Purpose
To evaluate the fine cytopathologic features and immunohistochemistry of eyelid sebaceous carcinoma.
Design
Retrospective clinicopathologic study.
Methods
Clinical records and microscopic glass slides of 12 patients diagnosed with sebaceous carcinoma were reviewed. Paraffin-embedded tissue recuts were immunoreacted for epithelial membrane antigen (EMA), Ber-EP4, p53, Ki-67, and adipophilin for cytoplasmic lipid. Invasive growth and intraepithelial spread were analyzed separately. Cytoplasmic and nuclear characteristics were correlated with the results of the immunohistochemical profiling.
Results
Five patients experienced recurrences, but no metastases or tumor-related deaths were discovered. The nuclei in 11 invasive tumor cells were typically round with finely divided, granular, or smudgy chromatin without prominent margination at the nuclear membrane; they exhibited small punctate nucleoli. Positivity for EMA (both diffuse and focal), p53 (72% of nuclei), and Ki-67 (45% proliferation index) was demonstrated. Adipophilin positivity in vesicular and granular forms was detected in paraffin sections in all invasive tumors, most prominently in moderately differentiated and well-differentiated lesions. Among 9 cases exhibiting intraepithelial extensions, 6 showed mostly granular positivity and 3 vesicular positivity. p53 identified residual atypical intraepithelial cells when conjunctival epithelial sloughing occurred.
Conclusions
Immunohistochemistry can make significant contributions to the diagnosis of sebaceous carcinoma. p53 and vesicular granular adipophilin positivity were highly reliable in supplementing the routine microscopic diagnosis of infiltrative tumors and both can be used in paraffin sections, thereby obviating cumbersome oil red O staining of frozen sections. The cells found in intraepithelial spread were strongly EMA and p53 positive, with more granular than vesicular adipophilin positivity.
In the largest published pathologic series from Europe of 5504 eyelid lesions excised between 1989 and 2007, sebaceous carcinoma accounted for only 29 examples (0.5% of total series) of all benign and malignant epithelial conditions (excluded from the series were 3340 chalazia). Among 6000 eyelid cases in the pathologic files of a major American eye institute, 40 cases (0.7%) were sebaceous carcinomas. Sebaceous carcinoma is nonetheless the second most common eyelid malignancy (5% of malignancies in this site), after basal cell and ahead of epidermoid (squamous) cell carcinomas, in the United States. Because of the generous number of sebaceous glands in the eyelids (intratarsal meibomian glands, Zeis glands attached to the cilia, and also those present in the caruncle and the brow), the periocular region is the most frequent single site for this malignancy throughout the entire integument (40% of all sebaceous carcinomas). Sebaceous carcinoma has also been discovered to occur as a primary lacrimal gland neoplasm. Delays in the correct diagnosis of sebaceous carcinoma can lead to recurrences, metastases, and ultimately death. Progressive education about this disease, leading to correct diagnosis and prompt, appropriate surgery, has reduced the death rate from 50% of cases in early studies to around 2%-10% in more recent ones. Tumors removed before 1970 in one series had a death rate of 24%, which fell to zero thereafter, probably because of heightened awareness as the result of a seminal publication in 1968. The morbidity of multiple surgeries attendant upon recurrences owing to incomplete eradication, however, still persists.
This investigation is aimed at devising an approach to further improve expeditious diagnostic accuracy and to determine the full extent of local tumor spread in the conjunctival epithelium. We have studied 12 patients with eyelid sebaceous carcinomas by concentrating on the fine details of cytomorphology and the use of 4 immunohistochemical markers: epithelial membrane antigen (EMA), p53, Ki-67, and adipophilin. Early and subsequent attempts to employ cytokeratin analysis have not caught on, in part possibly owing to unfamiliarity with these markers and to the ability of keratinocytes to modulate their cytokeratin expression in various diseases. Some authorities have concluded that other markers are more reliable than cytokeratins. Additional papers on eyelid tumors have contradicted each other with respect to the differential value and staining profiles of other immunohistochemical markers for sebaceous carcinoma vs mimicking epithelial cancers. For example, epithelial membrane antigen has been claimed to be negative in squamous cell carcinoma, while sebaceous carcinoma has been asserted to be Ber-EP4 positive ; these erroneous “findings” have also been carried over into major textbooks. In fact, it is now accepted that Ber-EP4 is negative in sebaceous carcinoma and EMA is negative in basal cell carcinoma, but positive in squamous cell carcinoma. That “primary” conjunctival epithelial sebaceous carcinoma has been alleged to be p53 negative while intraepithelial spread associated with infiltrating disease is p53 positive is an intriguing finding that has never been reassessed. It may have been the result of confusion with primary clear cell epidermoid carcinoma of the palpebral conjunctiva.
The results of adipophilin staining, which is capable of identifying intracellular lipid in formalin-fixed, paraffin-embedded sections, has not yet been reported in the ophthalmic literature in connection with eyelid sebaceous carcinoma. If proven to be helpful, its use would obviate oil red O staining of fresh tissue, which is frequently not performed because of failure to suspect a sebaceous tumor at the time of surgery. Furthermore, identification of oil red O positivity in conjunctival biopsy specimens for the presence of intraepithelial spread is notoriously fraught with pitfalls. The differential diagnostic specificity of the 2 morphologic forms of cytoplasmic adipophilin positivity (vesicular vs granular) is to an extent unsettled. We have attempted to resolve some of the foregoing immunohistochemical discrepancies to enhance the early and accurate diagnosis of eyelid sebaceous carcinoma and thereby facilitate optimal surgical therapy. We recognize, however, that reconciling discrepancies in contemporary immunohistochemical studies in regard to eyelid sebaceous carcinoma will continue to unfold as new data are brought forward.
Methods
This clinicopathologic retrospective study was conducted under the auspices of the Massachusetts Eye and Ear Infirmary’s Institutional Review Board (Protocol #13-056H), in compliance with the rules and regulations of the Health Insurance Portability and Accountability Act and all applicable federal and state laws, and in adherence to the tenets of the Declaration of Helsinki. After searching through the records of the David G. Cogan Laboratory of Ophthalmic Pathology at the Massachusetts Eye and Ear Infirmary from 2007-2013, 12 cases were considered acceptable to be selected for this study. This selection was based on histopathologic features displayed in formalin-fixed, paraffin-embedded, and hematoxylin-eosin-stained sections on glass slides. An essential criterion for inclusion was the availability of a paraffin block for further immunohistochemical studies derived from at least 1 surgery in the course of the disease that often included recurrences. The 12 patients had undergone a total of 30 surgical procedures. The grand total of separate specimens obtained and reviewed from all surgeries was 119 (including conjunctival map biopsies), of which 44 contained histopathologic evidence of either intraepithelial or infiltrative growth, or both. Patients’ medical records and clinical photographs were also reexamined in search of possible correlations.
Immunohistochemical staining was performed on 25 out of a total of 44 histopathologically tumor-positive specimens with available paraffin blocks on file. These were considered the most representative examples of the salient and often duplicative features; this restriction therefore reduced redundancy and cost. The results of multiple positivities of the same feature were aggregated for a single summary notation in Table 2 for each case. The studies were conducted in the Diagnostic Immunopathology Laboratory of the Massachusetts General Hospital. Immunostaining was done using the Leica Bond III (Leica Microsystems, Bannockburn, Illinois, USA) with appropriate controls to validate antibody quality. The following probes were used: epithelial membrane antigen (mouse monoclonal; Leica Biosystems, Newcastle, UK; prediluted); p53 (mouse monoclonal; Leica Biosystems; prediluted) for nuclear protein accumulation attributable to mutated tumor suppressor gene; adipophilin (clone AP125; Fitzgerald Industries Intl, Acton, Massachusetts, USA; 1:75) for cytoplasmic lipid; Ki-67 (mouse monoclonal; Dako, Carpinteria, CA; 1:200) for nuclear DNA replication; and Ber-EP4 (mouse monoclonal; Dako; 1:50) for basal cell carcinoma cytoplasm, and pancytokeratin (AE1/AE3: Leica Biosystems; 1:1000 and Cam 5.2: BD Biosciences, Franklin Lakes, New Jersey, USA; 1:100). Endogenous peroxidase activity was blocked by H 2 O 2 before antibody incubation. The chromogen diaminobenzidine was used and the tissues were counterstained with hematoxylin. Nuclear staining was recorded as percent positivity for p53 and Ki-67 (proliferation index), whereas cytoplasmic or cell membrane staining was noted to be positive or negative for EMA and Ber-EP4, respectively. Positive cytoplasmic staining for adipophilin was scored as 3+ (>50% of cells), 2+ (26%-50%), 1+ (6%-25%), and negative (≤5%).
| Patient # | Age/Sex | Laterality/Location | Symptoms at Initial Presentation | Total # of Surgeries a (n = 30) | Total # of Specimens b (n = 119) | Total # of Tumor-Positive Specimens (n = 44) | # of Recurrences | Length of Follow-up (Months) | Outcome |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 90/F | Right upper eyelid | Diffuse eyelid thickening | 4 | 8 | 7 | 2 | 134 | Alive without disease |
| 2 | 63/M | Left lower eyelid | Discrete eyelid mass, eyelid margin crusting and erythema | 2 | 5 | 3 | 1 | 72 | Alive without disease |
| 3 | 74/F | Left upper eyelid | Ulcerated lesion with crusting, madarosis | 2 | 9 | 7 | 1 | 53 | Alive without disease |
| 4 | 78/F | Right lower eyelid | Discrete eyelid mass, recurrent “chalazion,” surface crusting | 2 | 14 | 2 | None | 44 | Alive without disease |
| 5 | 94/M | Right upper eyelid | Discrete eyelid mass | 2 | 2 | 2 | 1 | 36 | Died of other causes unrelated to sebaceous CA |
| 6 | 70/M | Left upper eyelid | Discrete eyelid mass | 4 | 5 | 5 | 2 | 32 | Alive without disease |
| 7 | 70/F | Left lower eyelid | Diffuse eyelid thickening, conjunctival hyperemia | 2 | 5 | 2 | None | 18 | Alive without disease |
| 8 | 90/F | Left upper eyelid | Discrete eyelid mass with surface crusting | 1 | 1 | 1 | None | 14 | Died of other causes unrelated to sebaceous CA |
| 9 | 91/M | Left lower eyelid | Discrete eyelid mass with papillary projections of palpebral conjunctiva | 3 | 21 | 2 | None | 6 | Alive without disease |
| 10 | 88/F | Left upper eyelid | Indistinct mass in palpebral conjunctiva | 2 | 11 | 6 | None | 5 | Alive without disease |
| 11 | 49/F | Right lower eyelid | Diffuse eyelid thickening, madarosis, conjunctival hyperemia | 3 | 21 | 1 | None | 4 | Alive without disease |
| 12 | 72/M | Left upper eyelid | Discrete eyelid mass, irregular subconjunctival component, recurrent “chalazion,” history of colonic carcinoma | 3 | 17 | 6 | None | 3 | Alive without disease |
a Includes an incisional biopsy, if performed, followed at a later date by another wider surgery or separate map biopsies.
b Includes tumor excisions and map biopsies. There were 44 tumor-positive specimens out of a total of 119 manifesting infiltrative growth, intraepithelial spread, or both.
| Patient # | Surgery # | Predominant Pattern of Infiltrative Growth | Predominant Infiltrative Cell Type | Level of Differentiation (Vacuolization) in Infiltrative Cells b | Predominant Pattern of Intraepithelial Spread c | Epithelial Membrane Antigen | P53 % | Ki-67 PI | Adipophilin Staining Pattern (Grade) d |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | Trabecular | Epidermoid | Poorly differentiated | Pagetoid and meibomian gland carcinoma in situ e | Not done f | Not done | Not done | Not done |
| 1 | 2 | Trabecular | Epidermoid | Poorly differentiated | Carcinoma in situ | + (Diffuse) | 76.7 | 45.1 | Granular (+1) |
| 1 | 3 | Trabecular | Epidermoid | Poorly differentiated | Carcinoma in situ | + (Diffuse) | 82.1 | 32.4 | Granular (+1) |
| 2 | 1 | Not present | Not applicable g | Not applicable | Carcinoma in situ (intralobular) | + (Focal) | 85.9 | 43.0 | Vacuolar (+3) |
| 2 | 2 | Not present | Not applicable | Not applicable | Carcinoma in situ | – | 88.9 | Not available e | Vacuolar (+1) |
| 3 | 1 | Not present | Not applicable | Not applicable | Carcinoma in situ | + (Focal) | 79.1 | 65.8 | Granular (+2) |
| 3 | 2 | Microinvasion | Epidermoid | Poorly differentiated | Carcinoma in situ | + (Focal) | 79.4 | Not done | Granular (+2) |
| 4 | 1 | Not present | Not applicable | Not applicable | Carcinoma in situ | + (Focal) | Not done | Not done | Not done |
| 4 | 2 | Lobular with comedonecrosis | Basosquamous | Moderately differentiated | Pagetoid | + (Focal) | 64.1 | 46.7 | Granular (+2) Vacuolar (+1) |
| 5 | 2 | Trabecular and papillary | Basaloid | Well differentiated | Not present | + (Diffuse) | – | 50.6 | Vacuolar (+3) Granular (+2) |
| 6 | 1 | Lobular | Basosquamous | Poorly differentiated | Not present | Not done f | Not done | Not done | Not done |
| 6 | 2 | Lobular | Basosquamous | Poorly differentiated | Not present | + (Diffuse) | 81.0 | 37.3 | Vacuolar (+2) Granular (+1) |
| 6 | 3 | Lobular | Basosquamous | Poorly differentiated | Not present | + (Diffuse) | 85.4 | 41.3 | Vacuolar (+2) Granular (+1) |
| 6 | 4 | Lobular | Basosquamous | Poorly differentiated | Not present | + (Diffuse) | 90.4 | 42.8 | Vacuolar (+2) Granular (+1) |
| 7 | 1 | Lobular | Basaloid | Moderately differentiated | Not present | + (Focal) | 77.4 | Not done | Vacuolar (+1) Granular (+1) |
| 7 | 2 | Lobular | Basaloid | Moderately differentiated | Not present | + (Diffuse) | 83.1 | 52.3 | Granular (+1) Vacuolar (+1) |
| 8 | 1 | Lobular | Basaloid | Poorly differentiated | Pagetoid | + (Focal) | 58.9 | Not done | Granular (+2) Vacuolar (+1) |
| 9 | 1 | Not present | Not applicable | Not applicable | Carcinoma in situ | + (Focal) | 41.5 | Not done | Vacuolar (+3) Granular (+1) |
| 9 | 2 | Papillary | Basaloid | Well differentiated | Carcinoma in situ | + (Focal) | 33.7 | 45.2 | Vacuolar (+3) Granular (+2) |
| 10 | 1 | Lobular | Basaloid | Moderately differentiated | Pagetoid | + (Focal) | 76.4 | Not done | Vacuolar (+3) Granular (+1) |
| 10 | 2 | Lobular | Basosquamous | Moderately differentiated | Pagetoid | + (Focal) | 80.2 | 38.9 | Vacuolar (+2) Granular (+1) |
| 11 | 1 | Basal cell carcinoma–like | Basaloid | Poorly differentiated | Carcinoma in situ | + (Focal) | 49.1 | 27.4 | Granular (+1) Vacuolar (+1) |
| 12 | 1 | Lobular | Epidermoid | Poorly differentiated | Carcinoma in situ | + (Diffuse) | 48.6 | 62.6 | Granular (+1) |
| 12 | 2 | Lobular with comedonecrosis | Epidermoid | Poorly differentiated | Carcinoma in situ | Not done | Not done | Not done | Not done |
a Out of the total of 44 tumor-positive specimens, data on only 24 are summarized in the table. These are the most representative specimens for each case.
b Indirectly determined by degree of cytoplasmic vacuolization: well differentiated, >60% of lipidized cells; moderately differentiated, 25%-60%; poorly differentiated. <25%.
c Cell types of intraepithelial spread are not specifically categorized in the table but were either small, spindled, or ovoid in carcinoma in situ pattern, or large and polygonal in pagetoid pattern.
d +1, sparse positivity; +2, moderate positivity; +3, marked positivity.
e Pagetoid pattern was always found in epidermis and at eyelid margin either alone or together with carcinoma in situ in palpebral conjunctival epithelium.
f Not done means glass slides were reviewed, but tissue blocks were unavailable or inadequate for additional immunostaining.
g Not applicable indicates absence of infiltrative component precluding cellular characterization.
