Fig. 32.1
Allergens bind FcεRI and trigger Syk-mediated signaling pathway resulting in mast cell degranulation, eicosanoid mediator synthesis, and cytokine production. Ionomycin, a calcium mobilization agent, can also induce degranulation of mast cells
Ramasamy et al. reported association of three loci for either AR or grass sensitization through evaluation of genome-wide meta-analysis of genetic variants. The HLA variant rs7775228, which cis-regulates HLA-DRB4, was strongly associated with grass sensitization and weakly with AR. Variants in a locus near chromosome 11 open reading frame 30 (C11orf30) and leucine-rich repeat containing 32 (LRRC32), which was previously associated with atopic dermatitis and eczema, were also strongly associated with both phenotypes (rs2155219). The third genome-wide significant variant was rs17513503, which is located near transmembrane protein 232 (TMEM232) and solute carrier family 25, member 46 (SLC25A46). They also observed strong associations of both AR and IgE sensitization to grass with a common (minor allele frequency, 47 %) polymorphism in the 11q13.5 locus (Ramasamy et al. 2011).
Lu et al. (2011) found that the CT/CC genotypes in IL-4 C-590T were associated with a significantly decreased risk of mite-sensitized PER (adjusted odds ratio (OR) = 0.64, 95 % confidence interval (CI) 0.45–0.92), compared to the TT genotype. Moreover, persistent allergic rhinitis (PER) patients with CT/CC genotypes had significantly lower serum levels of total IgE than those with TT genotype (P = 0.001). However, there was no significant association of the IL-13 and IL-4RA polymorphisms with mite-sensitized PER (P = 0.05).
Kruse et al. (2012) reported genome-wide significant linkage to a novel AR locus at 1p13 and suggestive linkage to two novel regions at 1q31-q32 and 20p12. The locus has previously been demonstrated to have suggestive linkage with asthma (Denham). Possible candidate genes are the glutathione S-transferase M1 (GSTM1) (Ober and Hoffjan 2006) and acidic mammalian chitinase (CHIA) genes located at 1p12-p13, earlier shown to be associated with asthma (Bierbaum et al. 2005). Chromosome 20p12 is also a novel finding in regard to AR. However, it has previously been found to have linkage with atopy as well as asthma (Denham et al. 2008).
Several studies have found an association of filaggrin (FLG), a filament-associated protein that binds to keratin fibers in epithelial cells with various allergic conditions and diseases including atopy as well as atopic dermatitis (AD), asthma, and AR (Rodriguez et al. 2008).
At 2q13-q14, the interleukin 1 gene cluster (IL-1A, IL-1B, and IL-1RN) previously associated with AR (Joki-Erkkila et al. 2003).
At 20p13, a disintegrin and metalloproteinase domain 33 (ADAM33) gene residing almost 3 Mb from the maximum linkage signal has previously been associated with AR. Toll-like receptors 7 and 8 (TLR7 and TLR8) located at Xp22 confer susceptibility to several allergic diseases and among these are AR (Kruse et al. 2012).
Yousri et al. (Hussein et al. 2012) showed that the genotype and allele frequencies of the TLR2 Arg753Gln and TLR4 Asp299Gly polymorphisms are not significantly different between asthmatic children or allergic rhinitis as compared to controls (P > 0.05 for each) or even when compared further with IgE level. However, it is shown that the mutant allele of TLR2 or TLR4 polymorphisms was significantly associated with the moderate to severe groups compared to the mild group in both atopic asthmatics and allergic rhinitis group (P > 0.001 for each). In conclusion, their study demonstrated a lack of association of TLR2 and TLR4 polymorphisms with asthma and allergic rhinitis but suggests significant association between these genetic variants and the disease severity.
32.4.5 Allergic Rhinitis and Epigenetics
The immune system is heavily affected by environmental changes. One of the good and current examples of this situation is the remarkable increase of all immune diseases with urbanization. Likewise, rising prevalence of immune diseases in infancy indicate that there may be an essential early period of sensitivity. During fetal life, essential arrangements occur such as structure, function, and response patterns of many systems. So, elucidating early events may offer important insights into the pathogenesis of the disease, as well as the pathways of environmental influence.
A variety of environmental exposures in pregnancy such as maternal diet (Prescott and Saffery 2011), pollutants like cigarette smoke (Noakes et al. 2003), and microbial exposure (Prescott et al. 2008) have been shown to alter immune function. Conspicuously, the same determinants were identified as potential immune modifiers in epidemiological studies of allergic disease.
The effects of diet are potentially complex. More studies are needed to examine the effects of related dietary nutrients such as vitamins B2, B6, and B12, methionine, and choline, which may be implicated in epigenetic effects through their effects on folate-mediated one-carbon metabolism.
Belinsky et al. also showed that exposure to the ultrafine particulate matter found in pollution may also change DNA methylation in maternal and fetal DNA and may be associated with altered inflammatory response pathways.
There is current exciting evidence that some pathogens can affect the epigenetic profile of the host cell, influencing or mimicking mechanisms that participate in DNA methylation and histone modification (Arbibe et al. 2007). The studies also showed that allergy protection by in utero microbial exposure in rural farming environments is correlated with enhanced neonatal Treg (regulatory–suppressor T cells; these cells downregulate the immune system, thus keeping tolerance to self-antigens) function, FOXP3 expression, and associated epigenetic effects (hypomethylation) of the FOXP3 gene (Schaub et al. 2009).
Several specific nutritional changes have been correlated with the rising allergic propensity, including a decrease in omega-3 polyunsaturated fatty acids (n-3PUFA), soluble fiber, antioxidants, and other vitamins (Devereux et al. 2007), based on epidemiological associations and immunological effects. Similarly, antioxidants have been shown to have effects on T cell regulation and induction of IL-12 production by antigen-presenting cells (Utsugi et al. 2003). So, this could induce development of Th1 and repress Th2 responses. Devereux et al. showed an immunomodulatory role for maternal dietary antioxidants during pregnancy; this, combined with evidence that oxidative stressors can trigger epigenetic changes and alter disease risk (see below), indirectly suggests a role for these pathways (Breton et al. 2009).
A recent study in mice also showed that exposure to diesel exhaust particles augments the production of IgE after allergen sensitization through the hypermethylation of IFNγ and hypomethylation of the IL-4 locus (Liu et al. 2008). In placental tissue, nicotine has also been shown to alter cytokine production via NFκB (Dowling et al. 2007).
The current and remarkable rise in allergic diseases and the very early onset of disease indicate that in utero events have a more essential effect on immune development and allergic susceptibility than genomic inheritance. Above all, as epigenetic modifications are commonly reversible, this may provide for the development of novel therapeutic compounds that may be efficacious in arresting or even reversing the allergy epidemic (Prescott and Saffery 2011).
A recent large gene expression microarray study of unstimulated CD4+ T cells found no differences between allergic and nonallergic individuals (Hansel et al. 2008).
The existence of monozygotic (MZ) twins who are discordant for intermittent allergic rhinitis (IAR) suggests disease mechanisms that are independent of genetics (Bell and Spector 2011). Sjogren et al. for the first time performed mRNA and microRNA expression microarray analyses of CD4+ T cells from MZ twins discordant for IAR. They analyzed the CD4+ T cells outside the pollen season, after in vitro allergen challenge and during the pollen season without in vitro allergen challenge. The allergen challenge in vitro resulted in significant differences in mRNA and protein levels between the allergic and healthy twins. The cytokines IL-4, IL-5, and IL-13 were increased in the supernatants from allergic twins. In their study, they performed microRNA expression arrays of all the twin pairs, but found no significant differences. Next, blood samples were taken during pollen season from four MZ twin pairs discordant for IAR (Sjogren). Consequently they identified disease-relevant mRNAs and proteins that differed between the discordant MZ twins. No differences in microRNA expression were determined. Because the MZ twins are almost genetically identical, the observed altered expression of essential disease-associated genes and proteins may have epigenetic causes (Sjogren et al. 2012).
In conclusion, the role of genetic as well as epigenetic factors in the pathophysiology of nasal diseases, including allergic rhinitis, is becoming more evident. Environmental effects on genetic variants and epigenetics determine the fate of chronic nasal diseases. Clarification of such relationships and mechanisms will lead to novel molecular diagnostic and therapeutic approaches for these kinds of diseases.
32.5 Role of Genetics in Chronic Rhinosinusitis
32.5.1 Introduction
The first contact of the respiratory system with the external environment occurs in the nose, which is responsible for air filtering, humidification, and temperature regulation. Because of this close contact of the respiratory mucosa with a great variety of allergens and pathogens, upper respiratory illnesses are one of the most frequent diseases in humans. Chronic rhinosinusitis (CRS) is characterized by a chronic inflammatory condition of the sinonasal mucosa, but it is mostly defined by its clinical manifestation rather than inflammation pattern. CRS presents with chronic symptoms such as nasal congestion, anterior or posterior nasal drainage, hyposmia, and facial pain (Tomassen et al. 2011). It is highly prevalent. According to an analysis of the 2008 National Health Interview Survey data, almost 1 in 7 adults suffered from rhinosinusitis (Pleis et al. 2009). Additionally, it has a huge effect on quality of life and health-care expense in terms of antibiotic prescriptions filled, lost work days, and lost school days (Bhattacharyya 2009). The disease is characterized by chronic inflammation of the sinonasal mucosa, and because inflammation of the nasal and sinus mucosa often coincides, it is named “rhinosinusitis” in current literature. It is an attempt to stress the concept that patients present with symptoms attributable not only to the sinuses but also to nasal inflammation that might often, but not always, be present.
The term chronic refers to symptoms persisting for more than 12 weeks with no definitive resolution. Also, the most recent consensus definitions subclassify CRS into CRS without nasal polyposis (CRSsNP), CRS with nasal polyposis (CRSwNP), and allergic fungal rhinosinusitis (AFRS) (Hamilos 2011). A study showed that the inflammatory mediator profile in the nasal mucosa of patients with CRS without nasal polyps (CRSsNP) and CRS with nasal polyps (CRSwNP) is similar. It further strengthens the idea that rhinitis and sinusitis can indeed be classified as one disorder entity and supports the use of the term rhinosinusitis. Although primary allergic or upper respiratory tract infectious inflammation in the nose can cause sinus ostia obstruction and subsequent sinusitis, it remains unproved from prospective studies whether and how often this occurs.
Historically, chronic sinusitis has been considered a single unimodal clinical disease. But in recent years chronic sinusitis has been recognized as comprising several diseases with different causes, with each one characterized by a definite histological pattern and gene and protein expression. Identification of specific disease subgroups and their etiologies is important for correct differential diagnosis and to apply appropriate therapeutic intervention.
32.5.2 Chronic Sinusitis (CS)
For diagnosis of CS, it is important to see symptoms of nasal irritation, anterior and posterior rhinorrhea, and nasal congestion with the accompanying presence of pressure or pain in a “sinus” distribution area that last for more than 12 weeks (Slavin et al. 2005). But validation with nasal endoscopy or computed tomography (CT) imaging is important because there can be significant overlapping of these symptoms that are very likely to be of migraine or midfacial pain syndrome origin (Perry et al. 2004). Chronic sinusitis has been divided into two subgroups depending on presence or absence of NPs (Slavin et al. 2005). Some researchers determined a significant correlation between nasal polyposis and the presence of tissue eosinophilia. However, both the presence and degree of eosinophilia in NPs can be quite variable, and researchers did not observe eosinophilia in a large patient group with idiopathic nasal polyposis (Bachert et al. 2001). These results support the opinion that certain forms of CS such as allergic fungal sinusitis (AFS) (Schubert and Goetz 1998) and aspirin-exacerbated respiratory disease (AERD) (Mascia et al. 2005) may be more likely to produce NPs and present rich eosinophilic infiltrate into the sinus cavity. Polyposis can present as a complication in any form of CS. So it should not be used as the basis for diagnosis or decisions regarding treatment. Evaluation of the presence or absence of NPs can be especially important in clinical practice for identifying patients who are more or less likely to have eosinophilic disease and thereby also identifying patients who are more or less likely to respond to eosinophil-targeted medicines. However, it should be emphasized that, in practice, CS presents as a spectrum of disorders in which the level of eosinophilia and predilection for polyposis exist on a continuum. Although we will stress the different features of the pathology of eosinophilic and noneosinophilic forms of these diseases, in daily life patients can generally present with variable overlapping symptoms.
32.5.3 Remodeling Theories
When we look at the histological investigation of CRS, two different remodeling types are seen. One of them is CRSsNP, which is associated with fibrosis, basement membrane thickening, subepithelial edema, goblet cell hyperplasia, and mononuclear cell infiltration. The other is CRSwNP, which is associated with an edematous stroma with albumin deposition, pseudocyst formation, and subepithelial and perivascular inflammatory cell infiltration.
Remodeling is a dynamic balance between production and degradation of extracellular matrix (ECM) and is regulated by various factors among which TGF-b has a central role. The Treg cells are one of the most important factors in the remodeling process. TGF-b is also an essential factor in the remodeling process in the airways. It is responsible for attraction and induction of proliferation of fibroblasts, and it also causes upregulation of ECM synthesis. A recent study showed that TGF-b1 and 2 protein concentrations, TGF-b receptor I and III mRNA expression, and the numbers of activated pSmad 2-positive cells were significantly lower in patients with CRSwNP than control subjects. However, in patients with CRSsNP, TGF-b1 protein concentration, TGF-b receptor II and III mRNA expression, and the number of activated pSmad 2-positive cells were significantly higher than control subjects (Van Bruaene et al. 2009). Indeed, the upregulation of the TGF-b signaling pathway in patients with CRSsNP causes excessive collagen deposition associated with fibrosis, while its downregulation at the protein level in patients with CRSwNP causes edema formation and a lack of collagen production (Table 32.1).
Table 32.1
ARIA classification of allergic rhinitis
ARIA classification for allergic rhinitis | |
|---|---|
“Intermittent” rhinitis | “Persistent” rhinitis |
Symptoms are present: | Symptoms are present: |
≤4 days a week | ≥4 days a week |
or ≤ 4 consecutive weeks | and ≥4 consecutive weeks |
“Mild rhinitis” | “Moderate/severe” rhinitis |
None of following items are present: | ≥1 of following items are present: |
Sleep disturbance impairment of daily activities, leisure, and/or sport impairment of work or school work, troublesome symptoms | Sleep disturbance impairment of daily activities, leisure, and/or sport impairment of work or school work, troublesome symptoms |
Past investigations have focused on inflammatory differences, but recent information from studies comparing patients with CRSsNP and patients with CRSwNP showed that TGF-b proteins and their signaling might be suitable markers to distinguish between the different CRS subtypes.
32.5.4 Chronic Infectious Sinusitis
There is a significant loss of barrier and innate immune functions in all types of CS, making these patients highly prone to frequent and long-standing episodes of acute sinusitis. As a consequence, all forms of CS generally copresent with anaerobic bacteria, gram-negative organisms, Staphylococcus aureus, and other bacterial colonization in the sinuses. However, chronic infection (i.e., an episode of acute sinusitis persisting beyond 12 weeks despite antibacterial therapy) is less often the cause of CS, and, when present, the clinician should consider whether there is an underlying immune deficiency, HIV infection, immotile cilia syndrome, or cystic fibrosis. At the present time, investigation into the presence of allergic atopy or anatomic abnormalities is recommended for patients with CS because these factors might have a causative or modifying role on disease. The exact significance of these factors with relation to sinus inflammation is unknown, but their impairment might cause a decreased level of patient improvement (Rosenfeld et al. 2007; Table 32.2).
Table 32.2
Clinical manifestation of chronic sinusitis
1. Infectious | Idiopathic |
Associated with immune deficiency | |
2. Eosinophilic | AFS |
CHES | |
AERD | |
3. Noneosinophilic | Idiopathic |
Induced by anatomic tendency | |
Induced by Chronic rhinitis | |
4. Cystic fibrosis |
32.5.5 Noneosinophilic Sinusitis (NES)
Idiopathic noneosinophilic sinusitis (NES) is thought to result from chronic or recurrent blockage of the sinus ostia caused by anatomic predisposition like septal deviation viral rhinitis, allergic rhinitis, or other causes. As a consequence, these processes cause recurrent and prolonged bacterial infections, possibly in association with barotrauma of the sinus cavities and harm to the respiratory epithelium, ciliary destruction, prominent mucous gland, and goblet cell hyperplasia similar to bronchial epithelium in asthma disease, bacterial colonization, and biofilm formation (Payne et al. 2011). A mononuclear cell infiltrate with few neutrophils is observed in the inflammatory component of this form of sinusitis (19). If neutrophils are presented, it indicates recent infection, persistent infection, or CF. Prominent remodeling with dense deposition of collagen and other matrix proteins is characteristic for NES.
32.5.6 Molecular Basis of NES
Knowledge about the development of this disease is very limited, and as such, there are very few studies that have investigated whether there is a genetic component to NES. One study identified the plasminogen activator inhibitor 1 (PAI-1) gene as a possible candidate (de Alarcon et al. 2006). Subsequently, recent observations related to expression of PAI-1 and the thrombotic/fibrinolytic pathways further suggest a role for PAI-1 in CS (Shimizu et al. 2011). The 4G allele of PAI-1 is involved in the arrangement of fibrosis in asthmatic patients and especially in airway remodeling that leads to irreversible obstruction (Cho et al. 2001). In this study, overrepresentation of the 4G allele was observed in the NES group compared with the control group of subjects without sinus disease (0.53 vs. 0.45) (de Alarcon et al. 2006). In a recent study on subjects (excluding asthma, atopy, or aspirin intolerance and thus were more likely to have NES), it was found that patients with CS had an increased prevalence of a GG genotype at position 2,174 of the IL-6 promoter compared with a control group without sinus disease (odds ratio, 2.65) (Kosugi et al. 2009). There is an effect of IL-6 on the differentiation of naive CD4 T1 cells to TH17 lineage, so this result conforms with the finding of an increased TH17 signature in NE-NPs (Zhang et al. 2008). IL-6 is also important for plasma cell differentiation, and this function also is consistent with the previously discussed role for humoral immunity in patients with NES.
32.5.7 Chronic Hyperplastic Eosinophilic Sinusitis (CHES)
CHES is an inflammatory disease. It is characterized by intensive eosinophil accumulation in sinuses and also rarely accompanied with NP tissue. NPs generally associate with CF, AFS, and AERD. The presence of nasal polyposis (especially in association with asthma) has been recognized as suspicion for CHES diagnosis. But if we want to clearly diagnose CHES, histochemical staining of tissue for eosinophils or thorough determination of eosinophil-derived mediators such as major basic protein or eosinophil cationic protein is needed. The sinus tissue in patients with CHES presents a pronounced increase in cells that cause differentiation, survival, and activation of eosinophils that synthesize chemokines (e.g., CCL5, CCL11, and CCL24), cytokines (e.g., IL-5 and GM-CSF), and proinflammatory lipid mediators (e.g., cysteinyl leukotrienes [CysLTs]) (Perez-Novo et al. 2005). In addition, eosinophils are recruited and subsequently provide the essential growth factors for their own activation, proliferation, and survival (Hamilos et al. 1998). Thus, in contrast to NES, CHES behaves as an everlasting syndrome. As a consequence, it frequently does not respond well to surgery alone (Lee et al. 2010). The exact cause of CHES is not well understood. Some patients with CHES show allergic sensitization as determined by skin prick test or IgE immunoassay results. Aeroallergens usually do not reach healthy sinus cavities. When this disease involves blockage of the sinus ostia, it is more difficult for aeroallergens to gain access to the sinus (Gwaltney et al. 2000). Although aeroallergens do not reach the sinuses, their contact with nasal passage exacerbates sinus inflammation in these patients. Furthermore such nasal challenges increase eosinophil influx into the sinuses (Baroody et al. 2008). Since there is no direct access to the sinuses, some studies suggest that local and/or systemic lymphatic recirculation of inflammatory cells (such as eosinophils, eosinophil precursors (Denburg and Keith 2008), dendritic cells, and TH lymphocytes) between the nasal epithelium, nasal/sinus lymphatics, bone marrow, and sinus tissue may cause this disorder. Some research suggests that allergic IgE sensitization toward commensal fungi and bacteria colonies within the sinuses might be another disease mechanism (Ponikau et al. 1999). CHES shares a lot of histological and immunologic features with asthma and frequently associates with asthma, suggesting that CHES and asthma might include a similar idiopathic immune process, as airways, respectively (Braunstahl et al. 2001).
