Facial Nerve Injury and Decompression
Robert A. Sofferman
ANATOMY OF THE FACIAL NERVE
Temporal bone fractures following blunt trauma are the most common cause of facial nerve injury, with more than half caused by vehicular accidents (1). Other injuries related to sporting activities and assaults can similarly affect the temporal bone, especially falls that directly injure the occiput and transmit tremendous force to the skull base. Penetrating injury from weapons or missiles produce high-frequency fragmentation of the fallopian canal, mastoid air cells, and medial temporal bone, resulting in facial nerve injury and often transection of nerve integrity. Habercamp (2) has demonstrated that 75% of patients with gunshot wounds to the temporal bone have immediate-onset facial paralysis. Iatrogenic injury to the facial nerve by routine otologic surgery or during the process of neurosurgical procedures involving instrumentation along the floor of the middle cranial fossa may become apparent to the surgeon for the first time only in the recovery room as an unexpected complication. Each of these conditions has its own special anatomic, pathophysiologic, and management considerations. Understanding the comprehensive anatomy and topognostic information, electrophysiologic assessments, and basic science and clinical correlates allows a management schema that is somewhat predictable for each of these broad areas.
The facial nerve has the longest intraosseous course of any motor nerve in the body. Injury to the neurovascular supply of the facial nerve and intra- or extra-sheath hematoma within confined spaces may explain the special anatomic reasons for the development of facial paralysis in nontransection injury. Although the geniculate ganglion and proximal segments are amply supplied by branches of the labyrinthine artery and superficial petrosal branch of the middle meningeal artery, the vascular supply is essentially end-arterial with limited collateralization (3). The narrowest osseous path of the facial nerve is the labyrinthine segment, also the site of the most common physiologic impairment of facial nerve function in temporal bone trauma. These anatomic details might be among the most important factors responsible for the evolution of facial paralysis in the trauma condition under discussion (4).
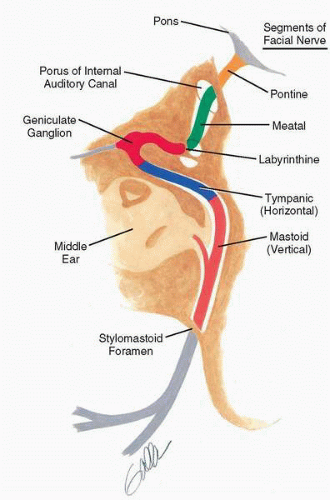
The facial nerve originates at the facial nucleus within the medulla, courses medially around the abducens nucleus in the pons, and exits the brainstem at the cerebellopontine angle (Fig. 36.1). For the purpose of surgical discussion, five anatomic segments of the facial nerve can be identified (3) (Fig. 36.2):
The pontine segment, between the brainstem and porus, measures 23 to 24 mm in length. At this point the facial nerve is anterior to the cochleovestibular nerve. The special sensory and visceral efferent components of the facial nerve pass in a separate bundle adjacent to the main motor trunk as the nervus intermedius.
The meatal segment, within the internal auditory canal, is 7 to 8 mm in length. The facial nerve passes superior to the falciform crest in the lateral aspect of the canal and is separated from the superior vestibular nerve by a vertical crest of bone (Bill’s bar).
The labyrinthine segment, between the meatal segment and geniculate ganglion, is 4 mm in length. The osseous canal surrounding the facial nerve is narrowest at the most proximal portion of the labyrinthine segment. This segment passes anterolaterally, paralleling the axis of the arcuate eminence of the superior semicircular canal, and passes superior and in proximity to the basal turn of the cochlea. The geniculate ganglion is triangular and averages 1.09 mm in length.
The tympanic or horizontal segment, between the geniculate ganglion and second genu, is 12 to 13 mm in length. The proximal edge of the geniculate ganglion is 5 mm anterosuperior to the posterior edge of the processus cochleariformis. The facial nerve passes superior to the oval window niche, a region where in approximately 55% of cases it is dehiscent (6).
The mastoid or vertical segment, between the second genu and stylomastoid foramen, measures 15 to 20 mm in length. At the second genu, the semicircular canal lies 0.5 mm posterosuperior to the facial nerve. The digastric ridge is a useful landmark just posterior to the stylomastoid foramen.
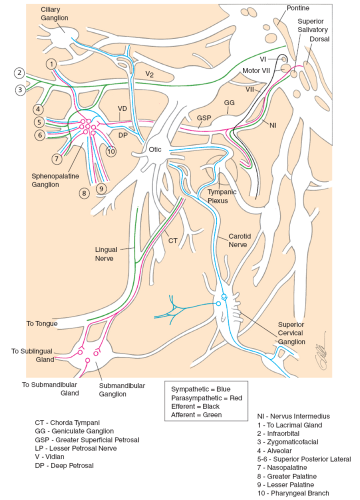 FIG. 36.1 Schematic diagram of the facial nerve (5). The motor division is shown in blue, the sensory division in black, and the parasympathetic pathway in red. (Reprinted with permission from Janfaza P, Nadol JB Jr, Galla RJ, et al. Surgical anatomy of the head and neck. Philadelphia: Lippincott Williams & Wilkins, 2001.) |
BLOOD SUPPLY OF THE FACIAL NERVE
The principal blood supply of the facial nerve originates from two opposite ends of the fallopian canal (2). The superficial petrosal artery, arising from the middle meningeal artery, enters the facial hiatus to supply the geniculate ganglion and horizontal and vertical segments of the facial nerve. The vessel anastomoses with the stylomastoid artery at the level of the oval window. The stylomastoid artery arises from the posterior auricular artery and passes through the stylomastoid foramen to supply portions of the mastoid cavity, tympanic membrane, and middle ear in addition to the facial nerve. There are anastomoses between the stylomastoid artery and posterior meningeal artery in the region of intersection of Arnold’s and facial nerves. Despite the anastomotic blood supply, the principal arterial supply enters from each end of the facial canal, with resultant potential for ischemia in the intervening segments. Ogawa and Sando (7) have described significant variations in blood supply at the various segments of the facial nerve. Based on cross-sectional histology, the labyrinthine segment seems to have a much poorer blood supply than either the horizontal or vertical segments. Single or multiple large veins accompany the facial nerve within the epineural sheath and often represent more than 50% of the cross-sectional area of the fallopian canal. These veins can bleed substantially during incision of the sheath.
MICROANATOMY OF THE FACIAL NERVE
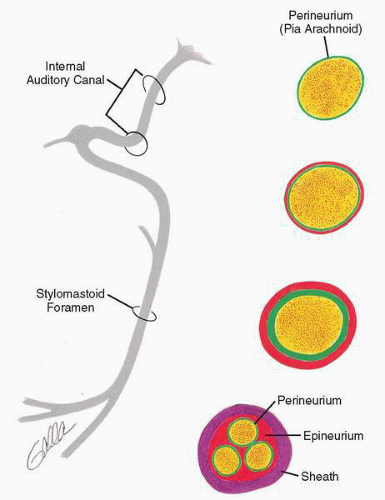
The microanatomy of the facial nerve is important to an understanding of facial nerve dysfunction and recovery (8,9). The motor division of the facial nerve consists of approximately 7,000 motor axons with cell bodies located in the medullary brainstem (10). The connective tissue component of the nerve consists of endoneurium, perineurium, epineurium, and nerve sheath (Fig. 36.3). The endoneurium consists of longitudinally oriented fibrous tissue around and between individual axons. The perineurium consists of parallel, circumferential, tightly arranged fibrous tissue around individual fascicles of the nerve. The epineurium
consists of loose connective tissue between and around fascicles and is continuous with the more dense sheath. The arteries, veins, and lymphatics lie just within the epineural layer. Arteries and venules lie between fascicles, whereas capillaries are found within the nerve sheath. This nomenclature is confusing, particularly for the facial nerve, which varies considerably in microarchitecture from segment to segment. For example, the facial nerve is monofascicular within its intratemporal course and develops a typical fascicular pattern as it emerges from the stylomastoid foramen (Fig. 36.3). The perineurium is extremely thin in the proximal intratemporal course and becomes thicker more distally.
consists of loose connective tissue between and around fascicles and is continuous with the more dense sheath. The arteries, veins, and lymphatics lie just within the epineural layer. Arteries and venules lie between fascicles, whereas capillaries are found within the nerve sheath. This nomenclature is confusing, particularly for the facial nerve, which varies considerably in microarchitecture from segment to segment. For example, the facial nerve is monofascicular within its intratemporal course and develops a typical fascicular pattern as it emerges from the stylomastoid foramen (Fig. 36.3). The perineurium is extremely thin in the proximal intratemporal course and becomes thicker more distally.
PATHOPHYSIOLOGY OF INJURY
In part, mechanisms of facial nerve injury may be ascribed to special anatomic considerations. Closed head trauma produces a deceleration injury, and some authorities have suggested that tethered neural structures are the most likely to be impaired during sudden deceleration (11). Two sites of consideration are the perigenicular region at the greater superficial petrosal nerve and the region of the facial nerve distal to the pyramidal eminence. The shearing forces produce intraneural contusion, edema, hemorrhage, and resultant ischemia.
In a cat experimental model, facial nerve transection at the stylomastoid foramen did not produce complete loss of distal electrical excitability until 72 hours had elapsed (12). The histopathology of facial nerve injury has been examined in a series of 12 patients with immediate traumatic facial palsy and eventual surgery for failed recovery 4 to 25 months after injury (13). Facial nerve biopsies revealed substantial axonal regeneration coexisting with other degenerating neurons. Axonal regeneration distal to the site of injury was blocked by fibrosis in the distal labyrinthine or proximal tympanic segments of the facial nerve in all cases. Others have theorized that intraneural fibrosis is a result of intraneural hematoma (14). Failed cases of nerve repair may be a result of suture line tension and subsequent fibrosis when primary anastomosis is attempted, arguing perhaps in favor of cable grafts for intralabyrinthine injury (15).
It is instructive to examine just what happens to patients with untreated traumatic facial paralysis and spontaneous complete or partial recovery. Adegbite et al. (16) reviewed 26 such patients who showed varying degrees of paralysis. Of patients with partial paralysis, 92.5% showed some evident recovery by 5 months, but only 10% of those with complete paralysis showed any recovery at this early time. Thus the course for recovery is more rapid for those with incomplete paralysis. Fisch (11) proposed that electroneurographic (ENoG) data in severe facial nerve injury in temporal bone trauma would be similar to that shown in his study on facial nerves with tumor and planned resection of the nerve. His analysis and that of Lieberherr et al. (17) suggest that the following ENoG results predict a favorable functional outcome: less than 90% degeneration for 6 days or no progression to greater than 95% degeneration within 14 days of injury. Thus previous concern for nerve compromise greater than 90% also requires examination of time relationships. Similarly, Gantz et al. (18) have examined the natural history of facial nerve injury in the viral insult of Bell’s palsy. Subjects who did not reach 90% degeneration on ENoG within 14 days of onset of complete paralysis all returned to House-Brackmann grade I (N = 48) or II (N = 6) by 7 months. Patients with greater than 90% degeneration on ENoG and no motor unit potentials on evoked electromyography (EMG) who elected to pursue conservative therapy had a 58% chance of poor outcome at 7 months (House-Brackmann grade III or IV [N = 19]). A similar group of patients who elected comprehensive middle fossa facial nerve decompression developed favorable recovery in 91% of cases (House-Brackmann grade I [N = 14] or II [N = 17]). However, the timing of decompression was critical: Patients having surgery within 2 weeks of onset of facial paralysis did well, but those submitted to surgery after 2 weeks had poorer results, similar to that of controls.
There may be some experimental validation for these clinical observations. Binns (12) produced a stretch injury at the second genu in cats and observed the expected facial paralysis and recovery at 6 to 8 weeks without treatment. Most animals subjected to facial nerve decompression of the second genu and vertical segment did not develop neural degeneration as long as surgery was performed within 48 hours. Decompression at 72 hours did not prevent neural degeneration. A separate study of compression injury in cats demonstrated clinical recovery if surgical decompression was performed within 12 days (19). Decompression between 14 and 21 days did not prevent synkinesis and reduction of electrical potentials.
ETIOLOGY OF TRAUMATIC FACIAL NERVE INJURY
Temporal Bone Fractures
Longitudinal Fracture
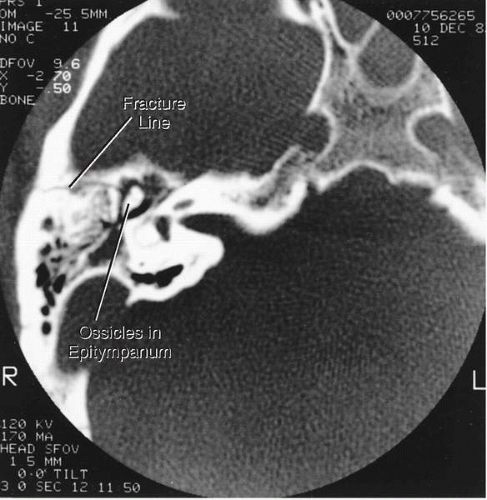
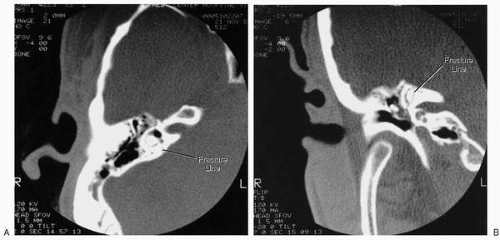
Seventy percent to 90% of temporal bone fractures are longitudinal as a result of a blow to the temporal or parietal skull, producing a fracture line that follows the long axis of the petrous pyramid (Figs. 36.4 and 36.5). The fracture crosses the floor of the middle cranial fossa near the foramen spinosum and courses anterior to the osseous labyrinth, injuring the facial nerve in 10% to 20% of cases at or near its labyrinthine segment. Fisch (11) has shown that 64% of longitudinal fractures involve the labyrinthine segment. The ossicular chain can be disrupted by fracture through the epitympanum, producing a conductive hearing loss. High-frequency sensorineural hearing loss occurs in approximately 20% of cases (20), presumably due to labyrinthine concussion. The fracture may pass through the external auditory canal, causing laceration of the skin and tympanic membrane. Bleeding and associated cerebrospinal fluid otorrhea
are common. Hasso and Ledington (21) have separated longitudinal fractures into anterior and posterior variants based on coronal computed tomographic (CT) scans. Posterior longitudinal fractures pass through the parietal skull and extend into the mastoid air cells, whereas anterior longitudinal fractures pass through the posterior and superior external auditory canal and tegmen. Both types may involve the eustachian tube, foramen lacerum, jugular canal, or carotid canal. Although facial paralysis develops in only 10% to 20% of longitudinal fractures, it is more common in well-pneumatized temporal bones, perhaps due to penetration of the nerve trunk by splinters of bone. Alternately, fixation of the facial nerve as it emerges from the internal auditory canal by the greater superficial petrosal nerve may predispose to traction injury. This may be associated with intraneural hematoma, leading to cicatricial obstruction of the fallopian canal. In a histologic study of a longitudinal fracture of the temporal bone occurring 1½ years prior to death, Eby et al. (22) found reactive osteitis, exophytic new bone formation, and narrowing of the labyrinthine segment of the fallopian canal. Longitudinal fractures are more often associated with delayed onset of facial paralysis, which carries a good prognosis and often does not require surgical intervention.
are common. Hasso and Ledington (21) have separated longitudinal fractures into anterior and posterior variants based on coronal computed tomographic (CT) scans. Posterior longitudinal fractures pass through the parietal skull and extend into the mastoid air cells, whereas anterior longitudinal fractures pass through the posterior and superior external auditory canal and tegmen. Both types may involve the eustachian tube, foramen lacerum, jugular canal, or carotid canal. Although facial paralysis develops in only 10% to 20% of longitudinal fractures, it is more common in well-pneumatized temporal bones, perhaps due to penetration of the nerve trunk by splinters of bone. Alternately, fixation of the facial nerve as it emerges from the internal auditory canal by the greater superficial petrosal nerve may predispose to traction injury. This may be associated with intraneural hematoma, leading to cicatricial obstruction of the fallopian canal. In a histologic study of a longitudinal fracture of the temporal bone occurring 1½ years prior to death, Eby et al. (22) found reactive osteitis, exophytic new bone formation, and narrowing of the labyrinthine segment of the fallopian canal. Longitudinal fractures are more often associated with delayed onset of facial paralysis, which carries a good prognosis and often does not require surgical intervention.
Transverse Fractures
Transverse fractures pass in a plane perpendicular to the long axis of the temporal bone (21) between the foramen magnum and middle fossa floor, traversing the osseous labyrinth or internal auditory canal (23) (Figs. 36.4 and 36.6). As a result, profound sensorineural hearing loss and vertigo are common, and in 50% of cases there is an immediate onset of facial paralysis. The site of facial nerve injury is the labyrinthine segment in 90% and meatal segment in 10% of cases (11). Occasionally, the fracture may extend to the jugular fossa or carotid canal, the latter resulting in a unilateral hemispheric infarction. Axial CT scans best demonstrate transverse fractures with associated injury of the temporomandibular joint, mandibular condyle, and skull base.
Complex Fractures
Severe trauma to the head may produce a combination of longitudinal and transverse fractures that are often severely comminuted and dislocated. In addition to the clinical sequelae described, cerebrospinal fluid otorrhea, rhinorrhea, and brain herniation may be additional complications. Gunshot wounds usually produce complex fractures with unpredictable injuries to neurovascular structures. Carotid arteriography is usually indicated in complex penetrating temporal bone injuries.
Facial Nerve Injuries in Children
Birth Trauma
The earliest recognizable facial nerve injury occurs with birth trauma (24). The deficit may be difficult to recognize except with facial asymmetry during crying. Facial paralysis may not be identified early due to the excellent facial tone present in children. The most common cause of facial nerve injury in the newborn is traumatic forceps delivery. In the infantile mastoid, the facial nerve is vulnerable at the stylomastoid foramen, lying just beneath the soft tissues of the upper neck and unprotected by overlying bone. Other causes of injury include neural compression by the maternal sacral promontory or by the fetal shoulder. The risk of facial paralysis increases in large primiparous infants and multiple fetuses. Neuropraxia can be distinguished from complete avulsion by electrophysiologic studies. Agenesis of the facial nerve can be excluded if electrical stimulation can be elicited at birth. If electrical excitability is lost after 72 hours, severe injury or avulsion at the stylomastoid foramen should be assumed, warranting surgical exploration (25).
Facial nerve injury secondary to temporal bone fracture is less common in children than in adults. In two series of temporal bone fractures, facial paralysis occurred in 10% to 20% of children, well below the incidence in adults (24) (Table 36.1). In a series of 26 children with traumatic facial nerve injuries, 61% of those requiring operative intervention recovered satisfactory facial function. Unsatisfactory results in this series were attributed to a delay of 4 or more weeks in therapeutic intervention.
TABLE 36.1 Incidence of facial paralysis in fractures of temporal bone
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|
|---|