Purpose
To evaluate the outcomes of epiretinal membrane (ERM) surgery in highly myopic eyes without traction maculopathy, and to compare them with those from non–highly myopic eyes.
Design
Retrospective nested case-control study from a cohort of 509 consecutive patients (509 eyes) who underwent pars plana vitrectomy with ERM removal.
Methods
Thirty-two highly myopic eyes (with a refractive error of more than −6.00 diopters [D]), which underwent surgery for isolated ERM, were included in the study. For each case studied, we selected from the same cohort 2 age-matched controls who had ERM surgery (n = 64 non–highly myopic eyes). The best-corrected visual acuity (BCVA), the central macular thickness (CMT), and the surgical complications were analyzed.
Results
The mean follow-up duration was 3.2 ± 1.5 years for the study cases and 3.4 ± 1.6 years for the control group ( P = .608). At the final follow-up examination, the mean logMAR BCVA had improved significantly, from 0.56 to 0.26 ( P < .001) for the case group and from 0.54 to 0.22 ( P < .001) for the control group. At the final optical coherence tomography (OCT), the mean CMT had improved significantly, from 433 to 314 μm ( P < .001) for the case group and from 428 to 303 μm ( P < .001) for the control group. There was no significant difference between the 2 groups as regards visual or CMT improvement ( P = .526 and P = .483, respectively). The incidence of surgical complications was not significant between the 2 groups.
Conclusions
The results of ERM surgery were not different in terms of anatomic and visual outcomes and surgical complication between highly myopic and non–highly myopic eyes.
Recent epidemiologic studies have identified an association between myopia and primary epiretinal membrane (ERM), particularly since the advent of optical coherence tomography (OCT); however, little is known about the outcomes of isolated ERM surgery in highly myopic eyes (ie, without traction maculopathy). Furthermore, in a recent study, ERM without retinal changes was found in 98 out of 214 eyes (45.7%) suffering from pathologic myopia. Since highly myopic eyes can exhibit abnormal peripheral retina, vitreous liquefaction, abnormal vitreoretinal interface, and retinochoroid atrophy in the macular area, one could reasonably hypothesize that the outcomes of surgery for ERM in highly myopic eyes without traction maculopathy will likely differ from those for non–severely myopic eyes. For example, it has been shown, concerning surgery for isolated macular holes, that the visual outcomes in highly myopic eyes are poorer than those for eyes that are not severely myopic. Hence, data with respect to the surgical outcomes of isolated ERM in highly myopic eyes are needed. The purpose of this study was to evaluate, in a large series of patients, the anatomic and visual outcomes of isolated ERM surgery in highly myopic eyes, and to compare them with the outcomes for matched control eyes.
Patients and Methods
Patients and Study Design
The proposed study is a case-control study nested in a retrospective cohort of 509 consecutive patients with either primary or secondary ERM (509 eyes) who underwent pars plana vitrectomy performed by the same vitreoretinal surgeon (C.M.) at the Quinze-Vingts National Ophthalmology Hospital (Paris, France), between January 1, 2004, and July 31, 2010. Informed consent was obtained from all patients before surgery. Retrospective review of patient records was approved by the Ethics Committee of the French Society of Ophthalmology. The study adhered to the tenets of the Declaration of Helsinki.
The cases involved all patients of the cohort who had high myopia (defined as a spherical equivalent refractive error of −6.00 diopters [D] or more, before any surgery), without myopic traction maculopathy on OCT, and who had been operated on for ERM. In order to avoid potential confounding bias and better evaluate anatomic and visual outcomes, we selected, for each case studied, 2 controls from the same cohort, which we matched to the study cases on age (±1 year), preoperative visual acuity (±0.02 logMAR), year of surgery (±1 year), and gauge of vitrectomy system.
Exclusion criteria for potential study cases and control patients included: follow-up of less than 12 months’ duration; eyes with secondary ERM, such as diabetic retinopathy, venous occlusion, retinal detachment, uveitis, or trauma; and presence of age-related macular degeneration, diabetic maculopathy, macular hole, foveoschisis, or foveal detachment.
Surgical Procedure
The decision to operate on the ERM was made based on a patient’s macular symptoms and on his or her individual occupational or vocational needs. In all of the patients a central vitrectomy was performed. If posterior vitreous detachment (PVD) was not already present, the posterior vitreous hyaloid was separated from the retina by active aspiration with the vitrectomy probe, and any visible vitreous strands that were adherent to the retina were removed. In highly myopic eyes, intravitreous triamcinolone (40 mg/mL, Kenacort; Bristol-Myers Squibb, New York, New York, USA) was used as a marker to facilitate visualization and removal of the adherent posterior cortical vitreous, while it was not used in non–highly myopic eyes. A micro-vitreoretinal blade was then used as a pick to create a small opening and a flap tear in the ERM. The flap was then grasped with end-gripping forceps and carefully started in a circular capsulorrhexis maneuver. Internal limiting membrane (ILM) was not intentionally peeled in all eyes (cases or controls) operated on before January 2006. Thereafter, ILM peeling was systematically performed, using indocyanine green (ICG) or brilliant blue (BB) dyes to stain and then remove the ILM.
Outcome Data
All data were assessed by a chart review, including the age and sex of patients, refractive error (in diopters), lens status, types of degenerative myopic maculopathy, duration of symptoms, induction of PVD, gas or fluid tamponade, duration of surgery, sclerotomy leakage, intraocular pressure (IOP), best-corrected visual acuity (BCVA), and OCT findings. Postoperative complications included severe hypertony or hypotony, suprachoroidal hemorrhage, retinal tear, retinal detachment, vitreous hemorrhage, and endophthalmitis.
Snellen visual acuity was converted into the logarithm of the minimal angle of resolution (logMAR) for statistical analysis. IOP was measured with the Goldmann applanation tonometer. Severe postoperative hypotony and hypertony were defined as IOP <6 mm Hg and ≥30 mm Hg, respectively. PVD status was established in the event the Weiss ring was visible during preoperative biomicroscopic examination, and as a function of intraoperative findings. For highly myopic eyes, B-scan ultrasonography (Humphrey Instruments, San Leandro, California, USA) was also systematically performed before surgery in order to better evaluate the PVD and to measure the mean axial length. For retinal periphery examination, a contact lens was used (Three Mirror Glass or Volk QuadrAspheric lens (Volk Optical Inc, Mentor, Ohio, USA), depending on the lens status) intraoperatively and a noncontact lens (Volk Digital Wide Field Lens and Volk Digital Superfields Lens [Volk Optical Inc]) preoperatively and postoperatively.
Every patient underwent a detailed ophthalmologic examination before and after surgery. Postoperative visits were scheduled for all patients 1 day, 1 month, 3-6 months, and 1 year after surgery, and then annually. OCT scans were carried out preoperatively, to confirm the diagnostic of ERM; at 3-6 months postoperatively, to confirm the successful removal of the ERM; at 1 year; and as deemed necessary thereafter (OCT3 Stratus [Carl Zeiss Ophthalmic Systems, Inc, Humphrey Division, Dublin, California, USA] was used before July 2008 and spectral-domain OCT thereafter [either Heidelberg Spectralis-OCT; Heidelberg Engineering, Heidelberg, Germany, or Cirrus HD-OCT; Carl Zeiss Meditec Inc, Dublin, California, USA]).
Statistical Analysis
Results were presented as mean ± standard deviation (SD) for continuous variables and as proportions (%) for categorical variables. Student t test and nonparametric Mann-Whitney were used to compare continuous data. Paired Student t test was used to statistically evaluate comparisons between preoperative and postoperative logMAR VA, IOP, and macular thickness on OCT. For binary outcomes, the stratified Cochran χ 2 test and Fisher exact test were used for intergroup comparisons of proportions when appropriate. P values of .05 or less were considered statistically significant. Statistical analysis was carried out using SPSS for Windows version 20.0 (SPSS, Inc, Chicago, Illinois, USA).
Results
Baseline Characteristics of the Cases
None of the patients in the initial cohort (n = 509) had previously undergone refractive surgery. We identified, from this cohort, 36 patients with high myopia (−6.00 D or greater). None of them had a history of choroidal neovascularization (CNV). Two patients had ERM associated with retinoschisis on OCT examination and 2 had ERM secondary to retinal detachment. These 4 highly myopic patients were not included in the analysis.