32.5.8 Molecular Basis of CHES
The close relation between CHES and NP formation has been shown in more than 30 studies addressing a possible genetic linkage. Several genes from the inflammatory pathway were found to relate/correlate with CHES, thus indicating a role for dysregulation of cytokine production in the pathogenesis of CHES. A few studies have replicated the relation between IL-1a and CS, similar to the relation between IL-1a and NPs. They found a greater risk related with the G allele at position 14,858 in exon 5 of the gene (Endam et al. 2010). Several studies have also found that the 2308 G-to-A polymorphism in the TNF-a gene is associated with CS and NPs (Batikhan et al. 2010); however, other studies were unable to replicate these findings (Endam et al. 2010).
A study in patients with asthma reported that a C-to-T polymorphism at position 2,590 of the IL-4 promoter may be connected to increased risk of asthma (Park et al. 2006). Two cohort studies have also shown this polymorphism to be associated with the development of NPs (Park et al. 2006). IL-1ra, IL-1 receptor-like 1, IL-33, and matrix metalloproteinase 9 have been determined as other relevant inflammatory genes. The MHC genetic region is very significant for the development of CHES. Various genes within this region have been especially associated with defects in antigen presentation as a cause for disease. A 2- to 5-fold increase in disease risk is associated with the HLA-DQA1* 0201 allele (Molnar-Gabor et al. 2000). Also, more than 12 other HLA alleles have been described in disease pathogenesis, but many of them have not been replicated in later studies.
32.5.9 Aspirin-Exacerbated Respiratory Disease (AERD)
AERD, also known as the Samter triad, is a condition characterized by NPs, asthma, and aspirin sensitivity (Samter and Beers 1968). Aspirin intolerance presents in 20 % of adult asthmatic patients. Incidence increases to 30 % if asthma is also associated with CS or nasal polyposis (Vally et al. 2002). The key features of this disorder are its association with severe and extensive pansinusitis and its propensity to develop de novo in adulthood. In patients who use aspirin regularly, if pansinusitis is present on CT scan examination, it may indicate aspirin sensitivity (Mascia et al. 2005). Nasal polyposis in AERD is aggressive with multiple polyps. Polypoid changes are characterized by rapid growth and frequent recurrence after surgery (Szczeklik and Sanak 2000). Despite the diffuse involvement of the sinuses with inflammatory tissue, the patient is asymptomatic, and compared with acute sinusitis or NES, patients seldom suffer from headaches or “sinus pressure.” However, anosmia is one of the consistent complaints and perhaps causes the greatest morbidity.
32.5.10 Molecular Basis of AERD
Increased leukotriene synthesis is typical for AERD, so genes related to leukotriene synthesis or response have been the center of attraction for many researchers. LTC4S is the most important rate-limiting enzyme in leukotriene synthesis. Some studies have found an A-to-C base substitution at position 2,444 of the LTC4S promoter, which increases LTC4S expression (Sanak et al. 2000). Some researchers subsequently identified a relationship between this base change and AERD (Kawagishi et al. 2002), while others have not (Van Sambeek et al. 2000). Thus, the significance of this substitution is still unclear. 5-lipoxygenase is the first enzyme in the leukotriene synthesis pathway. There are multiple variants of the tandem repeat GGGCGG within the promoter of the 5-lipoxygenase gene and Sp1, (In et al. 1997). One study found an increased risk for the development of AERD related with this gene (odds ratio, 5.0) (Choi et al. 2004). Additionally, another study showed that this polymorphism gives rise to the severity of airway hyperresponsiveness in patients with AERD (Kim et al. 2005). There are also other proteins in the leukotriene pathway associated with AERD such as CysLT1 and CysLT2 receptors. Finally, a few studies have reported a relation between the histocompatibility locus and the development of AERD, but these studies have not yet been replicated.
32.5.11 Allergic Fungal Sinusitis (AFS)
Sometimes, mold living commensally in the sinuses can cause activation of innate immune pathways and synergistically evoke robust TH2 lymphocyte and eosinophilic inflammatory responses. Initially, AFS was only attributed to Aspergillus species, but it is now known that many fungi species can be associated with AFS, including Cladosporium, Alternaria, Penicillium, Curvularia, and Bipolaris (Robson et al. 1989). The key feature of AFS is specific IgE sensitization, which is demonstrated by skin prick tests or serum immunoassays and measurement of increased total serum IgE concentrations. AFS generally develops in young, immunocompetent, and atopic subjects (Schubert and Goetz 1998). AFS has some features that distinguish it from other forms of eosinophilic sinusitis, such as its often being unilateral and limited to one or a few sinuses. Dense material fills and expands the sinuses and can typically be detected with CT scan (Mukherji et al. 1998). The mucous and inflammatory responses frequently occupy a space in the nasal cavity and lead to expansion into proximate tissue. This blocks the sinus ostia and subsequently causes bone absorption with resultant expansion into the orbits and cranium (Lydiatt et al. 1994).
32.5.12 Genetics of AFS
There is only one study that shows a genetic linkage with AFS. In a study of 74 subjects including 44 enrolled with AFS, a weakly significant association of disease was determined with the MHC class II allele HLA-DQB1*03 (Schubert et al. 2004), however, many subjects from the control group had at least one fungal species in the skin prick test.
