17 GLAUCOMA AND RELATED CONDITIONS
Ocular Hypertension (OHT)
G. Richard Bennett and Fiaz Zaman
ICD-9: 365.01
 THE DISEASE
THE DISEASE
Pathophysiology
OHT is a condition in which elevated intraocular pressure (IOP) is found without detectable glaucomatous damage on standard clinical tests, such as automated perimetry and optic nerve head evaluation. Elevated IOP is generally defined as an IOP greater than 21 mm Hg or 22 mm Hg by Goldmann tonometry. An estimated three to six million people in the United States, including 4% to 7% of those older than 40 years, are at increased risk of developing primary open-angle glaucoma (POAG) and subsequent visual loss. There is much evidence that substantial damage occurs to the optic nerve fibers before glaucomatous damage is detectable by current clinical methods. The Ocular Hypertensive Treatment Study (OHTS) found that 9.5% of untreated ocular hypertensives suffered clearly detectable clinical damage to the visual field and/or the optic nerve in the first 60 months of that study. Prophylactic medical treatment to lower IOP reduced the risk of developing damage to the optic nerve and visual field by more than 50% during the first 60 months of the OHTS.
Etiology
The precise etiology for OHT has not yet been determined, but most authorities attribute the elevated IOP to a reduced aqueous outflow facility in patients at risk.
The Patient
Clinical Symptoms
OHT is a condition without symptoms, which sometimes leads to difficulty in educating the patient with respect to the importance of close observation and possible treatment.
Clinical Signs
- OHT is associated with elevated IOPs but a normal optic nerve head appearance and normal threshold visual fields. Central corneal thickness (CCT), as measured by corneal pachymetry, is variable but may contribute valuable information with respect to risk of progression. The OHTS demonstrated a substantially higher risk to ocular hypertensives with large cup/disc ratios but relatively thin (<555 μm) CCTs.
- Gonioscopy will demonstrate normal anterior chamber depth and anatomy.
- Ophthalmoscopy will show a healthy optic nerve head with an intact neuroretinal rim 360 degrees.
Demographics
Groups at higher risk for progression to glaucoma include patients with higher IOPs (>25 mm Hg), African Americans, older patients, patients with large vertical cup/disc ratios (>0.5), and patients with thin central corneas (CCT < 555 μm).
Note: While younger patients may not have a higher rate of developing POAG per year, they have a greater life expectancy, longer duration of exposure to elevated IOPs, and thus a significant cumulative lifetime risk of developing glaucoma. Individuals of any age, with higher risks, will benefit from early prophylactic therapy.
Significant History
A family history of POAG and a personal history of vascular disease are considered significant risk factors by many authorities.
Ancillary Tests
- Threshold visual fields
- Pachymetry
- Optic nerve photography (stereo)
- Optic nerve and/or retinal nerve fiber layer imaging (HRT III, VCC GDx, OCT)
- Gonioscopy
The Treatment
The initial management is generally close observation. The OHTS demonstrated that patients with OHT are at risk for vision loss but that damage is generally slowly progressive and that most patients will never require treatment. It is critical to identify those patients at greatest risk (high IOP, thin CCT, large cup/disc ratio) and treat those patients. Treatment is usually topical medical therapy. Laser trabeculoplasty or surgical intervention would only be considered under extraordinary conditions without demonstrable visual field or optic nerve damage.
Initial Medical Therapy
The following medications are options for initial therapy:
- Topical prostaglandin analogs (latanoprost 0.005%, travoprost 0.004%, or bimatoprost 0.01% or 0.03%) once a day, typically at bedtime
- Topical β-blocker (timolol 0.25% to 0.5%, levobunolol 0.25% to 0.5%, betaxolol 0.25% or carteolol 1.0%) qam or b.i.d.
- Topical brimonidine 0.1% or 0.2% three times a day
- Topical carbonic anhydrase inhibitors (dorzolamide 2% or brinzolamide 1%) three times a day
Primary Open-Angle Glaucoma (POAG)
G. Richard Bennett and Fiaz Zaman
ICD-9: 365.11
 THE DISEASE
THE DISEASE
Pathophysiology
POAG is a progressive optic neuropathy that is associated with an elevated IOP, progressive cupping of the optic nerve, and visual field loss. IOP is thought to be elevated because of an obstruction to aqueous outflow through the trabecular meshwork.
Etiology
While the exact etiology is unclear, elevated IOP plays an important role in the development of POAG. Glaucoma probably has a polygenetic mode of inheritance.
The Patient
Clinical Symptoms
POAG is usually asymptomatic. In advanced stages, the patient may complain of decreased peripheral or central vision.
Clinical Signs
POAG is usually bilateral but can be asymmetric. The IOP is usually greater than 21 mm Hg; however, because IOP fluctuates, it does not have to be greater than 21 mm Hg on every examination. As many as 25% of patients with open-angle glaucoma present initially with IOPs lower than 22 mm Hg. Gonioscopy demonstrates an open anterior chamber angle without anatomic abnormalities and is an important procedure in the evaluation of a glaucoma suspect. There are no specific corneal or iris abnormalities. Fundus examination (best achieved by a binocular view of the posterior pole with a 60, 78, or 90-D lens, or a fundus contact lens) can reveal increased or asymmetric cupping, as well as notching or thinning of the neuroretinal rim (Fig. 17-1). Splinter disc hemorrhages, on or adjacent to the nerve head (Drance hemorrhages), are occasionally seen and may suggest that the glaucoma is not adequately controlled, especially in patients with relatively low pretreatment IOPs (Fig. 17-2). The OHTS suggests that Drance hemorrhages may reflect a comorbidity and should be viewed as a significant finding. Defects in the nerve fiber layer may precede progressive optic nerve cupping and may be observed more easily with the use of red-free (green) illumination and photography (Figs. 17-3 and 17-4). Common visual field defects include nasal steps, arcuate defects, and paracentral scotomas, often with significant asymmetry between the superior and inferior hemifields.
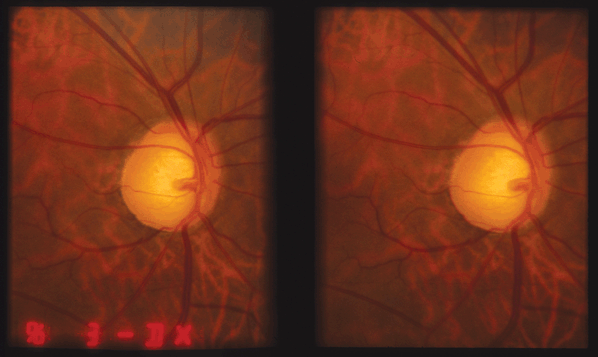
Figure 17-1. Stereo disc photo documenting large C/D ratio and inferior notch in PAOG.
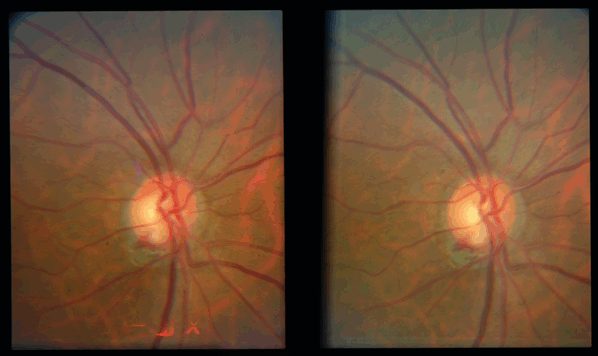
Figure 17-2. Stereo disc photo showing a small disc hemorrhage at 7:30.
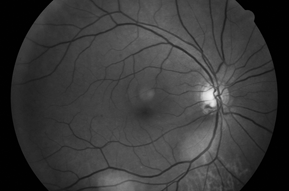
Figure 17-3. Red free photo showing an inferior disc hemorrhage with corresponding wedge nerve fiber layer defect.
Figure 17-4. Stereo disc photo showing a slit defect in the RNFL at 11:00.
Demographics
The risk factors for developing POAG include an elevated IOP, increased age, family history of glaucoma in a first-degree relative, Hispanic or African American race, high myopia (>8 D), thin central corneas, and possibly vascular diseases such as diabetes mellitus and systemic hypertension. While myopia may be a risk factor for developing glaucoma, possibly through alteration in the connective tissue support structure of the optic nerve, it does not appear to influence the rate of progression of glaucoma once treatment has been initiated. OHT is a significant risk factor, but most people with OHT do not develop glaucoma (optic nerve changes and visual field defects).
Significant History
- History of risk factors (family history of POAG, age, race, diabetes mellitus, myopia greater than 8 D, systemic hypertension, and/or systemic hypotension)
- A history of pulmonary disease, heart disease, kidney stones, depression, and drug allergies is important in determining appropriate therapy for glaucoma
Ancillary Tests
- Threshold visual fields
- Pachymetry
- Optic nerve head photography (preferably stereo images)
- Optic nerve and/or retinal nerve fiber layer imaging (HRT III, GDx, OCT)
- Color vision testing
- Diurnal IOPs
- Gonioscopy
The Treatment
There is not a “cookbook” for the treatment of POAG. Each patient must be considered individually to determine the appropriate treatment and aggressiveness of treatment. Factors to consider include age of the patient, medical history (avoid topical β-blockers in patients with emphysema, asthma, heart block, decompensated heart failure, symptomatic bradycardia), ocular history (prostaglandin analogs may exacerbate epithelial HSV keratitis, CME), drug allergies, comfort, convenience, and compliance. Finally, each practitioner should consider the safety, cost, dosage frequency, side effects, and efficacy of all medications.
Topical medical treatment may include
1. Prostaglandin Analogs
Prostaglandin analogs have become the major first-line drug in the treatment of open-angle glaucoma because of superior efficacy, convenience, and safety.
- Latanoprost (Xalatan 0.005%) at bedtime
- Bimatoprost (Lumigan 0.01% or 0.03%) at bedtime
- Travoprost (Travatan-Z 0.004%) at bedtime
2. Topical β-blockers
Topical β-blockers were traditionally the first line of medical therapy unless contraindicated by medical conditions.
A. Selective β-blockers
- Betaxolol (Betoptic 0.5% twice a day, Betoptic-S 0.25% twice a day)
B. Nonselective β-blockers
- Timolol maleate (Timoptic 0.25% twice a day, Timoptic 0.5% twice a day, Timoptic XE 0.25% or 0.5% qam, or Istalol 0.5% qam)
- Timolol hemihydrate (Betimol 0.25% or 0.5% twice a day)
- Levobunolol (Betagan 0.25% or 0.5% every day or twice a day)
- Metipranolol (Optipranolol 0.3% twice a day)—may be associated with uveitis
C. Nonselective with intrinsic sympathomimetic activity (ISA):
- Carteolol (Ocupress 1.0% twice a day)
D. Combination with dorzolamide (Cosopt twice per day = 0.5% timolol maleate + 2.0% dorzolamide)
- Combination with brimonidine (Combigan twice per day = 0.5% timolol maleate + 0.2% brimonodine)
3. Topical Carbonic Anhydrase Inhibitors (CAIs)
- Dorzolamide (Trusopt 2.0% three times a day)
- Brinzolamide (Azopt 1.0% three times a day)
4. Miotics
- Pilocarpine is started in low concentrations (0.5% to 1.0%) and increased (up to 2.0% to 6.0%) depending on the IOP and patient tolerance. Pilocarpine is frequently prescribed four times a day; however, two or three times a day dosing may be sufficient in some cases, particularly when used as concomitant therapy.
- Pilocarpine can also be prescribed as a gel (Pilopine gel 4% at bedtime).
- Other miotics (carbachol 1.5% to 3.0% three times a day, phospholine iodide 0.06%, 0.125%, 0.25% twice a day) are occasionally used in pseudophakic or aphakic patients.
- Pilocarpine can also be prescribed as a gel (Pilopine gel 4% at bedtime).
Note: Miotics are not commonly used in the United States today because of newer, more effective medications with fewer side effects.
5. Epinephrine compounds (not commonly used in the United States today)
- Dipivefrin (Propine 0.1% twice a day)
- Epinephrine 1% to 2% (Epifrin once or twice daily)
6. α-Agonists
- Apraclonidine (Iopidine 0.5% to 1% three times a day)—not generally used as a maintenance drug because of significant tachyphalaxis. Very useful in short-term lowering of IOP.
- Brimonidine 0.2% three times a day, 0.1% with Purite (Alphagan-P) preservative three times a day.
Oral Medical Treatment
Note: Oral medical treatment may be used in urgent care for very elevated IOPs but is used rarely today for chronic treatment of POAG because of significant side effects.
1. Carbonic Anhydrase Inhibitors
- Methazolamide (25- or 50-mg tablets) 25 to 100 mg orally two to three times a day
- Acetazolamide (250 mg tablets orally four times a day or 500-mg sustained release capsules orally twice a day)
Laser and Surgical Therapy
1. Laser Surgery
- Argon laser trabeculoplasty (ALT) is effective in reducing the IOP in approximately 70% to 80% of patients with POAG. The IOP frequently increases over time, and the success rate drops to 40% to 50% after 5 years.
- Selective laser trabeculoplasty (SLT) is as effective as ALT and may be repeatable after the treatment effect decreases over time.
2. Filtration Surgery
- Trabeculectomy has an 80% to 90% success rate in lowering IOP to ≤21 mm Hg in patients with POAG. Antimetabolites, such as 5-FU and Mitomycin-C, are useful in cases that are at an increased risk for failure.
3. Other Surgical Options
- Glaucoma drainage devices or tube shunt (Molteno, Baerveldt, Ahmed, etc.)
- Cyclodestructive procedures (cyclo- cryotherapy or laser transscleral cyclophotocoagulation, or endophotocoagulation of the ciliary processes)
- Nonpenetrating procedures (viscocanalostomy, canaloplasty, transciliary filtration)
Low/Normal-Tension Glaucoma (NTG)
G. Richard Bennett and Fiaz Zaman
ICD-9: 365.12
 THE DISEASE
THE DISEASE
Pathophysiology
Low-tension glaucoma or NTG is present when there is optic nerve cupping and/or visual field defects consistent with glaucoma, and IOPs less than 22 mm Hg without treatment. No other secondary cause is responsible for the cupping and visual field changes. It is critical to exclude contributing systemic diseases (vascular occlusive disease, space-occupying lesions, etc.) before establishing a diagnosis of NTG.
Etiology
NTG has been shown to be a pressure-dependent disease, with IOP lowering of 30% or more often proving beneficial in slowing the rate of progression. The etiology is elusive with non-IOP–dependent risk factors such as systemic hypotension, cardiovascular disease, atrial fibrillation, migraine syndrome, vasospasm, connective tissue disorders, anemia, sleep apnea, and hypothyroidism, potentially contributing to the disease process.
The Patient
Clinical Symptoms
Like many other types of glaucoma, patients are usually asymptomatic until the optic nerve damage is severe enough to cause significant visual loss.
Clinical Signs
Optic nerve cupping is present and can be asymmetric. Superficial disc hemorrhages and peripapillary atrophy appear to be more common in low-tension glaucoma compared to POAG and may have a greater significance with respect to progression (Fig. 17-5). Visual field defects are frequently like those in POAG. However, the defects may be denser and steeper and may involve the paracentral area with encroachment of fixation. IOP is consistently less than 22 mm Hg. The angle is open by gonioscopy without peripheral anterior synechiae. Other retinal and optic nerve anomalies (i.e., colobomas, optic nerve pits, tilted discs, myopic discs, optic nerve head drusen, ischemic optic neuropathy, hereditary optic neuropathies, infection, macular disease, retinal vascular occlusions) that may cause glaucomatous-like visual field defects must be excluded.
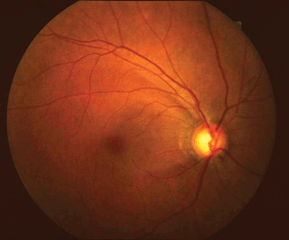
Figure 17-5. Flat fundus photo. Note the disc hemorrhage at 5:00, RNFL defects, and peripapillary atrophy.
Demographics
Low-tension glaucoma is more common in elderly patients. It is unusual to diagnose low-tension glaucoma in patients less than 50 to 60 years of age.
There appears to be no significant difference in the progression rate between myopic and nonmyopic normal tension glaucoma patients who initiated antiglaucoma therapy.
Significant History
It is important to note other causes of glaucoma or other conditions that could produce similar optic nerve and visual field changes.
Previous ocular trauma, steroid use, uveitis, nocturnal hypotensive episodes, hemodynamic crisis, sleep-disordered breathing or obstructive sleep apnea, anemia, increased blood viscosity, hypercoagulability, dyslipidemia, carotid artery disease, migraine, ischemic optic neuropathy, or intermittent angle-closure glaucoma should be investigated and excluded.
Patients taking oral β-blockers or instilling topical β-blockers at nighttime may be at increased risk for progressive vision loss because of nocturnal hypotension. Oral β-blockers can also mask elevated IOPs.
The diagnosis of NTG should be approached as one of exclusion. Suggested testing includes
- Automated visual fields—defects tend to be more focal, deeper and closer to fixation.
- Stereoscopic evaluation of the optic nerve (with a 60 or 78-D lens, fundus contact lens, or Hruby lens). Patients with NTG have a greater incidence of zone beta parapapillary atrophy (PPA) and more frequent disc hemorrhages than patients with POAG.
- Pachymetry is indicated as thin corneas can underestimate the true IOP.
- Gonioscopy excludes intermittent angle closure and other secondary causes of glaucoma.
- Optic nerve and/or retinal nerve fiber layer imaging (HRT III, GDx, OCT).
- Color vision testing.
- Hematologic studies including a CBC (for anemia or polycythemia), RPR, FTA-ABS (for syphilis), ANA (for connective tissue disorders), and an ESR and CRP (for temporal arteritis in the elderly) may be useful in some cases.
- Blood pressure and pulse should be measured and tilt testing should be considered. Some patients may benefit from ambulatory blood pressure monitoring.
- Diurnal IOP measurements (IOP measurements at different times of the day) should be obtained prior to initiating therapy to identify large variations in IOP.
- It may be appropriate to have the patient see an internist for a complete exam, emphasizing the need to investigate for cardiovascular disease and carotid occlusive disease.
- Neuroimaging (CT scan or MRI) should be considered in unusual or atypical cases, such as
1. Patients less than 50 years of age
2. Visual fields that are rapidly progressive, out of proportion to optic disc changes, or show defects that respect the vertical midline
3. Central vision loss (<20/40)
4. Asymmetry of the nerves or visual fields with symmetrical IOPs
5. Discs that show more neuroretinal rim pallor than cupping
One should also be suspicious when finding an APD that does not correspond to the degree of asymmetry in the visual fields or the optic discs.
The Treatment
Treatment for low-tension glaucoma is similar to POAG. The IOP should be lowered by at least 30% below the level where damage has occurred, which is often best identified by diurnal measurements. While not all untreated patients with NTG show progression, a faster rate of progression has been observed in women, patients with migraine headaches, and in the presence of disc hemorrhages.
Reduction in pressure may be accomplished by medical therapy, laser trabeculoplasty, or filtration surgery. Once the target IOP has been achieved, IOP measurements should be obtained at various times of the day to exclude diurnal fluctuations.
Medical Therapy
One should consider initiating a monocular trial of the medication so that the contralateral eye can be used as a control to access efficacy and adverse effects.
- Brimonidine 0.1% or 0.2% three times a day. (Some studies demonstrate that nighttime use could potentially lower the arterial blood pressure in certain patients, thus adversely affecting the diastolic ocular perfusion pressure [DOPP] to the optic nerve.)
- β-blockers: Betaxolol 0.25% to 0.5% twice a day, carteolol 1.0% twice a day, and so on. (Again, some authorities contend that topical nonselective β-blockers may interfere with perfusion to the ONH and may not be the ideal therapy in NTG.)
- Topical carbonic anhydrase inhibitors: Dorzolamide 2% or brinzolamide 1% three times a day—CAIs may improve the DOPP.
- Latanoprost (Xalatan 0.005%), travoprost (Travatan or Travatan-Z 0.004%), or brimatoprost (Lumigan 0.01% or 0.03%) once a day—like the CAIs, the prostaglandin analogues (PGAs) may help to improve the ocular perfusion pressure.
- Some studies have suggested the use of oral calcium-channel blockers in patients with low-tension glaucoma who have progressive cupping or visual field loss. Proof of their safety and efficacy awaits the results of future long-term studies; however, treatments for glaucoma based on systemic alterations of blood pressure and of ocular blood flow, do not appear to be viable options at this time.
- It is advised that NTG patients avoid taking antihypertensive agents just prior to sleep, as resultant lowering of blood pressure and reduced perfusion to the optic nerve may occur.
- Ginkgo biloba extract (40 mg p.o. t.i.d.) has been reported to improve visual field indexes in some NTG patients.
- Topical carbonic anhydrase inhibitors: Dorzolamide 2% or brinzolamide 1% three times a day—CAIs may improve the DOPP.
Laser and Surgical Therapy
- ALT or SLT may be effective in some low-tension patients; however, the results have been inconsistent.
- Filtration surgery is used when progressive optic nerve cupping and visual field loss occur and there is significant danger of loss of quality of life and the IOP cannot be lowered with more conservative therapy.
Steroid-Induced Glaucoma
G. Richard Bennett and Fiaz Zaman
ICD-9: 365.03
 THE DISEASE
THE DISEASE
Pathophysiology
Steroid-induced elevations in IOP appear to be secondary to decreased facility of aqueous outflow. There are several proposed mechanisms: (a) structural changes of the trabecular meshwork, (b) mechanical obstruction of the trabecular meshwork by steroid particles, extracellular matrix, or glycosaminoglycans, (c) debris in the trabecular meshwork because of inhibition of phagocytosis by trabecular meshwork cells, or (d) steroid-induced proteins (myocilin) may initiate the response.
Etiology
Elevated IOP has been associated most commonly with the use of topical steroid eyedrops or periocular repository steroid injections. The pressure response can occur within a week or may be delayed for months or years. Systemic steroids, steroid creams used on the skin near the eye or in areas away from the eye, inhaled steroids, injected steroids, implantable steroids, and nasal corticosteroids have all been associated with an increase in IOP.
The Patient
Clinical Symptoms
The patient is usually asymptomatic unless the IOP increases enough to cause corneal edema (decreased vision, halos, photophobia) or pain. The patient may have symptoms from the ocular disorder that is being treated with steroids (photophobia from iritis).
Clinical Signs
Prolonged elevations of IOP can cause optic nerve cupping and visual field defects similar to POAG. Clinical signs of the ocular disorder that is being treated with steroids may be seen (cells, flare, anterior or posterior synechiae, or engorged iris vessels from iritis), as can other ocular side effects of chronic ocular steroid use, such as posterior subcapsular cataracts or atrophy of the skin near the eyelids.
Demographics
Steroid-induced elevations in IOP are more common in glaucoma suspects and patients with preexisting glaucoma, high myopes (>5 D), diabetics, and in those with a family history of glaucoma.
Approximately one third of the normal population will develop a moderate increase in IOP following topical ocular corticosteroid use; however, 5% to 6% will show a markedly elevated IOP response.
Significant History
Significant history includes the type of steroid and the duration of use, previous ocular disorders, a history of diabetes mellitus or myopia, or a family history of glaucoma.
Ancillary Tests
- Threshold visual fields
- Stereo disc photographs
- Gonioscopy
- Pachymetry is indicated as thin corneas can underestimate the true IOP.
- Optic nerve and/or retinal nerve fiber layer imaging (HRT III, GDx, OCT)
The Treatment
Steroid-induced IOP elevations almost always respond within days to weeks of discontinuing the steroid. It is not always possible to abruptly stop the steroid because of the underlying ocular disorder being treated. In patients with iritis, it is often difficult to determine whether the increased IOP is because of steroids or inflammation. If iritis is still present, the steroid should be continued to treat the inflammation, with the addition of medical therapy to reduce the IOP. When the inflammation has decreased, the steroid can be tapered.
Options for treatment include
- Decreasing the effects of steroids by
- Discontinuing the steroid.
- Decreasing the frequency or strength of the steroid being used.
- Using a different type of steroid (fluorometholone, rimexolone, loteprednol, or medrysone are touted as being less prone to pressure elevations in some patients compared to prednisolone or dexamethasone). Of the commonly used ocular steroids, loteprednol is an ester steroid, which may shorten the active life of the drug, thus possibly reducing unwanted side effects.
- Using nonsteroidal anti-inflammatory eye drops (diclofenac 0.1%, nepafenac 0.1%, bromfenac 0.09%, ketorolac 0.5%), instead of steroids.
- Discontinuing the steroid.
- Medications to decrease the IOP may be needed if the IOP is very high or if the elevated IOP persists after changing, reducing, or stopping the steroid.
- β-blockers (timolol 0.25% to 0.5% twice a day, levobunolol 0.5% once or twice a day, Timoptic XE 0.25% to 0.5% once a day, carteolol 1.0% twice a day, betaxolol 0.25% to 0.5% twice a day).
- Apraclonidine 0.5% three times a day or brimonidine 0.1% or 0.2% three times a day.
- Carbonic anhydrase inhibitors (dorzolamide 2% three times a day, brinzolamide 1% three times a day, methazolamide 25 to 50 mg one to two tablets orally two to three times a day, or acetazolamide 250 mg orally four times a day or 500-mg sustained-release capsules orally twice a day).
- Miotics and prostaglandin analogs should be avoided if the patient has recently had ocular inflammatory disease.
- Removal of an injected intraocular or periocular steroid may be required.
- Laser trabeculoplasty is typically not effective for steroid-induced OHT
- Occasionally, glaucoma surgery is necessary if the IOP is at alarming levels (>50 mm Hg) or if the IOP does not respond to discontinuing the steroid and/or medical therapy.
- Laser trabeculoplasty is typically not effective for steroid-induced OHT
Glaucoma Associated with Inflammation
G. Richard Bennett
Fiaz Zaman
ICD-9: 365.62
 THE DISEASE
THE DISEASE
Pathophysiology
Intraocular inflammation can cause an elevated IOP by several different mechanisms. Inflammation can increase resistance to aqueous outflow by causing dysfunction or swelling of trabecular sheets and endothelial cells, or by causing a breakdown in the blood–aqueous barrier, allowing inflammatory cells or fibrin to accumulate in the trabecular meshwork. Production of inflammatory prostaglandins may produce elevated IOP by causing a breakdown in the blood–aqueous barrier. If posterior synechiae, secondary to inflammation, obstructs the flow of aqueous humor from the posterior to the anterior chamber, the peripheral iris will bow forward, causing iris bombe. This bowing can cause acute or chronic angle-closure glaucoma. Peripheral anterior synechiae formation may cause chronic angle-closure glaucoma. In many instances, multiple mechanisms occur simultaneously.
Etiology
There are many etiologies of intraocular inflammation (see sections on “Uveitis”). Frequently, the cause of iridocyclitis is never determined.
One must distinguish uveitic glaucoma from angle-closure glaucoma, which may produce a mild to moderate iritis.
The Patient
Clinical Symptoms
Photophobia, red eye, pain, and decreased vision may be present.
Clinical Signs
Elevated IOP, ciliary flush, flare and cells in the anterior chamber and anterior vitreous, miosis, keratic precipitates, and engorged iris vessels may be seen. Gonioscopy is important to determine whether the angle is open or whether peripheral anterior synechiae partially or completely close the angle. Chronically elevated IOP may cause optic nerve and visual field changes.
Demographics
The demographics of the group at risk will depend on the etiology of the intraocular inflammation.
Significant History
- Previous history of intraocular inflammation
- Duration of current symptoms
- Presence of systemic diseases associated with intraocular inflammation
- Sarcoidosis, tuberculosis, syphilis, etc.
- Ankylosing spondylitis
- Juvenile idiopathic arthritis
- Behçet’s disease, ulcerative colitis, Crohn’s disease, etc.
- Sarcoidosis, tuberculosis, syphilis, etc.
- History of trauma
Ancillary Tests
See the section on “Uveitis” for the evaluation and suggested ancillary testing for suspected etiologies of intraocular inflammation. Secondary causes of glaucoma should specifically be excluded. Usual glaucoma testing (visual fields, gonioscopy, optic nerve photography, etc.) may be indicated in some patients.
- Topical steroids are titrated, depending on the severity of intraocular inflammation. The specific steroid used will depend on the amount of inflammation and how well the IOP can be controlled. Traumatic uveitis may not require aggressive therapy, while some inflammatory situations require frequent (every hour) instillation of steroids. Prednisolone acetate 1.0% and dexamethasone 0.1% are potent anti-inflammatory agents and are very useful in treating moderate to severe inflammatory disease. However, these agents often show a higher incidence of steroid-induced elevated IOP when compared to “soft steroids” such as fluorometholone 0.1% or 0.25%. Rimexolone 1% or loteprednol 0.5% also offer potent anti-inflammatory properties with a potentially lower incidence of IOP elevation than prednisolone acetate, prednisolone phosphate, or dexamethasone. Certain inflammatory conditions (epithelial herpes simplex keratitis, fungal infections, etc.) may preclude intervention with topical steroids.
- Periocular or systemic steroids are sometimes indicated for severe inflammation or inflammation that is not responsive to topical steroids.
- Cycloplegic eye drops, one to three times per day (scopolamine 0.25%, homatropine 5.0%, cyclopentolate 1.0%, or atropine 1.0%), are used to decrease pain from ciliary spasm and decrease the potential for posterior synechiae.
- Topical nonsteroidal anti-inflammatory drops (ketorolac 0.5%, diclofenac 0.1%) are sometimes helpful as an adjuvant to topical steroids. They are titrated according to the level of inflammation.
- IOP Management
- Topical β-blockers (timolol 0.25% to 0.5% twice a day, levobunolol 0.25% to 0.5% twice a day, Timoptic XE 0.25% to 0.5% once a day, carteolol 1.0% twice a day, betaxolol 0.25% to 0.5% twice a day)
- Carbonic anhydrase inhibitors (dorzolamide 2% or brinzolamide 1% drops three times a day, methazolamide 25 to 50 mg one to two tablets orally two to three times a day, acetazolamide 250 mg orally four times a day or 500-mg sustained-release capsules twice a day)
- Topical apraclonidine 0.5% three times a day or brimonidine 0.1% or 0.2% three times a day
- Oral or intravenous osmotic agents if the initial pressure is very high (oral isosorbide 45% 1.0 to 1.5 g/kg, oral glycerol 50% 1.0 to 1.5 g/kg or IV mannitol 20% 1 to 2 g/kg over 45 to 60 minutes)
- Occasionally glaucoma filtration surgery with local antimetabolite therapy (mitomycin-C or 5 FU) is indicated if the IOP cannot be controlled medically
- Topical β-blockers (timolol 0.25% to 0.5% twice a day, levobunolol 0.25% to 0.5% twice a day, Timoptic XE 0.25% to 0.5% once a day, carteolol 1.0% twice a day, betaxolol 0.25% to 0.5% twice a day)
Note: Miotics (pilocarpine), prostaglandin analogs, and laser trabeculoplasty should be avoided while inflammation is present.
- The underlying systemic disease, if present, should be treated.
Glaucomatocyclitic Crisis (Posner-Schlossman’s Syndrome)
Nicky R. Holdeman
ICD-9: 364.22
 THE DISEASE
THE DISEASE
Pathophysiology
Glaucomatocyclitic crisis is an unusual type of acute, typically unilateral, recurrent uveitis, and secondary glaucoma.
The exact mechanism of this condition is unknown; however, trabeculitis may contribute to the pathogenesis, as tonographic studies indicate a reduction in outflow facility during an attack.
Etiology
While the exact etiology has yet to be defined, herpes virus, cytomegalovirus (CMV), and prostaglandin E have all been implicated as possible causes. Helicobacter pylori infection has been shown to occur significantly more often in Posner-Schlossman’s syndrome (PSS) patients than in the general population.
Some research has shown that about half of all presumed cases of PSS are CMV positive, but there are no clinical features that can differentiate between CMV-positive and CMV-negative eyes.
Increased levels of prostaglandins in the anterior chamber may account for the decreased facility of outflow and breakdown of the blood–aqueous barrier.
The Patient
Clinical Symptoms
In general, symptoms are remarkably mild and few in relation to the magnitude of the IOP.
The patient may be asymptomatic, or may report slight discomfort, redness, blurred vision, and halos if the IOP is significantly elevated and corneal epithelial edema is present.
Clinical Signs
Glaucomatocyclitic crisis is generally unilateral and is associated with a significant increase in IOP (>30 mm Hg and often between 40 and 60 mm Hg). Inflammation is minimal. There are a few cells and mild flare in the anterior chamber (grades ½ to 2+). Keratic precipitates appear for a few days; are small, flat, and nonpigmented; and tend to appear in the lower third of the cornea. There may be mild corneal epithelial edema and the conjunctiva will have only minimal injection. The angle is open, and there are no anterior or posterior synechiae. Slight pupillary constriction, dilation, or slowly reactive pupil may be observed in the affected eye.
Between attacks, all tests are within normal limits, including the IOP and outflow facility. On occasion, a “sentinel” keratic precipitate is seen during the quiescent phase.
Demographics
Usually occurs in patients between the ages of 20 and 50 years and is seldom seen after the age of 60.
Significant History
The patient may report intermittent episodes of unilateral blurred vision and ocular discomfort, as recurrent attacks are common. Recurrences tend to manifest at intervals of a few months to several years.
Ancillary Tests
The implication of CMV as an etiologic agent may be important, as effective treatments for CMV infection exist and have been used for many years. Confirmation of aqueous CMV, by polymerase chain reaction (PCR), would be a prerequisite before considering systemic antiviral agents (i.e., valganciclovir, gancyclovir) in these patients.
There appears to be an association of glaucomatocyclitic crisis with primary open-angle glaucoma. Some patients can experience optic nerve cupping and visual field loss due to repeated attacks or due to underlying POAG. Consequently, patients with PSS should undergo a complete glaucoma evaluation of both eyes (e.g., gonioscopy, pachymetry, nerve fiber layer analysis, stereoscopic disc photos, and baseline visual field-testing).
It is always prudent to exclude other common etiologies of uveitis by requesting an RPR, FTA-ABS, HLA-B27, and a CXR.
Glaucomatocyclitic crisis is self-limited and will usually resolve with or without treatment. The episodes typically last from several hours to a few weeks. Between attacks, the patient is asymptomatic, the IOP is normal, and there is no intraocular inflammation.
The treatment of glaucomatocyclitic crisis is aimed at controlling the IOP, decreasing inflammation, and maintaining ocular comfort during an acute attack. Further study will be required in order to elucidate the relationship between H. pylori infection and PSS and to determine if treatment for H. pylori may improve the clinical course of the disease.
During an acute episode, treatment may include
- Topical β-blockers q.d. or b.i.d.
- Topical α-agonists b.i.d.–t.i.d.
- Topical carbonic anhydrase inhibitors or oral CAI if necessary
- Topical steroids—prednisolone acetate 1% q.i.d.
- Cycloplegic agents if needed for comfort. Cycloplegics are seldom required as there is little ciliary muscle spasm and synechiae do not form.
- Oral NSAIDs and topical NSAIDs may be effective in reducing inflammation and in lowering the IOP in some patients.
- Should a CMV infection be confirmed, valganciclovir is a potential treatment. However, valganciclovir is an expensive medication with potential for renal and bone marrow toxicity.
Note: Patients with PSS may show an elevated IOP in response to topical steroids. Prolonged treatment with corticosteroids should be avoided.
Uveitis has been considered a relative contraindication to the use of PGAs due to the risk of inflammation and CME that have been reportedly associated with this class of glaucoma medications. Recent studies suggest that these problems are not likely to occur, and that patients with uveitic glaucoma may benefit from the significant IOP lowering effects of these drugs. However, it might be prudent to avoid PGAs in favor of other medications if possible and to use them as a latter option before proceeding to more invasive interventions.
Laser trabeculoplasty (ALT/SLT) is generally not effective in controlling the elevations in IOP associated with PSS.
Neovascular Glaucoma (NVG)
G. Richard Bennett and Fiaz Zaman
ICD-9: 365.63
 THE DISEASE
THE DISEASE
Pathophysiology
Retinal hypoxia and ischemia, leading to subsequent release of angiogenic factors such as vascular endothelial growth factors (VEGFs), provides the stimulus for neovascularization of the retina, iris (NVI), and the anterior chamber angle (NVA). Neovascularization of the anterior chamber angle consists of a fibrovascular membrane that can obstruct outflow through the trabecular meshwork either by covering the anterior chamber angle or by contracting, which causes peripheral anterior synechiae formation.
Etiology
The most common causes of retinal hypoxia, resulting in NVG, are diabetic retinopathy, central retinal vein occlusion (CRVO), and ocular ischemic syndrome (carotid occlusive disease). Some of the other etiologies include branch retinal vein occlusion, central retinal artery occlusion, chronic retinal detachment, chronic uveitis, intraocular tumors, radiation therapy, and trauma.
Clinical Symptoms
Occasionally NVG is asymptomatic. More often, symptoms include red eye, pain, decreased vision, and photophobia. Nausea, vomiting, and headache may be reported if the elevation of IOP is acute and/or the IOP is very high.
Clinical Signs
NVI (rubeosis) can be confined to the pupillary margin (Fig. 17-6) or may extend into the angle (Fig. 17-7). Conjunctival injection, corneal edema, iritis, significant elevations of IOP, hyphema, and ectropion uvea are frequently present. Gonioscopy may reveal an open angle with or without neovascularization or may demonstrate partial or complete closure secondary to anterior synechiae. Retinal examination may reveal proliferative diabetic retinopathy (PDR), retinal hemorrhages, or shunt vessels from previous vascular occlusions, or other retinal pathology that may help determine the cause of ocular ischemia.
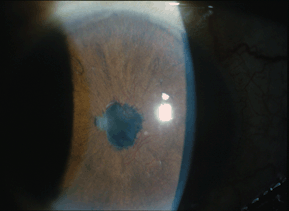
Figure 17-6. NVI pupillary margin with several areas of posterior synechia in a patient with NVG.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


