exactly the same position on the glaucoma graph (see Fig. 3), but because of different life management skills, medicinal therapy may be appropriate in one patient and surgical therapy in another.
The third indication for surgery for glaucoma is the presence of a glaucoma that has the capacity to cause deterioration in function occurring in a person who is unable to obtain continuing medical care or is unable to manage his or her life appropriately.
In summary, the indications for surgery in a patient with glaucoma are (1) when glaucoma is responsible for significant pain or for deterioration of the optic nerve or visual function despite maximum tolerated medicinal therapy and when the deterioration is occurring at a rate that will cause a deterioration in the patient’s function; (2) an IOP above 40 mm Hg on repeated determinations despite maximum tolerated medicinal therapy in an eye with useful vision; and (3) the presence of a glaucoma that has the capacity to cause deterioration in function occurring in a person who is unable to obtain continuing medical care or is unable to manage his or her life appropriately.
SELECTION OF THE APPROPRIATE SURGICAL PROCEDURE
Because the goal of treatment for glaucoma is maintenance or enhancement of health, the most appropriate method is the one that will lower the IOP the desired amount for the desired length of time with the least likelihood of causing side effects. One of the first things the physician must do when deciding on the most appropriate therapy is to select a final target pressure range.20,21,22,23 Although only an approximation, this target pressure range is helpful in planning. It is based primarily on the level of IOP known to have caused damage (Table 12), but the nature of the optic nerve may be a consideration. Some surgeons believe that the more damage the optic nerve has sustained before surgery, the more markedly IOP must be lowered. This approach sounds logical, but no studies support or deny the theory. Some authors also believe that the appearance of the optic nerve provides a valuable clue about its susceptibility to the damaging effects of IOP. The presence of an acquired pit of the optic nerve (a pseudopit) is a strong indicator of susceptibility.24,25
Table 12. Target Pressure Ranges: Goals for Lowering Intraocular Pressure | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
The target pressure range is based primarily on the known level of IOP at which damage developed (see Table 12) and to a lesser extent on a variety of other factors, such as the degree and pattern of optic nerve damage and considerations listed in Table 13. Table 14 gives rough guidelines for the amount of IOP lowering that different methods of treatment can accomplish.
Table 13. Glaucoma Damage to the Optic Nerve: Risk Factors | ||
|---|---|---|
|
If the decision has been made to perform surgery, the surgeon must consider how to perform the surgery to achieve the desired target pressure range (see Table 14). Although it is not always possible to achieve the desired target pressure range, certain procedures and approaches to surgery give the surgeon the ability to approximate the goal. A guarded filtration procedure performed essentially as described by Watson, for example, in a white patient without risk factors for failure usually will result in IOP of approximately 17 mm Hg, ranging from 15 to 19 mm Hg.26 In contrast, a full-thickness filtering procedure, such as a corneoscleral trephine, usually will result in a mean IOP of approximately 14 mm Hg, with a range of 12 to 16 mm Hg.26
To obtain increasingly lower IOP, it is necessary to modify surgical technique (Table 15). By performing guarded filtration procedures so that the sutures can be released postoperatively, with the use of either laserable or releasable sutures, a lower final IOP can be obtained in some cases. This technique allows a bleb to develop that is similar to that seen in a full-thickness filtration procedure. Such blebs tend to be thin, polycystic, and located directly at the limbus, in contrast to those seen after classic trabeculectomy, which are thicker, lower, more diffuse, and more posterior. When antifibrosis agents (antimetabolites and corticosteroids) are added to procedures designed to develop full-thickness filtration, IOP tends to be sharply reduced.27,28,29 The blebs associated with the use of 5-fluorouracil (5-FU) and mitomycin are an exaggeration of the full-thickness type of bleb: Often, they are extensive, sometimes involving 360 degrees of the anterior surface of the globe, and the conjunctiva tends to be thin and completely ischemic.30,31,32,33,34
Table 15. Methods Thought to Help Achieve Lower Final Intraocular Pressure After Filtration Procedure | |
|---|---|
|
The thin filtration blebs associated with the use of mitomycin or 5-FU may rupture spontaneously. They tend to be so high that the adjacent cornea becomes dry, with the development of an uncomfortable delle. Ptosis tends to develop, and patients often are photophobia One of the most serious concerns is the high incidence of endophthalmitis in patients with thin blebs. When full-thickness blebs were the routine type of glaucoma procedure, endophthalmitis would develop in approximately 1% of patients. When 5-FU was used to develop filtration blebs inferiorly, an 8% rate of endophthalmitis was reported.35,36,37,38,39,40,41,42 Hypotony, even in the absence of a thin bleb, introduces serious problems. The soft eye does not maintain a constant optical state, and it has a constantly changing amount of astigmatism that makes it impossible to correct. Each time the patient blinks, the amount of astigmatism changes. Macular and disc edema cause reduced central acuity and deterioration of the visual field, and the globe may have a constant, visceral ache. Patients with such eyes are not comfortable.