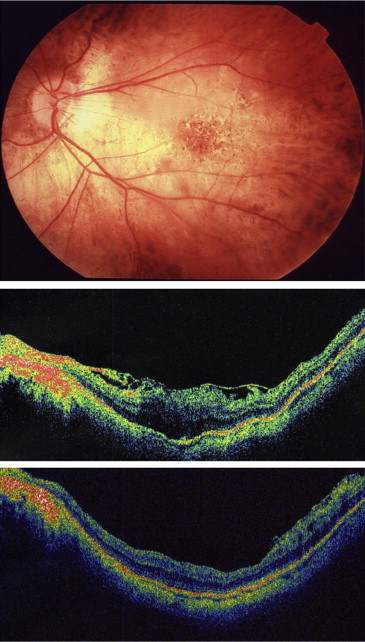
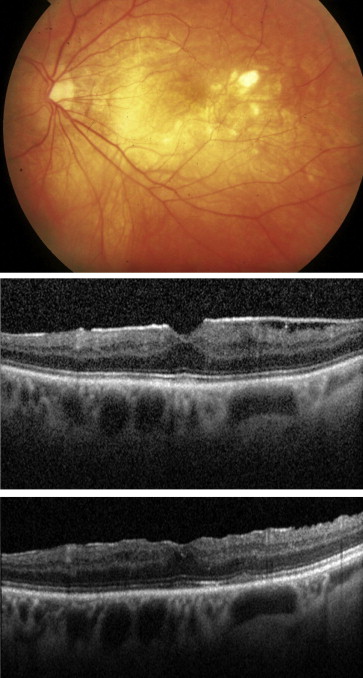
The mean age of the highly myopic cases (n = 32) was 59.88 ± 7.25 years, which was generally younger than the remaining patients of the cohort (68.73 ± 9.5 years) ( P < .001, Student t test). Fourteen of these were male and 18 female (sex ratio = 0.78). The mean axial length was 27.30 ± 0.97 mm. The preoperative BCVA (logMAR) was 0.56 ± 0.20, and the mean preoperative central macular thickness (CMT) was 433.1 ± 71.6 μm. Preoperative OCT confirmed the presence of ERM in all 32 of the eyes and showed that 8 eyes (25%) had posterior staphyloma. Thirty cases had diffuse thickening of the macula ( Figures 1 and 2 ), and 2 cases had cystoid macular edema. The ERM was in no cases associated with foveoschisis, macular hole, or macular detachment. Fundus examination of these highly myopic eyes revealed that 16 eyes (50%) had tessellated fundus alone, 5 eyes (16%) had diffuse chorioretinal atrophy ( Figure 1 ), 1 eye (3%) had diffuse chorioretinal atrophy with patchy atrophy ( Figure 2 ), and 1 eye (3%) had diffuse chorioretinal atrophy with lacquer cracks. None of the patients had CNV or macular atrophy. Twenty-six patients underwent 20-gauge vitrectomy and 6 underwent 23-gauge vitrectomy.
Comparison of Baseline Characteristics of the Patients in the Case and the Matched Control Groups
Sixty-four matched patients were selected from the initial cohort, as described in the Methods section. Table 1 sets forth the characteristics of the cases (n = 32) and matched controls (n = 64). Mean spherical equivalent refraction was −8.48 ± 2.75 D in the highly myopic group and 0.12 ± 1.83 D in the control group ( P < .0001, Student t test). The highly myopic group had a mean preoperative best-corrected visual acuity (logMAR) of 0.56 ± 0.20, while it was 0.54 ± 0.22 logMAR in the control group ( P = .618, Student t test). Mean preoperative macular thickness did not differ between the 2 groups ( Table 1 ). Compared to the controls, study cases were more likely to have ERM on the fellow eye; however, the difference was not significant ( Table 1 ).
| Characteristics | Cases (n = 32) | Controls (n = 64) | P Value |
|---|---|---|---|
| Age (y), mean (SD) | 59.88 (7.25) | 60.13 (7.22) | .873 a |
| Sex | |||
| Male | 14 | 28 | 1.000 b |
| Female | 18 | 36 | |
| Mean duration of symptoms (mo) mean (SD) | 11.70 (9.01) | 14.09 (12.28) | .373 a |
| Mean refractive error (diopters), mean (SD) | −8.48 (2.75) | 0.12 (1.83) | <.001 a |
| Preoperative best-corrected visual acuity (logMAR) mean (SD) | 0.56 (0.20) | 0.54 (0.22) | .618 a |
| Preoperative central macular thickness (μm) mean (SD) | 433.1 (71.6) | 427.8 (66.7) | .873 a |
| Lens status | |||
| Phakic | 21 | 47 | .336 b |
| Pseudophakic | 11 | 17 | |
| Laterality | |||
| Right | 18 | 37 | .884 b |
| Left | 14 | 27 | |
| ERM on the fellow eye (%) | 6 (19) | 4 (6) | .079 c |
| Gauge of vitrectomy system | |||
| 20-gauge | 26 | 52 | 1.000 b |
| 23-gauge | 6 | 12 |
Surgical Procedure
PVD was induced more frequently in the highly myopic group (23/32 eyes, 72%) than in the control group (29/64 eyes, 45%) ( P = .014, χ 2 test). In the highly myopic group, preoperative B-scan ultrasonography indicated complete PVD in 20 of 32 eyes (63%); however, this was confirmed during surgery in only 9 of these eyes. It could be observed during surgery that the PVD was incomplete in the remaining 11 eyes, with the vitreous still attached to the posterior pole. In the highly myopic group, ILM was peeled in 23 of 32 eyes (72%), using BB in 15 cases (47%) and ICG in 8 cases (25%). In the control group, ILM was peeled in 39 of 64 eyes (61%), using BB in 27 cases (42%) and ICG in 12 cases (19%). There was no difference in terms of incidence of ILM peeling ( P = .291, χ 2 test) and use of staining dyes ( P = .744, χ 2 test) between the 2 groups.
Gas tamponade (SF 6 ) was used in 3 eyes in the case group and in 1 eye in the control group, because of the occurrence of superior retinal breaks during surgery. Air tamponade was used only in the control group, in 2 eyes.
Visual Outcomes
The mean duration of follow-up was 3.3 ± 1.5 years for the case group (median 3.1 years, range 1-6 years) and 3.6 ± 1.6 years for the control group (median 3.4 years, range 1-7.3 years) ( P = .608, Student t test).
Mean logMAR BCVA improved significantly, from 0.56 ± 0.20 and 0.54 ± 0.22 at baseline to 0.32 ± 0.20 ( P < .001, paired Student t test) and 0.28 ± 0.20 ( P < .001, paired Student t test) at 1 year, and 0.26 ± 0.19 ( P < .001, paired Student t test) and 0.22 ± 0.18 ( P < .001, paired Student t test) at 3 years, for the case and control groups, respectively ( Table 2 ). At the final examination, mean visual improvement was 0.29 ± 0.20 logMAR (corresponding to a mean increase of 2.9 lines) for the case group and 0.32 ± 0.25 logMAR (corresponding to a mean increase of 3.2 lines) for the control group ( Tables 2 and 3 ). The 2 groups did not differ statistically as regards either final postoperative BCVA ( P = .279, Student t test) or visual changes ( P = .526, Student t test). Interestingly, 66% of patients in the case group and 61% in the control group gained 2 or more lines in visual acuity at the end of the study, whereas only 1 patient in the case group and 2 patients in the control group lost more than 2 lines as compared with baseline visual acuity ( Table 3 ). In the 2 groups, the mean increase in visual acuity did not differ as a function of whether patients had undergone ILM peeling or not ( P = .388 in the case group and P = .468 in the control group, Mann-Whitney test).
| BCVA (LogMAR), Mean (SD) | Preoperative (n = 96) | 1 Year After Surgery (n = 96) | 3 Years After Surgery (n = 56) | Final Examination (n = 56) |
|---|---|---|---|---|
| Global | 0.55 (0.22) | 0.27 (0.20) | 0.24 (0.18) | 0.23 (0.18) |
| Cases (n = 32) | 0.56 (0.20) | 0.30 (0.20) | 0.28 (0.19) (n = 22) | 0.26 (0.19) (n = 22) |
| Controls (n = 64) | 0.54 (0.22) | 0.26 (0.20) | 0.23 (0.18) (n = 34) | 0.22 (0.18) (n = 34) |
| P Value | .618 a | .344 a | .212 a | .279 a |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