32.6 Genetics of Cystic Fibrosis and Pathophysiology in Airways
32.6.1 Introduction
Cystic fibrosis (CF) is the most common lethal autosomal recessive genetic disorder, with a rate of approximately 1 in 2,500 live births among Caucasians. There are approximately 80,000 children and young adults with CF in the world. Genetic and nongenetic factors contributing to the disease and its variants have been widely investigated. Though the major gene responsible for the pathophysiology of CF is the cystic fibrosis transmembrane conductance regulator (CFTR) gene, recent research suggests that variations in other so-called modifier genes have an important influence on phenotypic differences in this disease. In recent years, multiple candidate modifier genes have been investigated, in particular, genes that are involved in the control of infection, immunity, and inflammation (Cutting 2005).
32.6.2 Rationale for Cystic Fibrosis
Subjects with CF typically present with disease in the lungs, sweat gland, pancreas, intestine (which is especially important during the newborn period), liver, and male reproductive tract (Welsh et al. 2001). CFTR controls chloride across the apical membranes of polarized epithelia (Anderson et al. 1991). Disruption in CFTR function inhibits the transport of sodium, chloride, and water across epithelial tissues, and so it causes insufficient hydration of mucous secretions in CF patients. Certain organs are eventually damaged from/by blockage in the luminal space and follows recurrent cycles of inflammation and fibrosis (Cutting 2007; Welsh et al. 2001). Many CF patients suffer from intestinal malabsorption and an abnormal nutritional status due to obstruction of the exocrine pancreas. The major cause of death in CF patient is complications arising from obstructive lung disease, a condition which occurs in approximately 90 % of patients (Cystic Fibrosis Foundation 2005).
32.6.3 Genetics and CF
Lung function measurements are notably different among CF patients with identical CFTR genotypes (e.g., F508del homozygotes) (Kerem et al. 1990). In fact, analysis of almost 40,000 patients in the CFTR2 database showed low correlation between CFTR mutations and FEV1. There are only a few mutations that cause a milder pancreatic phenotype (e.g., p. Arg455Glu) (De Braekeleer et al. 1997; Gan et al. 1995). In aggregate, these studies show that factors other than the CFTR genotype affect the progression of airway obstruction in CF.
Recurrence of complications in affected siblings at rates higher than in unrelated patients indicates a genetic effect, but care must be taken to account for the effect of similar environment for siblings. A more powerful approach is to compare monozygous (MZ) and dizygous (DZ) twin pairs for concordance for qualitative traits and correlation for quantitative traits. When MZ pairs show a stronger correlation than DZ pairs for a clinical feature, it shows that genetic factors may be responsible (Falconer 1965).
A higher correlation between composite measures of lung function and body mass index (BMI) was observed in 29 MZ versus 12 DZ twin pairs. This was the first twin-based assessment of the contribution of gene modifiers to CF disease severity and suggested genetic control of this trait (Mekus et al. 2000). Analysis of lung function and weight for height as independent measures did not show significant differences between the MZ and DZ twin pairs. Another comparison of 38 MZ pairs with six same-sex DZ pairs and 61 same-sex sibling pairs under 3 years of age demonstrated heritability of lung function based on FEV1 measurements ranging from 0.54 to 1.0 (Vanscoy et al. 2007). Variance analysis of 231 pairs of affected siblings showed an insignificantly higher estimate of heritability for the FEV1 measures (0.68–1.0) (Vanscoy et al. 2007). In aggregate, these studies show that genetic modifiers have an essential role in determining FEV1, a key measure of lung function, which is correlated with survival.
Collaco et al. recruited 134 MZ twins and 272 DZ twins and siblings when living together and after moving apart to estimate the relative effect of genetic and environmental factors on FEV1 among CF patients. Differences in lung function between MZ twin pairs while living together in the same house supplied an estimate of the effect of unique environmental and stochastic contributions. Changing the home environment to independent living was used to assess the effect of shared environment. The effect of genetic factors was estimated by comparing the similarities in lung function measures in MZ and DZ twin pairs when living together and subsequently when living apart. These methods showed that genetic and nongenetic factors had approximately equal effects on lung function. Analysis of 58 MZ twins and 568 DZ twins and siblings showed similar estimates for the genetic and nongenetic contributions to lung function variance (Stanke et al. 2011).
32.6.4 Modifier Genes in CF
Two independent studies with more than 500 patients combined showed that more than nine genes can be involved in modifying some feature of the CF phenotype. Several recent studies provide detailed lists of all the CF-related/modifier genes that have been studied thus far (Knowles 2006; Stanke et al. 2011).
These studies demonstrated the role of various modifier genes such as ADIPOR2, EDNRA, IFRD1, IL-8, MBL2, TCF7L2, MSRA, SERPINA1, and TGF-b1 in CF for pathologies of pulmonary function, liver disease, intestinal obstruction, diabetes, and infection. Three studies in CF patients showed earlier age of infection with Pseudomonas aeruginosa (Pa) to be related with mannose-binding lectin (MBL) deficiency genotypes. Lung disease severity, which is measured by FEV1 and infection status, is correlated with and two of them are changed by the age of the patient and by CFTR genotype. In aggregate, MBL2 genotype was found to be related to infection status more than the other variables (McDougal et al. 2010). Hence, deficits in MBL causes/can cause a predisposition to early infection with Pa, which leads to more severe lung disease than that observed in patients of the same age and CFTR genotype but who do not have MBL deficiency.
The Genetic Modifier Study (GMS), one of the largest CF genetic modifier studies to date, analyzed 808 F508del homozygotes drawn from the extremes of lung function (highest 30 percentile and lowest 30 percentile) and reported that alleles in the promoter (−509) and first exon (codon 10) of TGF-b1 are correlated with worse lung function (Drumm et al. 2005). This finding was studied in 498 patients with different CFTR genotypes and was separately confirmed when a haplotype composed of the opposite alleles at −509 and codon 10 was correlated with improved lung function (Bremer et al. 2008). Six studies including over 2,500 CF patients determined a relationship between TGF-b1 and CF lung function, while one study including 118 patients did not (Brazova et al. 2006) and another involving 171 patients (Arkwright et al. 2000) found a relation between worse lung function and the opposite alleles than those reported by Drumm et al. (2005) and Bremer et al. (2008).
Three SNPs in the highest ranking gene, the interferon-related developmental regulator 1 gene (IFRD1), were identified in the whole GMS sample and showed a relationship using transmission-based methods in the family-based CF Twin and Sibling Study (TSS) (Vanscoy et al. 2007). IFRD1 acts via transcriptional mechanisms to alter neutrophil function in response to bacterial infection, as demonstrated by cell- and mouse-based studies.
It was demonstrated that variants in the interleukin-8 (IL-8) gene correlated with lung function. This result supported the idea that modification of CF lung disease may be caused by altered neutrophil response to infection (Hillian et al. 2008). IL-8 is a mediator that has a role in neutrophil chemotaxis and is distinctly increased in the airway secretions of CF patients.
There are other mechanisms, which seemingly contribute to CF lung pathology as demonstrated by evidence that variants in the endothelin receptor type A (EDNRA) gene correlate with lung disease severity. Correlation between a variant in the 3’ untranslated region of EDNRA was identified in 709 F508del homozygous patients in the GMS study and replicated in three independent samples of CF patients. Also, alleles of the EDNRA variant are associated with differences in RNA transcript level, which indicates a possible functional role. Given that variation of EDNRA has been implicated in vasoconstrictive diseases as a result of effects on smooth muscle function, it was hypothesized that this gene may modulate CF lung disease by changing smooth muscle tone in the airways and vascular system (Darrah et al. 2010).
32.6.5 Cystic Fibrosis and Nasal Findings
Clinical manifestations in the upper airways (UAW) occur in almost 100 % of CF patients, appearing as recurrent sinusitis, rhinitis, and/or nasal polyposis (Cepero et al. 1987; Ramsey and Richardson 1992). The frontal sinuses seldom develop in these patients, perhaps because of the early occurring/earlier occurring disorder of sinusitis which hinders pneumatization (Ledesma-Medina et al. 1980). Sinusitis onset and nasal polyposis commonly occur between 5 and 14 years of age, with adult onset being unusual.
Most patients with CF (over 90 %) (Umetsu et al. 1990; Weber et al. 1999) develop chronic and recurring rhinosinusitis with or without nasal polyps. Modified mucus composition and viscoelasticity cause decreased mucociliary clearance and blockage in paranasal sinus drainage ostia, thereby promoting local inflammation, hypoxia, and increased carbon dioxide partial pressure. Mucosal edema generally develops after impaired ciliary function and bacterial colonization, usually by Staphylococcus aureus and Pseudomonas aeruginosa (Batsakis and El-Naggar 1996; Cimmino et al. 2003).
Franco et al. reported a relation between sinonasal symptoms and cystic fibrosis. They found most common symptoms like cough (45 %), oral breathing (44 %), sleep disorders (42 %), and nasal obstruction (37 %) in CF patients. Twenty-eight patients (28 %) had purulent nasal discharge, and 41 % had medial bulging of the nasal lateral wall (Franco et al. 2009). It is reported that nasosinusal involvement may worsen pulmonary disorder (Daniel 2006). Hence, otorhinolaryngologists should investigate these patients in more detail for signs of pulmonary diseases. A recent study in Brazil (Sakano et al. 2007) demonstrated more attention to the nasosinusal findings of CF patients, because CF is genetically very heterogeneous, with many types of mutations and a wide diversity in clinical presentations (Gentile and Isaacson 1996).
ΔF508 homozygosity was found more frequently in the patients undergoing sinus surgery (58 %) compared with a control population (48 %) (Moss and King 1995). Lastly, a study reported that ΔF508 homozygosity was associated with clinical severity of paranasal sinus diseases and with the presence of polyps on endoscopy in 113 patients (Jorissen et al. 1999).
32.6.6 CF and Nasal Polyposis
Nasal polyposis in CF patients was first described almost 50 years ago (Lurie 1959), but there is little known about its pathophysiology (Hulka 2000). The prevalence of nasal polyposis varies by population (Sakano et al. 2007). The incidence of nasal polyps has been observed in 6 to 48 % of cases (Shwachman et al. 1962) by the time cystic fibrosis is diagnosed. Nearly 4 % of patients already have symptomatic nasal polyposis when their diagnosis of CF is established, and it is expected that nearly 14 % of patients will undergo surgical intervention for their nasal polyp disease (Cimmino et al. 2003).
Weber et al. showed that nasal polyps were estimated in 39.1 % of CF patients and, interestingly, all of them were older than 6 years of age, presenting with recurrent pneumonia in 82.6 %, pancreatic insufficiency in 87 %, and malnutrition in 74 %. No correlation was seen between nasal polyps and sweat chlorine concentration, genotype, clinical signs of severity, and nasal symptoms. Nasal polyps regressed in seven patients treated with topical steroids, while six patients showed complete resolution (Weber and Ferrari 2008).
Some researchers reported that patients with nasal polyposis had better pulmonary function, however a higher rate of Pseudomonas aeruginosa colonization, more hospitalizations, and more prevalence of allergy to Aspergillus fumigatus than the comparison group. They found no statistically different genotype distribution between the group with polyposis and the control group. But they also emphasized that the prevalence of the compound heterozygous genotype is higher within the nasal polyposis group than within controls (Cimmino et al. 2003).
32.7 Role of Genetics in Nasal Polyposis
32.7.1 Introduction
The nasal polyp is one of the final manifestations of chronic inflammation. Nasal polyposis is a chronic inflammatory disorder of the upper respiratory tract that 1 to 4 % of the human population suffers from (Pawankar 2003). The lamina propria of nasal polyps usually presents great numbers of eosinophils and lymphocytes. In chronic inflammation, inflammatory cells produce neuropeptides, cytokines, and growth factors. These molecules lead to an extensive network of cellular interactions. In addition, resident structural cells can synthesize many of these molecules. Fibroblasts, epithelial cells, and endothelial cells help to organize the inflammatory process in nasal polyps (Liu CM et al. 2002).
Recently, it has been shown that there are proinflammatory cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-1b (IL-1b) in the epithelial and endothelial cells of nasal polyps. Also, cell adhesion molecules such as very late antigen-4 (VLA-4) have been found on the surface of eosinophils, while integrins such as vascular cell adhesion molecule-1 (VCAM-1) have been shown on the surface of the small venules of the nasal polyp. Lastly, the presence of chemokines such as regulated upon activation normal T cell expressed and secreted (RANTES), eotaxin, and IL-8 in the epithelium of the nasal polyps has been determined.
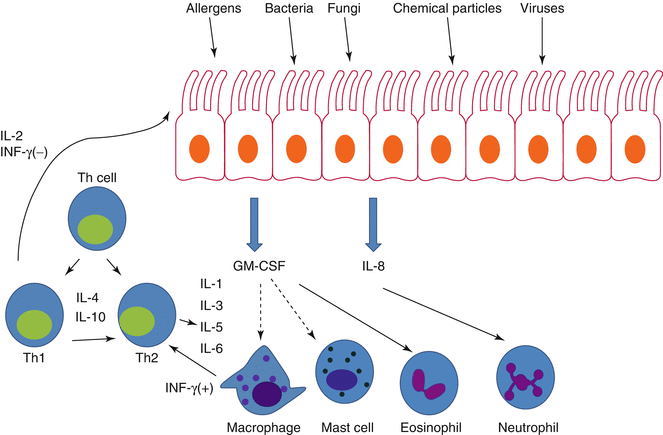
The nasal polyp tissue and the nasal mucosa have a sufficient collection of inflammatory molecules to combat efficiently against different agents such as allergens, bacteria, fungi, chemical particles, and viruses that come into the nose from the external environment. One of the most significant cells to offer an immune response may be the lymphocyte subpopulations. The percentages of TH1 lymphocytes (which produce IL-2 and interferon-α [ INF- α]) and TH2 lymphocytes (which produce IL-4 and IL-5 cytokines) in the nasal pharyngeal tonsillar lymphocytes and peripheral blood lymphocytes have been determined in patients with nasal polyposis (Bernstein et al. 2001). These same researchers have described the lymphocyte subpopulations and cytokines in nasal polyps (Bernstein et al. 2004).
32.7.2 Mucosal Irritation and the Role of Staphylococcal Exotoxin
As the nasal polyp symbolizes a final point in chronic inflammation, it is difficult to describe the initial events that trigger the inflammatory process in the lateral wall of the nose. Some substances, such as allergens, bacteria, viruses, air pollutants, and fungal elements, enter the submucosa of the lateral wall of the nose and damage the airway epithelium. These irritants lead to changes in some of the possible modifications of the respiratory epithelium that may take place after entrance of these particles. These changes include the following: first, the synthesis of inflammatory eicosanoids, which are potent cell activators and chemoattractants; second, proinflammatory cytokines such as TNF-α and IL-1, which have major effects on growth, differentiation, migration, and activation of inflammatory cells; and, third, specific cell adhesion molecules, which have an essential role in managing the inflammatory cell. Lastly, major histocompatibility class II antigens have a crucial role in antigen presentation to T cells (Salik et al. 1999) and are also responsible for consequent activation of T cells. Figure 32.2 shows the possible changes in respiratory epithelium after the entrance of bacteria, virus, allergens, and fungal elements.