The likelihood of achieving a surgical success is also considered when weighing the pros and cons of surgery and the specific procedures to be selected (Table 16).
Table 16. Factors Related to Success or Failure of Glaucoma Surgical Procedures in Patients with Open-Angle Glaucoma | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
A variety of factors that relate to each person will affect the likelihood that a guarded filtration procedure will succeed. Those predisposing to success are shown in Table 17 and those tending to cause failure in Table 18. When it appears that the likelihood of success with a guarded filtration procedure is less than the surgeon and patient desire, then either another procedure must be chosen or the guarded filtration procedure must be modified. A tube-shunt is a reasonable option but is far more likely to be associated with troublesome extraocular muscle movement abnormalities leading to disturbing double vision postoperatively. This is, of course, not a limiting factor in a patient in whom vision is essentially limited to one eye. Titrating the final IOP following a tube-shunt procedure is also more difficult than it is with a guarded filtration procedure. Because of the seriousness of the complications associated with their use, cyclodestructive procedures are usually employed only after other surgical methods have failed or in specific situations (see Table 1).
Table 17. Factors Predisposing to a Low Intraocular Pressure as a Result of a Guarded Filtration Procedure | |
|---|---|
|
Table 18. Risk Factors for Failure of a Filtration Procedure | |
|---|---|
|
Because many glaucomas progress with IOPs in a relatively low range (e.g., less than 15 mm Hg), there is increased interest in trying to develop a low final IOP. Because 5-FU and mitomycin C can result in lower final IOPs, they are commonly used with guarded filtration procedures. Some surgeons recommend using antimetabolites routinely in all cases. This is not our recommendation. It is our belief that the use of an antimetabolite must be factored into the decision of what type of surgery should be done so that the surgeon considers all the other factors that make a particular procedure appropriate or inappropriate for a particular patient. In addition, antimetabolites, at least mitomycin C, cannot be titrated, as was hoped, to increase the beneficial effects and decrease the troublesome complications. Table 19 provides a rough set of guidelines for suggested use of antimetabolites in association with guarded filtration procedures based on the desired final level of IOP and the number of risk factors for failure. These are the same risk factors indicated in Table 18.
Table 19. Suggested Use of Antimetabolites: Rough Guidelines | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
The underlying diagnosis is another factor to consider in selecting a procedure. This is often the most important determining consideration. Rough guidelines are provided in Table 20.
Table 20. Advisability of Surgical Treatment in Selected Types of Glaucoma | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The anticipated effect of the surgery on the patient’s quality of life must also be considered (Table 21).
Table 21. Expected Effect of Glaucoma Surgery on the Patient’s Quality of Life | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||
The procedure should also be able to be repeated easily with a reasonable likelihood of success. The likelihood that a glaucoma surgical procedure will be followed by troublesome symptoms will vary, of course, according to the seriousness of the condition, the nature of the patient, and the skill of the surgeon.
Specific Considerations Regarding Relief of Pain and the Treatment of Blind Eyes
Glaucoma can be associated with mild, moderate, or extremely severe discomfort. However, it is not the absolute level of pressure that causes the pain but the change in IOP and the rapidity with which that change occurs. Patients can maintain IOPs of 50 or 60 mm Hg with no discomfort. On the other hand, eyes in which the IOP varies 10 or 15 mm Hg in a day, for example, from 15 to 30 mm Hg, are usually productive of moderate but troublesome pain. A classic symptom of a failed cyclodialysis cleft is excruciating pain when the IPO goes from around 10 to around 40 mm Hg. There is a tendency to consider that the pressure level itself is the cause of the pain and, therefore, to try to relieve pain by lowering IOP. This is rarely successful, however, except in those cases in which the pressure is fluctuating more than 10 mm Hg during the day, such as those in whom the IOP rises more than 20 mm Hg rapidly. Patients with unstable glaucoma, such as occurs in association with the exfoliation syndrome, may have repeated measurements of IOP in a normal or slightly elevated range and yet may be uncomfortable. The probable cause is the variable IOP, ranging from 15 to 35 mm Hg during the day. In such cases surgery may become indicated because of deterioration of the optic nerve despite normal pressure. Patients with blind, painful eyes from IOPs around 40 mm Hg resulting from neovascular glaucoma usually are uncomfortable because of the ischemia or the inflammation and not because of the elevated pressure. The appropriate treatment for these cases is usually topical atropine and corticosteroids and not surgery designed to lower IOP.
Treatment of Blind Eyes
It is almost never appropriate to perform any surgical procedure other than an injection of retrobulbar alcohol or chlorpromazine or an enucleation or evisceration on a blind eye. Although the incidence of sympathetic ophthalmia is low, sympathetic ophthalmia is such a totally devastating complication that its potential occurrence mitigates against performing procedures that have the capacity to cause it. Sympathetic ophthalmia has been reported to occur with surgical peripheral iridectomy, filtration procedures of every kind, cyclocryotherapy, cyclophotocoagulation, and cyclodialysis. Retrobulbar alcohol tends to produce problems of its own, including numbness in the skull, ptosis, and strabismus. The experience with retrobulbar chlorpromazine is less extensive but promising because the pain relief it provides is usually excellent without production of the problems associated with retrobulbar alcohol. An injection of chlorpromazine, 25 mg, is placed in the retrobulbar space.
The most appropriate therapy for the blind, comfortable eye is no therapy. The most appropriate therapy for the eye that is blind and has moderate discomfort because of markedly fluctuating IOP is a long-acting agent such as slow-release timolol; once the pressure is stabilized, the timolol often may be stopped.
When patients learn that they have glaucoma, they may have an immediate decrease in the quality of life because of the anxiety that they experience as a result of knowing that they have a potentially blinding disease. Telling a patient that surgery may become necessary usually increases the degree of fear. Patients with glaucoma are often asymptomatic. The recommendation of surgery to prevent the loss of vision may be difficult for these patients to accept. They must have implicit trust in their surgeon. Their concerns are justified; surgery does not always work. Surgery does not always control the disease, and often it makes the vision worse. Consequently, fully informed consent must be obtained. The surgeon must be certain that the patient understands the risks of having surgery, the risks of not having surgery, the benefits of having surgery, and the benefits of not having surgery. In addition, the patient must be given a realistic idea of what to expect.
SURGICAL TECHNIQUES
Glaucoma surgery is best performed under the operating microscope. In some cases, high magnification is not essential. In others, such as certain portions of guarded filtration procedure, it increases the likelihood of a successful operation. Care should be taken to avoid photic damage to the retina, especially in patients who have far advanced disease. The light from the operating microscope should not focus on the posterior pole. We usually perform most aspects of glaucoma surgery with side illumination rather than the axial illumination from the microscope. When axial illuminator is used, it usually is reduced in intensity, and the cornea is covered.
