Chapter 52 External Beam Radiotherapy for Thyroid Malignancy
The standard management of differentiated thyroid cancer with surgery and with or without radioactive iodine as described elsewhere in this book is well established (see Chapter 51, Postoperative Radioactive Iodine Ablation and Treatment of Differentiated Thyroid Cancer). The role of external beam radiotherapy (XRT) is more controversial. As in all aspects of thyroid oncology, there is a paucity of randomized controlled trials to either support or refute the role of external beam radiotherapy. Evidence comes almost exclusively from retrospective data, so conclusions have to be interpreted with caution. That being said, although it is not a substitute for appropriate surgery, we do believe that there is sufficient evidence to support the use of external beam radiotherapy in selected cases. This chapter reviews the evidence and gives our recommendations for its use. We will outline the role not only for differentiated thyroid cancer, but for anaplastic and medullary thyroid cancer and non-Hodgkin’s lymphoma.
Differentiated Thyroid Cancer
XRT for Gross Disease
That external beam radiation therapy can control unresected thyroid cancer has been known for some time. As far back as 1966, Sheline et al. reported their experience with a follow-up of 1 to 15 years; 8 out of 15 patients with gross residual or unresectable papillary thyroid cancer were alive with no evidence of disease.1 In a large series from Hong Kong of 217 patients with gross residual papillary thyroid cancer, 70% of whom had had external beam radiation, the 10-year local relapse-free rate (LRFR) and cause-specific survival (CSS) were significantly better in patients who were treated with external beam radiation than in those who were not (63.4% versus 24%, p < 0.001 for LRFR and 74.1% versus 49.7% for CSS, p = 0.01). The improvement in local control was seen in both subgroups with and without palpable residual disease after surgery.2 We have reported from Princess Margaret Hospital that of 33 patients with gross residual or unresected differentiated thyroid cancer treated with external beam radiation, the LRFR at 5 years was 62% and the CSS was 65%.3 More recently, 15 patients were treated definitively for gross disease with differentiated thyroid cancer at the MD Anderson Hospital; 4 showed a complete response, 3 had partial response, 6 had stable disease, and only 2 experienced progression through treatment. All 4 patients with complete disease response remained free of locoregional relapse with follow-up of 13, 14, 29, and 63 months.4
XRT without Gross Disease
The question remains, is there a role for external beam radiation therapy in patients who have had a resection of all gross disease? About 8% of patients with papillary thyroid cancer have extrathyroidal extension, and especially in older patients this is a poor prognostic factor.5,6 Mazzaferri and Jhiang showed that local tumor invasion into the neck structures was associated with higher 20-year recurrence rates and higher 20-year cancer mortality rates than tumor confined to the thyroid gland.7 When tumors with extrathyroid extension recur, the recurrent local tumor is less likely to take up 131I and the patient is also more likely to die of the disease.8 This suggests that surgery and 131I may be inadequate treatment in older patients with extrathyroidal extension.
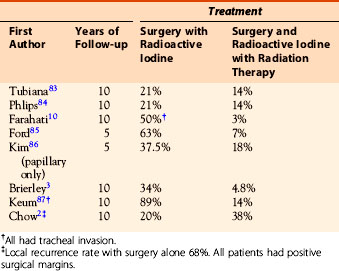
Many single institutional retrospective reviews have addressed the role of external beam radiotherapy after all gross disease has been resected. Some of the earlier studies suggest that external beam radiation has no benefit or even a deleterious effect, but these contain a large number of patients with good prognostic features that would not be expected to derive benefit. This is also true for a randomized clinical trial from Germany that failed to complete because of failure to recruit.9 Although an analysis of patients randomized or registered and compared with those not randomized showed no benefit from external beam radiation, half the patients had T3 disease and would not be expected to be at high risk and overall only 26 received external beam radiation. Most retrospective studies do, however, show a benefit in selected cases (Table 52-1). The most persuasive older study was from Faraharti et al., who reported a decreased locoregional and distant failure in patients over the age of 40 with extrathyroidal extension and lymph node involvement when treated with adjuvant radiation.10 All patients in this study, unlike many others studies, had therapy consisting of total thyroidectomy, 131I, and thyroid-stimulating hormone (TSH) suppression in addition to external beam radiation. External beam radiation was a predictive factor for improvement in time to locoregional recurrence (p = 0.004) and locoregional and distant failure (p = 0.0003). In the most recent data from Princess Margaret Hospital, there was a higher 10-year CSS (81% versus 65%) and a higher LRFR (86% versus 66%) among patients older than 60 years with extrathyroidal extension but no gross residual disease who had been treated with external beam radiation compared with those who had not.3 In a study from Hong Kong, in 251 patients with positive margins after surgery, both age and size of the tumor were additional prognostic factors. The 10-year LRFR was 32% when no 131I or external beam radiation were given, 57% with external beam radiation, 80% with 131I, and 90% when both 131I and external beam radiation were given.2 The benefit from external radiation was only seen in patients with T4 disease and was not seen in patients with close but negative margins (0.01 to 3 mm) or T3 disease (minimal extrathyroid extension to sternothyroid muscle or perithyroid tissue).
Table 52-1 Local Recurrence Rates after Adjuvant External Radiation Therapy for High-Risk Disease Differentiated Thyroid Cancer
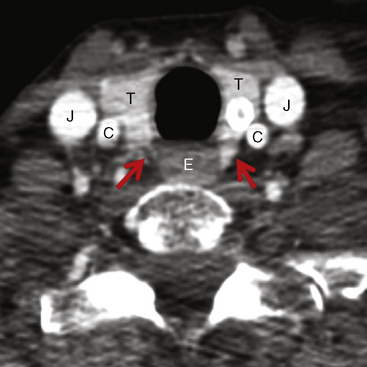
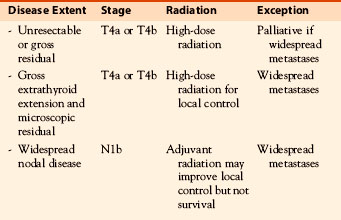
These studies indicate that external beam radiation in addition to surgery and 131I improves local control and possibly survival in high-risk patients, defined as older patients with gross extrathyroidal extension at the time of surgery that is resected but with microscopic disease present postoperatively (i.e., R 1 resection). Typically these are patients with extrathyroidal extension into the tracheoesophageal groove (Figure 52-1) involving the recurrent laryngeal nerve, trachea, or larynx in whom resection with negative margin would require loss of function and in whom if the disease recurred extensive ablative surgery (such as pharyngolaryngectomy) would the only surgical option.
The use of external beam irradiation to treat the primary tumor should be considered in patients over age 45 with grossly visible extrathyroidal extension at the time of surgery and a high likelihood of microscopic residual disease, and for those patients with gross residual tumor in whom further surgery or RAI would likely be ineffective.11
At Princess Margaret Hospital, the current recommendations are that older patients (50 plus) who have gross extrathyroidal extension (cT4) and probable microscopic disease or positive surgical margins are considered for postoperative external beam radiation in addition to 131I. It is also considered for highly selected younger patients with locally extensive residual disease (i.e., T4b) or extensive T4a disease with poor prognosis based on histologic features. Lymph node involvement by itself is not an indication for external beam radiation therapy because regional control is usually achieved with neck dissection in combination with postoperative radioiodine. Although Chow et al. reported that external beam radiation improved 10-year LRFR in patients with N1b disease (58% versus 70%, p < 0.0001) and also for patients with nodes larger than 2 cm, in their multivariate analysis it was more radical surgery and 131I, but not external beam radiation, that improved lymph node control.2 As discussed later, at Princess Margaret Hospital we reserve external beam radiation as an option to nodal beds for failure after neck dissection and 131I. However, it may be of value for patients with extensive and locally invasive extracapsular extension of lymph node disease at presentation.
Local or Regional Recurrence of Differentiated Thyroid Cancer
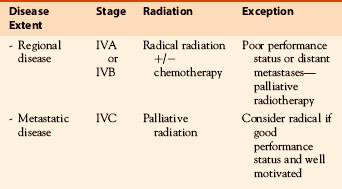
Most differentiated thyroid cancers that recur do so in the cervical lymph nodes, and these are usually adequately treated by neck dissection and 131I. Neck node recurrence after ablation of the thyroid bed may not represent failure of 131I if there was extensive uptake by residual thyroid tissue resulting in an inadequate dose to lymph nodes containing metastatic disease. In this situation, further therapeutic 131I after surgery may be of benefit. However, occasionally repeated recurrences are seen in the neck despite appropriate therapy, and for these patients, external beam radiation may be appropriate. If after a second administration of 131I there is further extensive nodal recurrence, one should consider external beam radiation after surgery. For recurrences in the thyroid bed with local invasion into the tracheoesophageal groove or adjacent structures, with a positive resection margin, external radiation should be considered after 131I therapy. This approach is similar to that described previously for patients who initially present with locally invasive disease (see Table 52-2 and Figure 52-4 for differentiated thyroid cancer radiotherapy treatment summary recommendations and case report).
Anaplastic Thyroid Carcinoma
Anaplastic thyroid carcinoma is among the most deadly of human cancers (see Chapter 26, Anaplastic Thyroid Cancer and Thyroid Lymphoma). Comprising only a minority (< 5%) of thyroid cancers, it nonetheless accounts for up to half of thyroid cancer deaths.12,13 Unlike differentiated thyroid malignancies, the majority of anaplastic thyroid cancer patients do not survive 6 months following diagnosis. Anaplastic disease typically presents as a rapidly growing, infiltrative neck mass causing dyspnea, dysphagia, or neck pain. It is frequently inoperable at presentation because of extensive invasion of adjacent blood vessels, trachea, esophagus, and soft tissues, or the presence of distant metastases in at least half of cases.14 Nonetheless, incidental discovery of focal de-differentiated disease within a resected differentiated tumor does occur, and such cases warrant aggressive management because significantly reduced survival can be expected in a significant proportion of such patients.
Optimal management of anaplastic thyroid cancer remains controversial given the rarity of presentation and absence of randomized data. Multimodality therapy is the cornerstone of aggressive treatment. Adjuvant external beam radiation is indicated following surgery, regardless of the completeness of resection. If surgery is not feasible, definitive chemoradiotherapy or hyperfractionated radiotherapy can be offered.15–20 Clinical results with definitive radiation-based treatment have remained poor through the years; results appear to be best for patients able to undergo complete surgical removal of disease prior to radiation.17,18,21–26 The published literature suggests that dose escalation and hyperfractionation may help to offset the rapid proliferation and radioresistance of anaplastic disease.19,21,27 The addition of aggressive cytotoxic chemotherapy to hyperfractionated radiation has been studied for several decades.15,20 Unfortunately, significant treatment toxicity was observed in early series28 and radiation fraction sizes and cumulative doses were scaled back in subsequent trials.20 The therapeutic ratio of current versions of this approach remains poor, with no substantive improvements in treatment toxicity or disease control rates. A recent series of 30 patients prospectively enrolled onto a clinical trial were treated to 40 Gy in twice-daily 1.25 Gy fractions with two cycles of 60 mg/m2 doxorubicin and 120 mg/m2 cisplatin; these patients yielded a 10-month median overall survival.17 Twenty-four patients underwent surgery, 20 of whom subsequently underwent chemoradiation. A notable minority of patients (27%) enjoyed durable 3-year survival, albeit at the cost of frequent grade 4 pharyngitis, esophagitis, and marrow toxicity.
As noted later, intensity-modulated radiation therapy (IMRT) serves as a potential means to safely deliver high radiation doses to appropriately selected patients. Nonetheless, the recently published experience of modern three-dimensional radiation therapy (3DRT) or IMRT technique for anaplastic disease from MD Anderson Cancer Center demonstrated survival outcomes in 56 patients, which closely resemble those from older published reports for older conventional techniques.29 Median follow-up was 4 months (range: 1 to 56 months) for the entire cohort and 28 months (range: 12 to 49 months) for surviving patients. Kaplan-Meier estimates of disease-specific survival (DSS) and overall survival (OS) at 1 year were both 19%. Median DSS and OS were identical at 3 months. For patients irradiated aggressively with the intent to deliver definitive dose, Kaplan-Meier estimates of DSS and OS at 1 year were 29%, with a median DSS and OS of 7 months. Twenty-five patients with the most realistic chance to benefit from locoregionally aggressive treatment (i.e., with no evidence of distant metastases at the time of irradiation) were analyzed separately. These patients received a median dose of 60 Gy (range: 38 Gy to 66 Gy) with a median fraction size of 2 Gy (range: 1.25 Gy to 2.2 Gy). All patients received concurrent chemotherapy, and 9 (36%) received twice-daily fractionation. DSS and OS at 3 years for this subpopulation was 21% and 16%, respectively. DFS and OS at 3 years for 12 patients who had complete surgical resection with no gross residual disease prior to aggressive post-op chemo/radiotherapy were both 33%, consistent with older reports and with the notion that durable survival can indeed be seen in this subgroup of patients if aggressive multimodality locoregional treatment is pursued. In contrast, in a series from Princes Margaret Hospital of 23 patients irradiated aggressively, mostly without chemotherapy, the 1-year survival rate was 46.1% and the median survival was 11.1 months. For 14 patients treated with a single daily fraction, the median survival was 10.3 compared to 13.6 months (p = 0.3) for patients given 60 Gy in 40 fractions of 1.5 Gy each over 4 weeks.30
Improvements in late toxicity with IMRT are difficult to assess for anaplastic disease, given the complicating issue of competing risk of rapid local and distant disease progression. Obvious reductions in acute treatment toxicity were not seen with use of IMRT in the series discussed earlier and were most likely the result of a continued need for wide-field coverage and concurrent administration of chemotherapy. Given the toxicity and efficacy shortcomings of conventional chemoradiotherapy, the importance of developing novel biologically targeted approaches now supersedes that of technical improvements in radiation delivery. Recently published data have demonstrated single-agent activity in mixed study populations with radioiodine-refractory differentiated or undifferentiated thyroid disease for multitargeted tyrosine kinase inhibitors, such as axitinib and sorafenib (see Chapters 55, Medical Treatment for Metastatic Thyroid Cancer, and 26, Anaplastic Thyroid Cancer and Thyroid Lymphoma).31,32 There are compelling mechanistic rationales for investigating these or similar agents as potential radiosensitizers, and future clinical trials may provide promise for improved control of this otherwise radiorefractory disease. In addition to effective radiosensitizers, better ways of selecting patients who will benefit from an aggressive approach are required so that patients with a limited life expectancy are spared the significant toxicities associated with a multimodality therapy (see Table 52-4 and Figure 52-6 for anaplastic radiotherapy treatment summary recommendations and case report).
Medullary Thyroid Carcinoma
Surgery is the main curative treatment modality for medullary thyroid carcinoma (MTC). Because regional lymph nodes are involved in approximately 50% of sporadic patients, meticulous neck and superior mediastinal dissections are often required. Radioactive iodine has no role in the management of MTC. For those who have normal calcitonin and CEA levels following surgery, especially if obtained following calcium stimulation, the prognosis is excellent.33,34 However, a significant proportion of patients will have postoperative residual disease as evidenced by high serum calcitonin and CEA levels.35 Additional factors that predict for a poorer prognosis include extrathyroidal invasion, postoperative gross residual disease,36 and clinical stage.34,35 In these patients, further investigation to locate for regional or metastatic disease is required, with computed tomography (CT) scans, somatostatin and bone scans, and18 FDG-PET.37–40 A higher sensitivity of detecting subclinical disease has been reported for18 F-dihydroxyphenylalanine (18F-DOPA) in comparison with 18FDG-PET.41,42 Subclinical nodal disease in the neck and superior mediastinum is relatively common,43,44 and if detected on PET scan, surgical reexploration should be considered to remove the nodal disease. Reoperation can achieve normalization of calcitonin levels.45 If further surgery is not possible or if residual disease is still evident despite repeated good quality surgical clearance, adjuvant external radiation to the thyroid bed and regional nodal tissue may be considered if no (or minimal) systemic spread of disease has been identified. Without further treatment, approximately half of these high-risk patients will recur in the neck. Radiation doses of 40 to 50 Gy in 2 Gy fractions to lymph nodes in the neck and upper mediastinum followed by a boost to the thyroid bed to a total of 50 Gy resulted in a locoregional control rate of 86% at 10 years.36 The present approach with IMRT adopts the dose/fractionation used in differentiated thyroid cancer, with IMRT delivering 63 to 66 Gy to gross disease and “high-risk” areas, and 54 to 56 Gy (in 30 to 33 fractions over 6 to 6.5 weeks) to cervical and superior mediastinal nodal regions. This treatment does not affect overall survival, but locoregional control is an important goal because cervical relapse can have a deleterious impact on the patient’s quality of life. In 34 patients with stage IVa to stage IVc disease treated to a median dose of 60 Gy with IMRT, the 5-year locoregional relapse-free rate was 87%, and the treatment morbidity was considered acceptable.46 Other investigators have confirmed similar results (see Table 52-3 and Figure 52-5 for MTC radiotherapy summary treatment recommendations and case report).47–51
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree