Purpose
To investigate the features of corneal epithelial basement membrane dystrophy using spectral-domain optical coherence tomography (SD OCT) and to examine the reliability of SD OCT in distinguishing epithelial basement membrane dystrophy from the normal cornea.
Design
Diagnostic test study.
Methods
Forty-five individuals with epithelial basement membrane dystrophy and 45 age- and sex-matched controls with normal corneas were examined, and SD OCT scans of their corneas were performed. In vivo confocal microscopy was performed to confirm or rule out the diagnosis of epithelial basement membrane dystrophy. The structural corneal changes occurring in eyes with epithelial basement membrane dystrophy based on SD OCT findings were described.
Results
Epithelial abnormalities were observed in 86 of 87 eyes with epithelial basement membrane dystrophy (45 patients) on SD OCT scans. The 2 main features were the presence of an irregular and thickened epithelial basement membrane duplicating or insinuating into the corneal epithelium layer, or both, and the presence of hyperreflective dots. In some cases, we detected hyporeflective spaces between the corneal epithelial layer and the Bowman layer similar to a corneal epithelial detachment. This corneal epithelial detachment sometimes was associated with a cleavage with a stair-step appearance within the corneal epithelial layer. We found a perfect correlation between in vivo confocal microscopy and SD OCT findings in the diagnosis of epithelial basement membrane dystrophy (κ = 0.98).
Conclusions
SD OCT provides an accurate assessment of the structural changes occurring in eyes with epithelial basement membrane dystrophy. These changes, visible on SD OCT scans, are easily detectable and permit an accurate diagnosis, especially in patients with no biomicroscopically visible corneal changes.
Corneal epithelial basement membrane dystrophy (Online Mendelian Inheritance in Man 121820) —also known as Cogan microcystic dystrophy, map-dot-fingerprint dystrophy, or microcystic corneal dystrophy—is the most common of the anterior corneal dystrophies. It usually develops between the ages of 20 and 50 years and is characterized by bilateral, and frequently asymmetric, subepithelial fingerprint lines, geographic map-like lines, and epithelial microcysts (or dots) shown by slit-lamp examination. In some families, the condition seems to have an autosomal dominant method of inheritance. Most authors, however, consider this condition to be an age-dependent degeneration of the cornea. Although, as a general rule, epithelial basement membrane dystrophy is asymptomatic, approximately 10% of patients develop painful, recurrent epithelial erosions. This dystrophy is characterized, histologically, by an abnormal basement membrane that protrudes into the epithelium and the presence of intraepithelial microcysts.
Over the last decade, in vivo confocal microscopy has provided greater insight into the morphologic anomalies that occur in eyes with corneal epithelial basement membrane dystrophy, thereby improving our understanding of the disease. This technique has become a helpful tool for the diagnosis of corneal epithelial basement membrane dystrophy, especially when clinical features are atypical; however, the examination necessitates direct contact, which inadvertently may cause more damage to an already fragile eye. Recently, the development of spectral-domain (SD) technology has dramatically improved imaging speeds. Of the currently available SD devices, the RTVue (Optovue Inc, Fremont, California) is the only SD optical coherence tomography (OCT) system that has been approved by the Food and Drug Administration for both corneal and retinal imaging.
We performed a prospective study of patients with corneal epithelial basement membrane dystrophy seeking treatment at an ophthalmic unit in Paris. The main aim of the study was to investigate the SD OCT features of corneal epithelial basement membrane dystrophy. We also evaluated whether these SD OCT features permit eyes with corneal epithelial basement membrane dystrophy to be distinguished from normal eyes.
Methods
Ethics Approval
This study adhered to the tenets of the Declaration of Helsinki. All of the patients involved were informed that the study comprised several procedures (SD OCT examination and an in vivo confocal microscopy examination) and that their data would be recorded and be used in the study. All of the patients gave their informed consent to participate. The study was a prospective study, approved by the Ethics Committee of the French Society of Ophthalmology (IRB 00008855 Société Française d’Ophtalmologie IRB no. 1).
Patients
We performed a diagnostic test study using prospectively collected data from patients consecutively seeking treatment at a reference center for corneal dystrophy at the Quinze-Vingts National Ophthalmology Hospital (Paris, France) between January 2014 and May 2014. Ocular assessment was carried out according to the standard operative procedures of the unit. Exclusion criteria included any prior ocular surgery, trauma, contact lens wear, or associated corneal pathologic features.
Data Collection
All of the patients studied underwent a complete eye examination, including a history of recurrent erosion, visual acuity assessment, anterior segment slit-lamp biomicroscopy, and funduscopy. All of the patients were examined by 3 corneal specialists (L.L., V.B., and B.A.). For each patient, best spectacle-corrected visual acuity was measured using a conventional Snellen chart and slit-lamp photographs were obtained.
Patients who had typical geographic patches, subepithelial fingerprint lines, or dot-like opacities on slit-lamp examination were diagnosed with corneal epithelial basement membrane dystrophy. SD OCT imaging of the cornea was performed on all eyes, followed by an in vivo confocal microscopy examination.
Controls
To determine whether SD OCT was able to distinguish between normal and abnormal epithelial basement membrane, we conducted a comparative study that included normal patients for whom no ophthalmic symptoms or corneal anomalies were observed on slit-lamp biomicroscopy or during in vivo confocal microscopy examination. To obtain comparable groups, we selected 1 control for each patient studied who was matched to the study patient by age (±5 years) and gender. Most of these patients were working as volunteers at the hospital or were patients seeking treatment at our department who were eligible for refractive surgery. All matched patients were examined by the same 3 corneal specialists (L.L., V.B., and B.A.). All controls underwent SD OCT imaging of the cornea, followed by an in vivo confocal microscopy examination.
Anterior Segment Spectral-Domain Optical Coherence Tomography
A Fourier-domain OCT system (RTVue) with a corneal adaptor module was used. The system works at a 830-nm wavelength, with a scan speed of 26 000 axial scans per second. Depth resolution is 5 μm (full-width half-maximum) in tissue. For each patient (case and control), 10 high-resolution horizontal scans (with 0.5-mm intervals) and 10 high-resolution vertical scans (with 0.5-mm intervals) were made to evaluate all structural corneal anomalies. All SD OCT examinations were performed by the same corneal specialist examiner (E.B.), who was masked to the case or control status and the clinical features of the patients. All OCT scans then were analyzed by 2 corneal specialist observers (M.E. and O.S.). To assess the interobserver agreement, the 2 observers were not given access to the patients’ clinical and topographic data. To evaluate the intraobserver reliability, OCT scans were reanalyzed 2 weeks later by the same 2 observers (M.E. and O.S.).
In Vivo Laser Scanning Confocal Microscopy
In vivo confocal microscopy was performed using the Heidelberg Retina Tomograph 3/Rostock Cornea Module (Heidelberg Engineering GmbH, Heidelberg, Germany) confocal microscope. All examinations were performed by the same examiner (I.G.). The examiner was masked to the case or control status, clinical features, and SD OCT results of the patients studied. In vivo confocal microscopy was performed on all patients subsequent to the SD OCT examination. The images obtained from in vivo confocal microscopy consisted of 2-dimensional high-resolution optical sections covering a 400 × 400-μm area, with 1-μm transversal resolution and 4-μm longitudinal optical resolution. Several images of the different layers of the cornea were produced. This technique was used on each eye for 5 minutes, and no complications related to in vivo confocal microscopy evaluations were noted.
Statistical Analysis
Results are presented as mean ± standard deviation for continuous variables and as proportions (%) for categorical variables. The paired Student t test was used to compare continuous data between the patients and the matched controls. For binary outcomes, the stratified Cochran χ 2 test and Fisher exact test were used for intergroup comparisons of proportions when appropriate.
For the analysis of SD OCT results, interobserver and intraobserver agreements were assessed using a χ 2 test and the κ statistic, the latter permitting an assessment of the level of agreement that takes into account agreement occurring by chance. κ statistics were interpreted using a rating scale suggested by Landis and Koch: values ranging from 0 to 0.20 indicating slight agreement, 0.21 to 0.40 indicating fair agreement, 0.41 to 0.60 indicating moderate agreement, 0.61 to 0.80 indicating substantial agreement, and more than 0.80 indicating almost perfect agreement. Agreement between the SD OCT and the masked in vivo confocal microscopy techniques in the diagnosis of corneal epithelial basement membrane dystrophy also was assessed by the κ statistic. P values of .05 or less were considered statistically significant. Statistical analysis was carried out using SPSS for Windows version 20.0 (SPSS, Inc., Chicago, Illinois, USA).
Results
Clinical Evaluation of Patients with Corneal Epithelial Basement Membrane Dystrophy
Forty-five patients were identified for inclusion in this study, including 19 men and 26 women with a mean age of 48.05 ± 10.75 years (range, 23 to 67 years). Thirty-six patients (80%) had a history of recurrent ocular pain in one (9 patients) or in both (27 patients) eyes. Among the 9 patients who had no history of ocular pain, 4 patients (8.9%) sought treatment at the department as a result of blurred vision in one (2 patients) or in both (2 patients) eyes. The remaining 5 patients were asymptomatic, and the diagnosis of corneal epithelial basement membrane dystrophy was made, or suspected, by slit-lamp examination during routine eye examinations. Best spectacle-corrected visual acuity ranged between 20/20 and 20/100 (mean ± standard deviation, 20/23 ± 20/80). Patients with painful recurrent epithelial erosion initially were treated with a variety of medications, such as tear substitutes, antibiotic drops, or vitamin A ointment. At the time of examination, patients had received no treatment or had used only artificial tears. None of the patients had been treated with stromal puncture or phototherapeutic keratectomy.
In 79 eyes (88.9%) of 40 patients, corneal epithelial basement membrane abnormalities were diagnosed using slit-lamp examination: 15 eyes had a pattern of typical map-dot-fingerprint changes, 20 eyes had a map-dot pattern, 8 eyes had a map-fingerprint pattern, 7 eyes had a dot-fingerprint pattern, 14 eyes had only dot-like findings, 5 eyes had a map pattern, and 10 eyes had miscellaneous basement membrane abnormalities. For 8 eyes (9.2%), no corneal anomalies evocative of epithelial basement membrane dystrophy were revealed on slit-lamp examination, and the diagnosis of corneal epithelial basement membrane dystrophy was made only after performing the SD OCT and in vivo confocal microscopy examinations. After clinical, SD OCT, and in vivo confocal microscopy evaluations, 3 patients (6.7%) were diagnosed with corneal epithelial basement membrane dystrophy in 1 eye only. Dry eye superficial punctate keratitis was diagnosed using slit-lamp examination in 16 eyes.
Clinical Evaluation of the Control Participants
Forty-five age- and sex-matched controls were selected. These included 19 men and 26 women with a mean age of 45.05 ± 12.25 years (range, 22 to 60 years). Thirty controls were hospital workers, and the remaining 15 controls sought treatment at our department for presbyopia refractive surgery. At the time of examination, none of the controls had ocular symptoms or treatments, and none had a history of ocular trauma or ocular surgery. Best spectacle-corrected visual acuity for the control group ranged between 20/16 and 20/30 (mean ± standard deviation, 20/20 ± 20/100). No corneal anomalies were found during slit-lamp examination. The baseline characteristics of the 2 groups (cases vs controls) were not different (see Table 1 ).
| Cases | Controls | P Value | |
|---|---|---|---|
| Mean age (SD), y | 48.05 (10.75) | 45.05 (12.25) | 0.407 a |
| Gender, no. | |||
| Male | 19 | 19 | 1.000 b |
| Female | 26 | 26 | |
| Mean BSCVA (SD), Snellen | 20/23 (20/80) | 20/20 (20/100) | 0.738 a |
Spectral-Domain Optical Coherence Tomography Examination
In eyes with corneal epithelial basement membrane dystrophy, the epithelial basement membrane usually was irregular and thickened and appeared more hyperreflective than the epithelial basement membrane in a normal control eye ( Figures 1 and 2 ). This thickened epithelial basement membrane was sometimes very compromised with the appearance of small hyperreflective elevations associated with undulation and elevation of the corneal epithelium ( Figure 2 , Middle and Bottom). This thickening frequently was associated with an area of duplication of the epithelial basement membrane and a protrusion of the basement membrane into the corneal epithelium layer ( Figure 3 ). These insinuations of the epithelial basement membrane into the corneal epithelium layer usually corresponded to the map-like or the fingerprint lesions observed during slit-lamp examination.
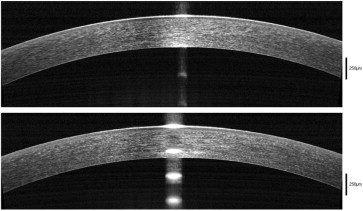
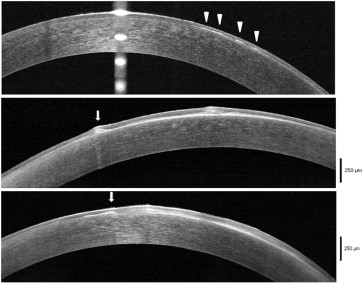
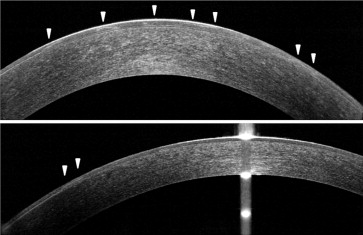
Another important feature observed in SD OCT scans was the presence of hyperreflective dots, with or without posterior shadow cone, in the middle of the corneal epithelium layer ( Figure 4 ), corresponding to the dots (also known as epithelial cysts) observed via slit-lamp examination. In some cases, the hyperreflective dots were more superficial ( Figure 5 ). In cases where an abnormal epithelial basement membrane protrusion was found, the dots were always beneath the abnormal epithelial basement membrane ( Figure 6 ).
In some eyes, we observed localized or extensive separations between the corneal epithelial layer and Bowman layer, with a hyporeflective space between the 2 layers ( Figure 7 ). These corneal epithelial detachments were separated by areas of normal adhesion between the corneal epithelium and Bowman layer ( Figure 8 ). The corneal epithelial detachments noted by SD OCT scan corresponded to the map-like lesions observed through slit-lamp examination. The patients with these abnormalities had a history of recurrent corneal erosion. In 2 patients, this separation between the corneal epithelial layer and Bowman layer continued as a cleavage within the corneal epithelial layer with a stair-step aspect ( Figure 9 ). On slit-lamp examination, these patients had fingerprint-like lesions in both eyes.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