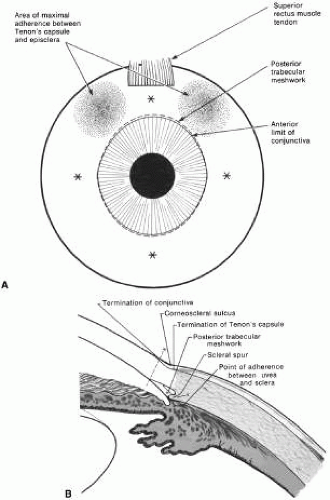
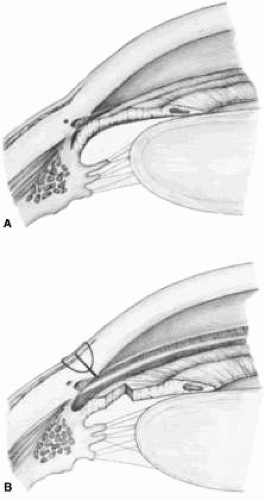
The surgical anatomy of the anterior chamber angle in an average eye is shown in Figure 4A and B. Landmarks will differ markedly in eyes that have abnormalities. In the myope, the space between the anterior limbus and the iris root is greater; in the hyperope, it is less. An incision at the corneoscleral sulcus may enter the anterior chamber far anterior to the trabecular meshwork in the myope but may enter the posterior chamber in the hyperope.
Because glaucoma in infants is rare, its surgical treatment is not discussed in detail. However, guarded filtration procedure is described later and may be employed with satisfactory results in many patients with congenital glaucoma (trabeculodyspenesis).
PREOPERATIVE CARE
The reader should refer to the earlier section of this chapter on evaluation of the person. The goal is to move the patient from conditions auguring failure toward those promising success. Obviously, many factors cannot be changed. In the simplest terms, the healthier the eye and the less likely it is that scarring will develop when the eye is injured, the greater the chance for success. Preoperative care is discussed in greater detail in the sections that describe the operative procedures.
Many people take aspirin products routinely. Aspirin comes in many forms, and it is part of many compounds. Because it predisposes patients to bleeding, it should be discontinued, if prudent, 2 weeks before surgery. Most people who are taking aspirin are taking it for relatively unimportant indications; some simply may have heard that aspirin can help prevent heart attacks. However, in some patients, aspirin may be an important part of treatment, and discontinuation of the aspirin must be coordinated with the physician who ordered the aspirin. Wherever feasible, it should be discontinued preoperatively.
Other agents also predispose patients to bleeding, for example, dipyridamole and anticoagulants, such as dicumarol. These agents should be discontinued an appropriate time before surgery to allow the blood to return to a normal clotting state. Some patients may have to take heparin instead of dicumarol at the time of surgery. This substitution must be individualized and coordinated with the physician managing the patient’s anticoagulants.
PARACENTESIS
A paracentesis should be part of virtually all intraocular glaucoma surgical procedures. This opening in the cornea facilitates management of complications such as bleeding or flat anterior chamber; allows deepening of the anterior chamber at the time of surgery; provides an entry for acetylcholine or sodium hyaluronate; allows the surgeon to determine at the time of surgery how much filtration, if any is occurring through the guarded filtration procedure or sclerostomy; permits safe development of a bleb at the conclusion of surgery; and allows the surgeon to detect whether any tears or leaks are present in the conjunctival flap. The major risk of the procedure is that it may damage the lens. This damage can be avoided by making sure that the instrument used to develop the paracentesis never points toward the lens; holding the instrument parallel to the iris surface eliminates the risk of damaging the lens.
Fixation of the globe is critical. The point of fixation should be directly in line with the intended direction of the paracentesis. If the paracentesis is to be made at the 10 o’clock position, extending exactly inferiorly, the sclera should be held directly superior to the 10 o’clock position (Fig. 5). If the surgeon prefers a horizontal paracentesis, starting at the 3 o’clock position, then the globe must be fixated precisely at the 3 o’clock position. An instrument with fine teeth provides good fixation. Examples are the Bonn or Barraquer-Colibri fine-toothed forceps. The tips should be separated only slightly, approximately 1 mm, and then pressed hard against the sclera. Conjunctiva, Tenon’s capsule, and episclera are not adequate sites for fixation.
We prefer to use a new, sharp, short, disposable 27-gauge needle on a 2- or 5-ml syringe. The needle is held, bevel up, absolutely parallel to the iris surface. In an eye with an iris bombe or a flat anterior chamber, the needle actually may be pointing anteriorly. The tip of the needle is placed against the cornea in the desired position, and the globe is pulled by the fixating hand in the direction opposite to that in which the needle is pointing. For example, if the needle is held horizontally, at the 3 o’clock position of the left eye, then the sclera is grasped at the 3 o’clock position and pulled laterally (temporally) (see Fig. 5).
The needle tip enters the cornea. If there is normal or elevated pressure and the chamber is deep, the needle penetrates the cornea after making a paracentesis that is approximately 1 to 2 mm long. If the eye is soft or the chamber is shallow, the needle remains in the cornea and does not penetrate the anterior chamber. This position is desirable in cases in which the cornea is thin, such as in the buphthalmic eye. If the intracorneal track is longer than 2 mm, then the needle and syringe are depressed back toward the apex of the orbit; they are depressed toward the floor of the operating room (see Fig. 5, top right). The tip of the needle must not be angled toward the iris-lens; it must be kept parallel to the iris. As the needle and syringe are pushed toward the floor, the needle changes the curvature of the cornea, permitting it to enter the anterior chamber when advanced. The needle is advanced by a combination of pulling the globe and pushing the syringe. The importance of firm fixation and of introducing the needle against traction provided by the fixation must be stressed. Also, it is essential that the needle not be angled toward the iris, but kept parallel to the iris surface.
Once the needle enters the anterior chamber, it is more clearly visible than when it is intracorneal (see Fig. 5, bottom right). The tip is advanced carefully about 1 to 3 mm, until the surgeon is sure that the endothelium has been completely penetrated. The needle is then withdrawn.
If the paracentesis has been made with a no. 25 needle, a no. 30 blunt-tipped needle can be introduced later with ease. If the cornea is especially thin or if the surgeon wishes the fit to be especially tight, a 30-gauge needle should be used for the paracentesis.
When re-entering the paracentesis track, the blunt needle must be directed exactly parallel to the original track and must hug the posterior (internal, deep) aspect of the track. Often, the neophyte struggles unnecessarily to get the no. 30 blunt-tipped needle into the anterior chamber through a no. 27-size paracentesis. However, when the blunt-tipped 30-gauge needle is angled in the direction of the floor, that is, toward the iris, and slid along the internal aspect of the paracentesis, it enters gracefully. Alternatively, use a sharp 27-gauge needle for the paracentesis and a sharp no. 30 needle for later entry.
USE OF SLIP KNOTS AND RELEASABLE SUTURES
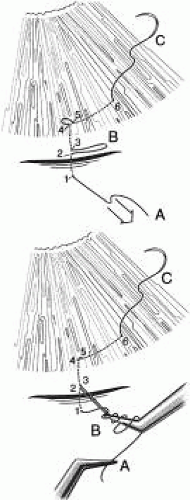
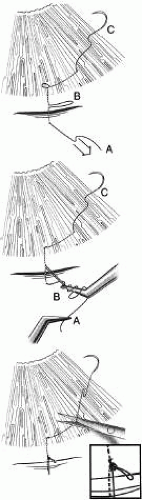
The use of a slip knot is helpful in achieving the desired level of tightness of the scleral flap in a guarded filtration procedure. Slips knots also have the advantage of being easier to bury than the usual surgeon’s knot. They cannot be used to close incisions under tension, but are appropriate for almost all other situations. We use them routinely. Figure 6 illustrates the method of tying slip knots. See p. 24 for a discussion of releasable sutures (Fig. 7).
IRIDECTOMY
Iridectomy deserves separate treatment in this chapter because, with rare exceptions, its basic purpose is different from that of all other glaucoma surgery. Iridectomy often is performed not to lower IOP but to correct an anatomic aberration, narrowness of the anterior chamber angle (Fig. 8). It is important to explain to the patient why the iridectomy is recommended. Patients usually conclude that glaucoma surgery has as its purpose the lowering of IOP, and unless enlightened to the contrary, most patients anticipate that IOP will be lower after iridectomy.
Diagnosis and Classification
An extensive description of the diagnosis and classification of the angle-closure glaucomas cannot be given here.1,43 However, the essential component in the diagnosis of the angle-closure glaucomas, gonioscopy, must be mentioned. Differentiation between optical contact and actual adhesion between the iris and the cornea cannot be made without the use of indentation gonioscopy; therefore, the correct diagnosis of the angle-closure glaucomas demands the appropriate use of a gonioscopic lens that can be used in indentation gonioscopy8,44 (Fig. 9). We prefer the Zeiss four-mirror lens on an Unger handle. For the diagnosis of angle-closure glaucoma to be certain, the ophthalmologist must be certain that the symptoms could only be the result of angle closure and that the anterior chamber angle actually has closed. Thus, the search for peripheral anterior synechiae, characteristically between the 10 and 2 o’clock positions of the eye, assumes great significance.
Peripheral iridectomy is described in moderate detail because it is our opinion that the general ophthalmologist should still be fully competent in performing a standard surgical iridectomy. Nd:YAG laser iridotomy is clearly the procedure of choice in patients needing an iridotomy. However, it is not always possible. In patients in whom the cornea does not become sufficiently clear to allow a laser iridotomy, an incisional surgical iridectomy becomes necessary. In addition, incisional iridectomy may be necessary in some patients with a diagnosis of pupillary block glaucoma in whom a persistent, patent peripheral iridotomy cannot be achieved with laser techniques.
Preoperative Care
Most attacks of acute primary angle-closure glaucoma can be treated medically. The longer the attack, the higher the pressure, the more the eye has been damaged, and the less likely the patient is to be harmed by the medications used to treat the attack, the more vigorous the treatment should be. When the attack is brief, the inflammatory signs minimal, and the pressure relatively low, pilocarpine 1% every 5 minutes for four doses often is adequate. Apraclonidine 1% often is helpful in treating patients with high IOP or in cases in which lowering IOP is not expected to be accomplished with the use of pilocarpine alone. When treatment must be maximal, the following routine may be followed: pilocarpine 1% every 5 minutes for four doses; timolol 0.5% immediately and again in 1 hour; apraclonidine 1% immediately; acetazolamide 500 mg intravenously; and an osmotic agent (isosorbide, 30 mg/kg body weight in routine cases; mannitol 20%, 70 ml/kg body weight intravenously in the nauseated patient who can tolerate a large sudden increase in blood volume; or anhydrase glycerol orally, 1 ml/kg body weight in the patient who can take oral medication but is likely to have urinary retention). Topical steroids are appropriate if the eye is inflamed. Occasionally, forceful compression of the anterior chamber with an instrument such as the Zeiss four-mirror gonioprism can push the angle open and help to end the angle-closure attack. Once the attack has been stopped, it is helpful to allow the eye to quiet before proceeding with the iridectomy. Weak pilocarpine administration, such as 1% two or three times a day and aqueous suppressants, such as timolol and acetazolamide, should be continued until the time of surgery. If surgery is indicated on the fellow eye, it may be appropriate to perform an iridectomy on that eye while waiting for the involved eye to quiet. Pilocarpine should be used with caution in both the involved eye and the fellow eye because it can predispose the eye to angle closure by increasing the degree of papillary block or causing the lens-iris diaphragm to move anteriorly.
Occasionally, it is impossible to break an attack. In such cases, it is advisable to proceed with surgery promptly, despite the presence of high IOP. Here, a retrobulbar injection of anesthetic agent is appropriate and may help lower the IOP. If unsuccessful, the pressure will be brought down easily by paracentesis at the time of surgery.
When a peripheral iridectomy is performed, the pupil should be as small as possible. The use of mild pilocarpine is an easy, effective approach. In cases in which the IOP remains elevated or the sphincter is nonfunctional, however, reducing the size of the pupil may be difficult. Multiple instillations of pilocarpine are not recommended in such cases. Usually, they only make the patient systemically ill and the eye inflamed; they do not reduce the size of the pupil. If the sphincter is functional, the pupil will contract quickly once the IOP has been lowered. On the other hand, if the surgeon is planning to perform a sector iridectomy, it is best to have the pupil dilated as widely as possible. Such a sector iridectomy probably is the preferred procedure in patients with severe iris atrophy, a dilated fixed pupil, or a cataract that is limited to the visual axis.