Results
Clinical Findings
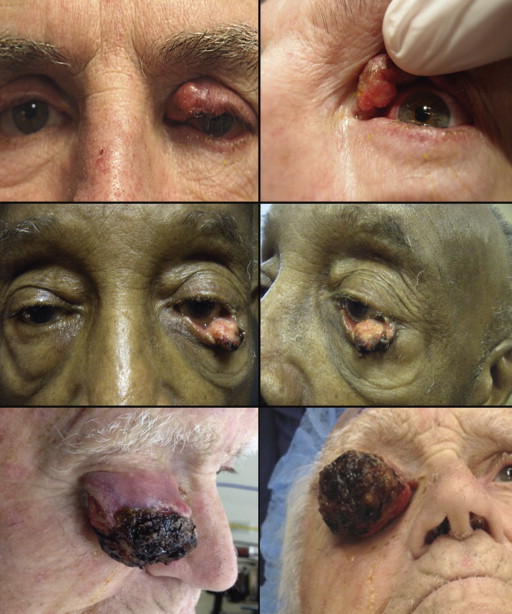
The clinical features of the cases involved in this study are summarized in Table 1 . Among the 12 cases, 7 patients were female and 5 were male. The mean age at presentation was 77 years. The right eye was affected in 4 cases and the left in 8. The upper eyelid was involved in 7 patients ( Figure 1 , Top left and Top right) and the lower in 5 ( Figure 1 , Middle left and Middle right). The tumor initially presented as a discrete mass or eyelid nodule in 7 patients, while diffuse eyelid thickening was initially seen in 3 patients and was usually associated with conjunctival hyperemia. One patient had a subconjunctival multinodular protruberant growth ( Figure 1 , Top right), another had blunt papillary projections of the palpebral conjunctiva ( Figure 1 , Middle right), and another developed a neglected, ulcerated lesion with crusting and madarosis ( Figure 1 , Bottom left and Bottom right); all these tumors had a vaguely yellow hue. An unbiopsied preauricular lymph node enlargement was detected in only 1 patient. One patient had had colonic carcinoma resected 5 years earlier. The median number of surgical procedures per patient was 2 (range of 1-4); the median number of specimens obtained for each patient over the entire course of his or her disease was 9 (range of 1-21). Six cases were initially misdiagnosed as chalazion, blepharitis, basal cell carcinoma, and/or squamous cell carcinoma before the final diagnosis of sebaceous carcinoma was eventually made. Local recurrences occurred in 5 cases. The mean length of follow-up was 35 months. Ten patients are alive without disease and 2 have died of causes unrelated to their tumors.
Histopathologic Findings
The gross specimens consisted of a variety of tissue types from the different cases at initial surgery or upon recurrences. They encompassed biopsies, partial- or full-thickness eyelid resections, additional strips of tissue to assess adequacy of surgical margins, and biopsies of nodules and small biopsies of conjunctiva performed to map out the extent of intraepithelial tumor. The main histopathologic features are summarized in Table 2 . Out of a total of 44 tumor-positive specimens, only 24 are listed in Table 2 . These are the most representative specimens from surgeries done on each case. Of these 44 tumor-positive specimens (out of 119 removed from the patients), 24 displayed both infiltrative growth and intraepithelial (“pagetoid”) spread, 12 had infiltrative growth only, and 8 had intraepithelial spread only. Since each patient had more than 1 kind of tissue result, the summary per patient is that 8 had both infiltrative and intraepithelial growth within their accumulated specimens throughout the full course of their disease (including recurrences), 3 manifested infiltrative growth only, and 1 displayed intraepithelial growth only (an in situ carcinoma of an underlying meibomian gland was eventually found).
The main growth pattern evidenced by the majority of the patients’ invasive tumors was lobular without peripheral palisading or clefting, wherein the tumor cells comprised variably sized units ( Figure 2 , Top left and Top right). These were generally small to medium-sized aggregates. Differentiation in the form of vacuolization often became more prominent centrally in the lobules with outer, more primitive nonvacuolated cells. In many moderately differentiated tumors, however, vacuoles were evenly distributed throughout the lobules. Prominent comedonecrosis consisting of total cellular dissolution (mimicking holocrine secretion) in the centers of the lobules was noted in 2 cases ( Figure 2 , Top left), although subtler degrees of central liquefactive holocrine dissolution were seen in several other lesions. Papillary growth was detected in 1 case ( Figure 2 , Middle left), whereas trabecular ( Figure 2 , Middle right), infiltrative or strand-like ( Figure 2 , Bottom left), and basal cell carcinoma–like ( Figure 2 , Bottom right) patterns were noted in the remainder of the cases. No instance of an “Indian file” infiltrative pattern was observed. As an incidental finding, 3 biopsies revealed histopathologic features of a lipogranuloma (chalazion), probably attributable to blockage of normal meibomian excretory ducts by tumor.
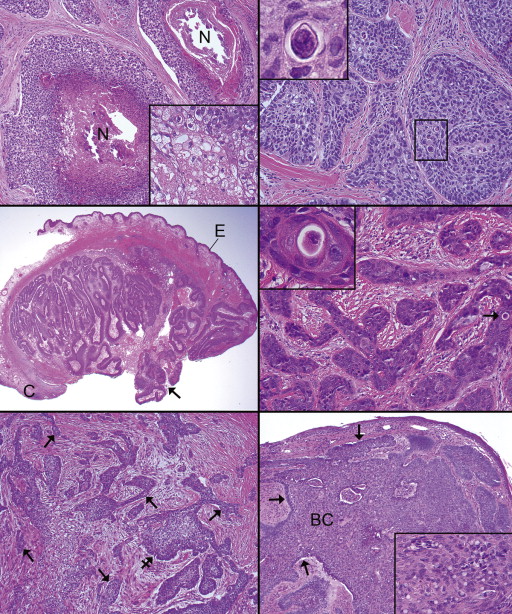
Several cell types and levels of differentiation among the various lesions often overlapped from lesion to lesion and were even combined within the same lesion, in that different fields could offer disparate architectures and cellular typologies. With 1 exception in which de-differentiation occurred, all the other recurrences preserved the same overall architecture and level of differentiation. The main cell types encountered, in decreasing order of frequency, were the basaloid, basosquamous, and epidermoid. Sebocyte-like cells were a subset of cells discovered only in well-differentiated tumors ( Figure 3 , Top left and inset, and Top right and inset). They manifested either mulberry-type vacuolization or a frothy appearance of total cytoplasmic vacuolization of various-sized spaces considered stereotypical for sebaceous carcinoma. In our material this feature was focally in only 3 lesions, generally with a background basaloid cellular population identified. Five cases had tumors composed mostly of small “blue” or basaloid cells, displaying a high nuclear-to-cytoplasmic ratio and inconspicuous vacuoles ( Figure 2 , Bottom right and inset; Figure 3 , Top left). The basosquamous and epidermoid cell types predominating in 7 cases consisted of small to medium-sized cells with modest to moderate amounts of eosinophilic cytoplasm ( Figure 3 , Middle left and Middle right). These cell types sometimes displayed easily overlooked scattered dyskeratotic cells ( Figure 2 , Top right and inset, Middle right and inset). Among the epidermoid lesions ( Figure 3 , Bottom left and inset), small foci of central keratin compaction simulated holocrine secretion ( Figure 3 , Bottom right); pancytokeratin immunostaining disclosed intense positivity in these foci ( Figure 3 , Bottom right inset). According to a tripartite designation of level of differentiation adopted herein (well differentiated, >60% vacuolated cells; moderately, 25%-60%; poorly, <25%), the patients’ invasive nodules were mostly poorly differentiated ( Figure 3 , Bottom right), 3 were moderately differentiated ( Figure 3 , Middle left), and 2 were well differentiated ( Figure 3 , Top left and Top right). If a lesion displayed an organoid growth pattern with central, highly vacuolated cells (sebocyte-like) surrounded by a thick cuff of less differentiated basaloid cells, it was designated as well differentiated ( Figure 3 , Top left).
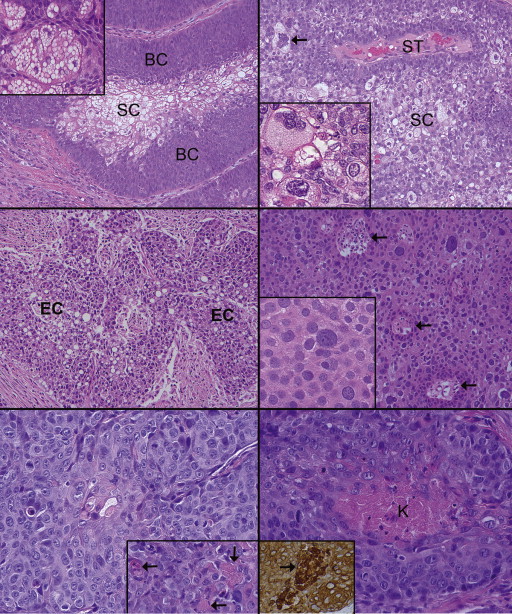
The nuclear features of the invasive cells in our cases were also helpful in distinguishing sebaceous carcinoma from other tumors. The typical, well-preserved nucleus not marred by poor fixation or tissue compression during surgery or tissue processing was round or oval and endowed with finely divided, ink-drop or smudgy chromatin that did not marginate at the nuclear membrane ( Figure 4 , Top left and Top right). The nucleoli were punctate and hematoxylinophilic, with either 1 or 2 per nucleus. Pleomorphism in the form of large, atypical, or bizarre nuclei ( Figure 3 , Top right and inset) and cells with multiple nuclei ( Figure 3 , Top right, main panel) were observed in about half of the cases and distributed in a random fashion. Mitotic activity was high in most tumors, with 2-3 mitoses detected per high-power field ( Figure 4 , Top left and Top right including inset). The normally round or oval shape of the nucleus was sometimes deformed, indented, or eccentrically displaced by cytoplasmic vacuoles in the moderately differentiated and well-differentiated tumors ( Figure 3 , Middle left, and Figure 4 , Top right inset). In lesions with more pronounced epidermoid characteristics, the nuclei tended to be vesicular with more prominent nucleoli ( Figure 3 , Bottom left and right).
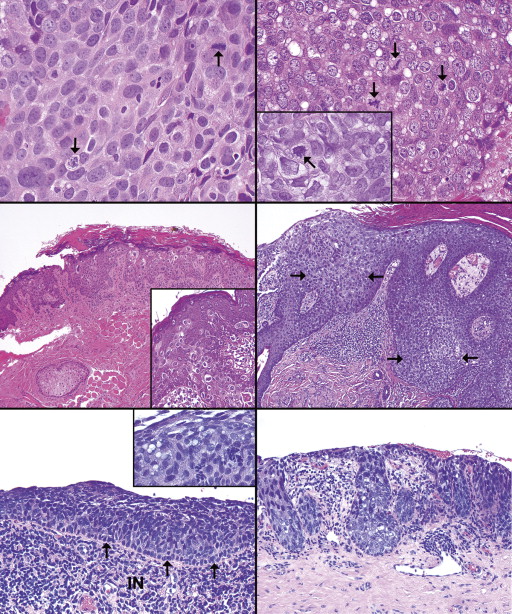
Two patterns of intraepithelial spread of sebaceous carcinoma cells were found. At the eyelid margin and in the epidermis of the skin only pagetoid spread was detected, whereas in the palpebral conjunctival epithelium, either a pagetoid or carcinoma-in-situ pattern could be featured. Intraepithelial sebaceous carcinoma cells exhibit nuclei that are 50%-100% larger than those of neighboring normal keratinocytes. One case was remarkable because the entire disease was restricted to the epithelium, notably an in situ carcinoma of a meibomian gland with accompanying eyelid margin intraepidermal spread. Tumor cell extension into adnexal structures (mostly follicles) was detected in 3 cases with intraepidermal involvement. The predominant pattern within the conjunctival epithelium mimicked an epidermoid (squamous) carcinoma in situ in 7 patients’ lesions. Less common in the conjunctiva was the pagetoid pattern (4 cases), constituted by atypical cells percolating individually or in small clusters at various levels of an identifiably normal, preexistent epithelium. In the vicinity of the orifices of the meibomian gland ducts, the pagetoid pattern was created ( Figure 4 , Middle left and inset; and Middle right) by cells that usually possessed clear cytoplasm or vacuoles. Parakeratosis was also displayed by the involved segments of epidermis and eyelid margin ( Figure 4 , Middle left and Middle right). In the palpebral conjunctival epithelium, atypical epithelial involvement occurred mostly as a carcinoma-in-situ pattern with spindly “blue” basaloid cells entirely replacing the normal epithelium as well as that of the pseudoglands of Henle ( Figure 4 , Bottom left and inset, and Bottom right; Figure 5 , Top left).
Three unusual features were noted in association with intraepithelial sebaceous carcinoma spread: partial preservation of the normal basal germinal cells of the conjunctival epithelium, above which were the atypical sebaceous cells ( Figure 4 , Bottom left); intense chronic inflammation of the subjacent substantia propria ( Figure 5 , Top left, left panel); and surface cell multivacuolar differentiation developing apically above multiple layers of primitive spindled basaloid tumor cells—observed in 1 case with papillary features ( Figure 5 , Top left, right panel). True pagetoid colonization of the conjunctival epithelium stood out as clear cells retracted from the surrounding keratinocytes ( Figure 5 , Top right, top and bottom panels). The skip areas of conjunctival intraepithelial involvement were common and would probably have been interconnected in a 3-dimensional reconstruction of tumor spread ( Figure 5 , Middle left, top and bottom panels). A highly suggestive feature of intraepithelial sebaceous carcinoma cells was a tendency for focal or extensive cell sloughing or dyscohesiveness in a third of the lesions ( Figure 5 , Middle right, top and bottom panels).
Immunohistochemical Findings
All infiltrative lesions stained positively for EMA. Five lesions with better-differentiated features manifested uniform staining of the cell membranes in the lobules ( Figure 5 , Bottom left), whereas only focally erratic collections of such cells were discovered ( Figure 5 , Bottom right and inset) in 5 poorly differentiated tumors. p53 was highly positive in all but 1 of the invasive growths (average of 71.7%, range of 33.7%-90.4%) ( Figure 6 , Top left). It could not be determined whether the negative result was the consequence of a technical failure or a true attribute of the tumor. In the positive lesions both the pleomorphic and smaller tumor cell nuclei immunoreacted positively. Ki-67 ( Figure 6 , Top left, inset) in the infiltrative components of the tumors had an average proliferative index of 45.1% (range 27.4%-65.8%). A reliable proliferation index could not be calculated for the intraepithelial spread because of difficulty in distinguishing between normal basal cell mitotic activity and tumor cell mitoses.
With respect to intraepithelial tumor spread, which was present in 9 out of the 12 cases in at least 1 specimen throughout the course of the disease and its recurrences, EMA was dramatically positive in the colonizing tumor cells ( Figure 6 , Top right, top and bottom panels). The adjacent epidermis or conjunctival epithelium displayed either faint or no basilar and suprabasilar cell staining. Likewise, p53 vividly immunoreacted with the tumor cells in the epidermis, marginal eyelid epithelium, and conjunctival epithelium ( Figure 6 , Middle left, top and bottom panels). Adjacent basilar cells of the epidermis ( Figure 6 , Middle left, top panel) were less intensely positive, and the basilar cells of the palpebral conjunctival epithelium were generally negative. The pseudoglands of Henle of the palpebral conjunctiva were not uncommonly replaced by p53-positive neoplastic cells surmounted by a thin surface of nonneoplastic epithelial cells or only focally positive surface cells ( Figure 6 , Middle right, top panel). Areas where most of the conjunctival epithelium had sloughed still harbored atypical hyperchromatic basilar cells that were p53 positive ( Figure 6 , Middle right, bottom panel). All infiltrating patterns of the tumors that we studied were Ber-EP4 negative.
Adipophilin was strongly positive in the most differentiated infiltrating tumor cells ( Figure 6 , Bottom left) and compared favorably with correlative oil red O staining ( Figure 6 , Bottom left, inset). There were vagaries in adipophilin staining, in that different regions of infiltrative growth with the same histopathologic features might be strongly or weakly positive or completely negative, as was also discovered with respect to “skip” areas of negativity in frankly involved segments of surface epithelium. Tumor cells with well-differentiated or sebocytic features of capacious, frothy, and totally vacuolated cytoplasm were usually highly positive ( Figure 6 , Bottom right), but in some instances they were either weakly staining or nonstaining, as if their lipid contents had been partially or totally released. The basaloid cells that frequently enveloped the collections of sebocytic cells were nonstaining. In more closely analyzing the nature of the adipophilin cytoplasmic staining, positivity was attributable to clear-cut vesicular (vacuolar) rimming by immunoreactant ( Figure 7 , Top left, left panel) detected in the better-differentiated lesions. On the other hand, fine nonvacuolar granular or particulate staining ( Figure 7 , Top left, right panel) was detected in the poorly differentiated lesions, particularly those in which basaloid or epidermoid cells were featured. Basosquamous lesions could manifest both types of adipophilin morphologies. In lobules displaying central comedonecrosis, adipophilin positivity occurred in both vesicular (vacuolar) and granular forms ( Figure 7 , Top right and inset). Vacuolated cells in lobules with moderate differentiation in comparison to full-blown sebocytes were also positive, but less intensely so ( Figure 7 , Middle left). In the lesion with a basal cell carcinoma–type growth pattern (but lacking any connection to the epidermis and possessing an accompanying intraepithelial component—see Figure 2 , Bottom right), small scattered clusters of adipophilin-positive cells displaying vesicular forms were randomly scattered throughout the tumor cell lobules ( Figure 7 , Middle right).