Operative Technique
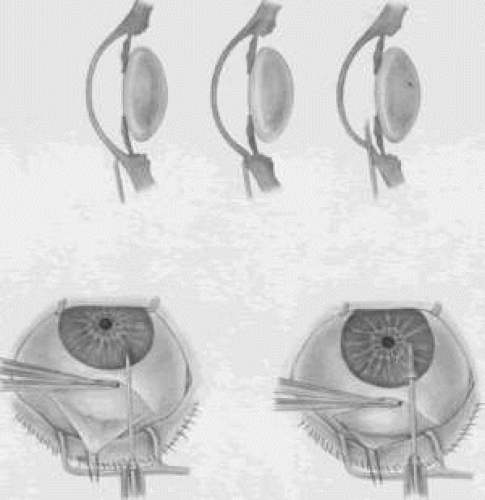
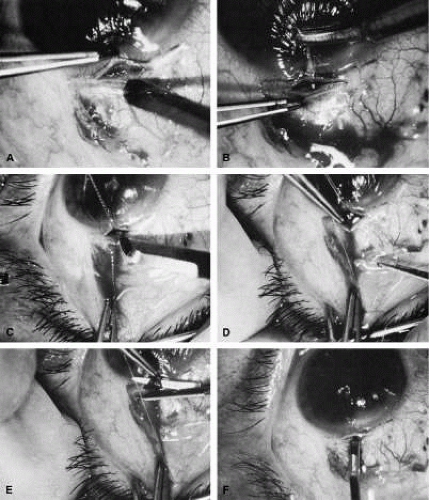
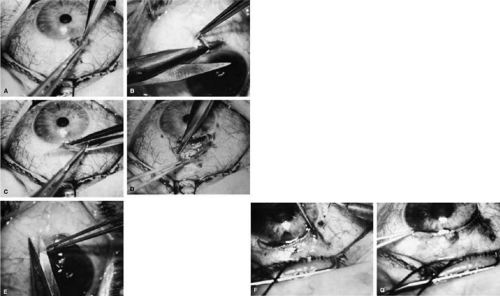
Adequate anesthesia for surgical iridectomy performed with a blade is provided by topical administration of an agent such as proparacaine 0.5% eye drops. Tetracaine also is effective, but it appears to have a slightly more irritating effect on the corneal epithelium. Proparacaine 0.5% given approximately every 30 seconds for ten doses 10 minutes before the procedure and then followed by about five instillations after the eye has been prepared and draped, immediately before the surgery, almost always gives satisfactory anesthesia. It is even possible to place a superior rectus bridal suture without causing undue discomfort.
In most cases, it is preferable to perform a facial nerve block with a modified O’Brien block.45 This approach allows easier and more comfortable placement and retention of the speculum. Because the procedure usually lasts only about 5 to 15 minutes, a short-acting injection anesthetic, such as lidocaine, is appropriate. The relevant anatomic considerations are shown in Figure 4A and B. The operative technique is shown in Figure 10. The iridectomy should be performed so that the only instruments that enter the anterior chamber are the needle that is used to develop a paracentesis track and the tip of the blade that is used to make the corneoscleral incision. The advantages of the paracentesis so greatly outweigh the minute risks associated with it that we believe that it should be performed routinely. The technique is described in detail earlier in this chapter and is illustrated in Figure 5. When properly performed, this procedure is virtually without risk, even in patients with extremely shallow or flat anterior chambers. Without such an opening into the anterior chamber, the integrity of the incision cannot be tested, blood in the anterior chamber cannot easily be irrigated away, and chamber deepening cannot readily be performed.
To prevent the tip of the knife used to develop the corneoscleral incision from damaging the iris or lens, it is preferable to use a broad blade, such as the no. 67 Beaver blade. Multiple small scratches are made, with the surgeon verifying that the depth of the incision is uniform from end to end. Some surgeons prefer an anteriorly shelved incision because it tends to close more easily; in the past, many surgeons did not place a suture through this type of incision. However, a perpendicular incision allows the iris to prolapse more readily, gives better visualization, and facilitates the procedure. The availability of fine suture material allows tight closure of the incision with minimal irritation or difficulty. The scissors used to perform the iridectomy should be sharp and should be tested immediately before the procedure to ensure their proper operating condition.
We prefer the use of a preplaced suture, as seen in Figure 10. This approach provides a clearer view of the process of creating the corneal incision, it aids in prolapsing the iris, and it allows for immediate closure with a suture that the surgeon knows is perfectly placed. Any of a number of sutures is satisfactory. Absorbable polyglactin [Vicryl (Ethicon, Somerville, NJ)] works well and has the advantage of not requiring removal. A 9-0 nylon suture also is satisfactory; it is thick enough to be used to retract the tissue yet fine enough to be well tolerated.
The small fornix-based flap can be closed by stretching it toward one side, especially if a radial relaxing incision approximately 2 mm long and extending from the limbus has been performed (Fig. 11). This closure can be done with the same suture used to close the corneoscleral incision, or it can be coapted with the wet-field cautery.
If the scissors performing the iridectomy are held as shown in Figure 12 the iridectomy will tend to be broad based but will remain basal, reducing the chance that the patient will have a disturbing sense of double vision postoperatively. It is important to stress that in a properly performed iridectomy, it is not necessary to insert the iridectomy forceps into the anterior chamber. To do so unjustifiably increases the risk of damage to the cornea, lens, and zonules. If a fine-toothed forceps such as a Bonn forceps is employed, the surgeon must be careful to pull the iris out of the anterior chamber far enough to ensure that the iridectomy will he penetrating. A nontoothed forceps, such as a McPherson tying forceps, offers the relative advantage of providing a less secure grip on the iris, requiring the surgeon to grasp more tissue. The disadvantage, however, is that control of the tissue is less certain, and the forceps must be inserted further into the incision.
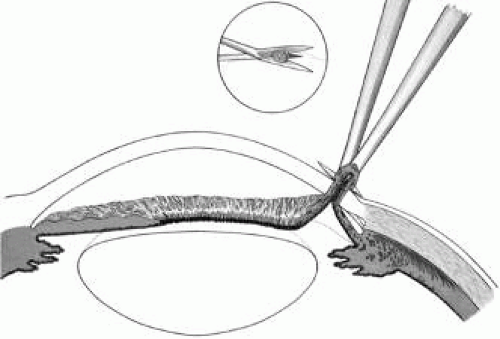 Fig. 12. Proper technique for performing iridectomy. (Spaeth GL. Glaucoma Surgery. In Spaeth GL (ed). Ophthalmic Surgery: Principles and Practice. Philadelphia: WB Saunders, 1990.) |
After the iridectomy is complete, the iris usually will return spontaneously into the anterior chamber unless it has suffered damage as a result of high pressure (as occurs in severe attacks of angle closure glaucoma). In patients with a reactive sphincter, it usually is necessary only to release the iris from the grasp of the incision. The iris can be stroked into position with the use of a blunt instrument on the corneal surface. Attempting to reposit the iris by directing a stream of irrigating solution into the incision is not recommended; the solution may enter the posterior chamber, forcing even more iris out of the incision. If the surgeon has difficulty restoring the iris to its proper position, injection through the previously placed paracentesis track of freshly mixed acetylcholine usually will solve the problem. In the eye with a dilated fixed pupil and a flaccid iris, it often is preferable to perform a sector iridectomy.
Once the pupil is entirely round, the suture placed previously in the corneoscleral incision can be pulled up and secured promptly. Injecting balanced salt or acetylcholine solution through the previously placed paracentesis can deepen the chamber. In patients who have had recent angle-closure attacks and in whom peripheral anterior synechiae may be newly developed, forceful deepening of the anterior chamber at this point may tear open the synechiae and restore normal function of the anterior chamber. The procedure of chamber deepening must be done cautiously, with careful monitoring of IOP. A gonioprism, such as the Zeiss four-mirror lens, can be used with an operating microscope to monitor the effect on anterior chamber angle.
At the close of the procedure, the pupil should be round, the chamber deep, and the incision sufficiently closed that there is no leakage. The IOP should be approximately 15 to 30 mm Hg, being raised to that level by injection through the keratostomy. Such a pressure may help prevent subsequent choroidal detachment and allows the integrity of the incision to be tested. If the corneoscleral incision has been properly placed and sutured, there should be minimal transient astigmatism. Vision should return to the preoperative level within several days.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree