15 Endoscopic Transsphenoidal Approach to the Sella
First performed by Schloffer over a hundred years ago, the transsphenoidal approach has become the standard approach for the removal of sellar lesions.1 In the early 1990s, Cushing2 and Hirsch3 went on to develop and refine the trans-sphenoidal approach. However, the approach fell out of favor after Cushing abandoned it for the transcranial approach. It was not until the 1950s and 1960s, with the addition of intraoperative fluoroscopy and the operating microscope, that Guiot4 and Hardy5 revitalized the transsphenoidal approach. The next major stage in the evolution of pituitary surgery began when Guiot et al6 introduced the use of endoscopy into transsphenoidal surgery in 1963. The endoscope was later reintroduced into clinical practice in the early 1990s.7 Since that time larger contemporary endoscopic series by Jho and Carrau8,9 and by Cappabianca et al10–13 have established the endoscopic, endonasal, transsphenoidal approach as an accepted technique and arguably new standard of care in pituitary surgery14 (Fig. 15.1).
For surgery within the sella, the endoscope has numerous advantages over the microscope. The endoscope produces a more panoramic view of the operative field.15 The endoscopic, endonasal approach may be associated with less postoperative swelling, avoidance of nasal packing, improved patient comfort, decreased length of stay, and less postoperative complications.16,17 With this approach, normal mucociliary transport is preserved because the nasal passage is not disrupted and the mucosa of the sphenoid sinus remains intact. Most importantly, the endoscope dramatically improves intra- and suprasellar visualization, especially with the use of angled scopes.18–20 Angled scopes can be advanced into the sphenoid sinus and sella to explore around corners to look for residual tumor, particularly laterally, against the medial wall of the cavernous sinus (Fig. 15.2). Similarly, extended approaches can be used to remove tumor in the suprasellar cistern and cavernous sinus.21–25 As a result the endoscopic approach has a lower incidence of residual tumor on postoperative magnetic resonance imaging (MRI) scans.26 The main disadvantage of the endoscope compared with the microscope is the lack of stereoscopic vision. However, this is not a significant limitation once the surgeon gains sufficient experience.18,27,28 In addition, three-dimensional stereoendoscopes are now available that provide stereoscopic vision.29–31
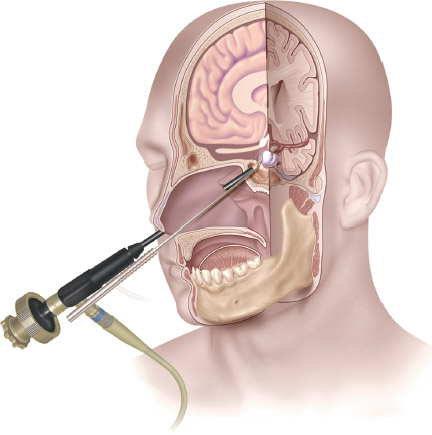
Fig. 15.1 The endonasal endoscopic approach to the sella is the most direct, minimally invasive approach to intrasellar lesions.
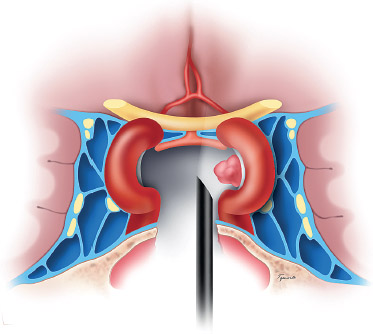
Fig. 15.2 Angled endoscopes can be used to visualize residual tumor in the lateral recesses of the sella, adjacent to the cavernous sinus, which would not be visualized with the microscope. (From Anand VK, Schwartz TH. Practical Endoscopic Skull Base Surgery. San Diego: Plu-ral Publishing; 2007. Reprinted with permission.)
 Indications
Indications
The endoscopic, endonasal, transsphenoidal approach is suitable for pituitary tumors, both microadenomas and macroadenomas. Other lesions that are confined to the sella or that enlarge the sella and extend into the suprasellar cistern, such as some craniopharyngiomas and Rathke cleft cysts, are also suitable candidates for this approach (Fig. 15.3).
Surgery continues to be the standard of care for hormone-producing pituitary tumors, such as ones that cause Cushing’s disease or acromegaly, and for nonsecreting pituitary macroadenomas that cause either optic nerve compression or have documented growth.28,32 For asymptomatic nonsecreting pituitary tumors that are stable in size and not compressing the optic chiasm, either conservative management with expectant observation or surgical resection should be considered, depending on the situation. Prolactinomas are not routinely removed as a first-line therapy. However, when surgical resection is considered, a preoperative prolactin level >250 μg/L has been shown to predict a lower probability of cure from surgery than is obtained with medical management. If the prolactin level is <250 μg/L, and the tumor is a microadenoma with no cavernous sinus involvement, then the chance of surgical cure is almost 90%.33 Other factors that support surgical debulking include significant visual loss, apoplexy, and resistance to medical treatment.
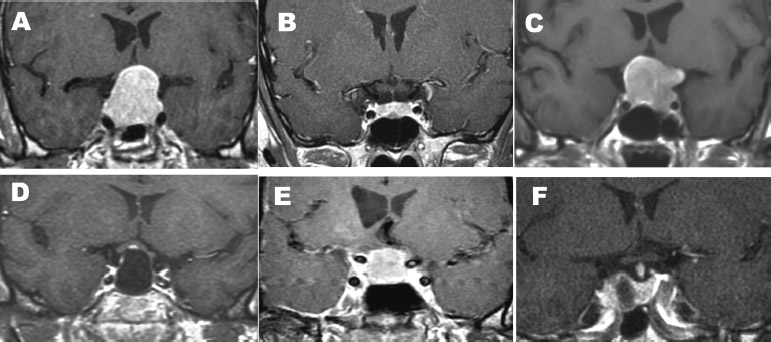
Fig. 15.3 Multiple types of intrasellar and intra- and suprasellar lesions are suitable for the endonasal, endoscopic transsphenoidal, transsellar approach: (A) pituitary macroadenomas with suprasellar extensions, (B) pituitary microadenomas, (C) macroadenomas with moderate lateral extension, (D) Rathke cleft cysts, and (E) intrasellar craniopharyngiomas. A unilateral approach can be used for small tumors such as that shown in B. Note that in this case, a right-sided approach can be used, and only half of the sphenoid sinus needs to be opened because there is a septum that marks the lateral extent of the tumor. A unilateral left-sided approach can be used for tumors that are on the right side of the sella, such as the adenoma shown in F. (From Anand VK, Schwartz TH. Practical Endoscopic Skull Base Surgery. San Diego: Plural Publishing; 2007. Reprinted with permission.)
The role of surgical intervention for Rathke cleft cysts is somewhat controversial. Hypopituitarism is improved in only approximately 20 to 30% of patients, but symptoms of optic chiasm compression improve in 98%.34,35 In addition, headaches are relieved in approximately 90% of patients and can be an indication for surgery.36 Although some authors recommend aggressive removal of the cyst wall to prevent recurrence, this strategy increases the risk of postoperative hypopituitarism.37 Drainage and subtotal cyst wall removal is a less risky procedure and prevents recurrence in up to 84% of cases.13,34–36 Endoscopic, endonasal resection of Rathke cleft cysts is an effective minimally invasive approach.13,35 Evidence of squamous metaplasia in the cyst wall predicts recurrence, as does residual cyst on postoperative imaging.34,36
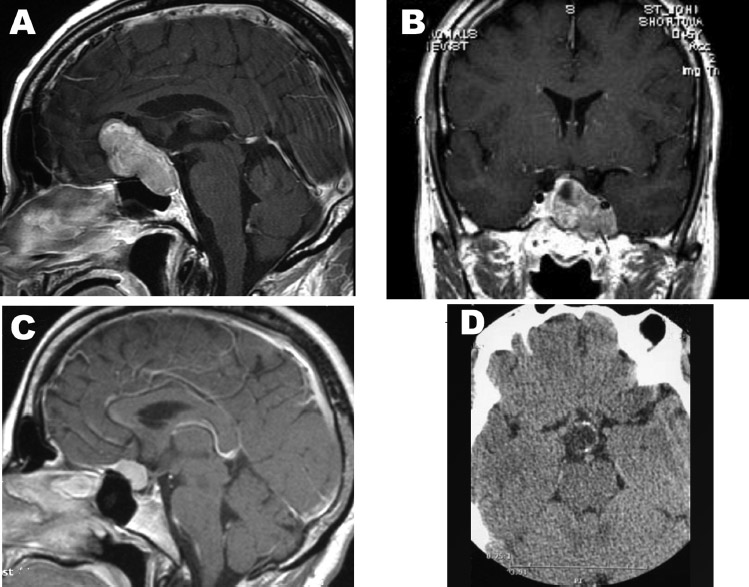
Fig. 15.4 Considerations in choosing the transsellar approach. (A) A pituitary macroadenoma with significant suprasellar extension that extends anteriorly. This tumor cannot reliably be completely removed with a sellar approach but requires a more extensive approach such as the endonasal, endoscopic, transplanum, transtuberculum approach. In some cases removal of the posterior ethmoids is required. (B) A tumor with significant cavernous sinus extension cannot be completely removed through a transsellar approach. If the goal is to remove the entire tumor, a transcavernous approach is required. However, a limited resection followed by radiosurgery to the cavernous sinus remnant may be more prudent in this situation. (C) Meningiomas of the tuberculum sellae can extend into the sella and mimic pituitary adenomas. These tumors require a larger transtuberculum opening for removal. (D) Calcification is the hallmark of the craniopharyngiomas, which can be removed only through a transsellar approach if there is significant involvement of the sella. (From Anand VK, Schwartz TH. Practical Endoscopic Skull Base Surgery. San Diego: Plural Publishing; 2007. Reprinted with permission.)
The standard treatment for craniopharyngiomas has been surgery for many years. The transsphenoidal approach is appropriate when an enlarged sella is present. Purely intrasellar craniopharyngiomas are rare and occur only 4% of the time. Intra-or suprasellar lesions occur 21% of the time, and primarily suprasellar lesions are observed 75% of the time.38 Controversy remains regarding conservative versus aggressive resection.39 Aggressive surgery is associated with better control but also greater morbidity secondary to hypothalamic and pituitary injury. The trans-sphenoidal approach has been broadened to lesions other than purely intrasellar craniopharyngiomas.40 Application of the endoscope to the transsphenoidal approach for craniopharyngiomas takes advantage of the less invasive nature of the approach with improved intra-and suprasellar visualization.41
 Contraindications and Considerations
Contraindications and Considerations
The endonasal, endoscopic, transsphenoidal approach to the sella can be used to resect the majority of suprasellar tumors that arise within and enlarge the sella by using straight and angled endoscopes. However, for exceptionally large tumors with a considerable amount of anterior suprasellar extension, we do not advise a direct transsellar method (Fig. 15.4A). In such cases, a transplanum, transtuberculum approach to enlarge the bony and dural opening can be applied, which provides a direct view of the suprasellar component of the tumor to allow a safe and maximal resection.
Another major consideration is the lateral extent of the lesion, into the cavernous sinus (Fig. 15.4B). Only when the tumor completely encircles the carotid artery can cavernous sinus involvement be guaranteed (Fig. 15.5B). Often, compression of the medial wall can mimic invasion (Fig. 15.5C,D), and in these cases the entire tumor can be removed through an endonasal, transsellar approach. Lateral extension into the cavernous sinus is not an absolute contraindication, but the decision should be made preoperatively whether the goal of surgery is to decompress the optic apparatus by resecting sellar and suprasellar components of tumor or to remove intracavernous tumor by opening the cavernous sinus. If the goal is to remove tumor from the cavernous sinus, there are two options. In some circumstances, the tumor passes through a fenestration in the medial wall of the cavernous sinus. By using angled endoscopes and instruments, this opening can be followed from within the sella into the cavernous sinus to remove intracavernous tumor. The alternative method is a transethmoidal or transpterygoidal approach, which provides a more direct exposure of the parasellar region.42 In cases when intracavernous tumor is intentionally left, stereotactic radiation therapy can be used to control growth as long as the tumor mass is at least 4 to 5 mm from the optic apparatus.43
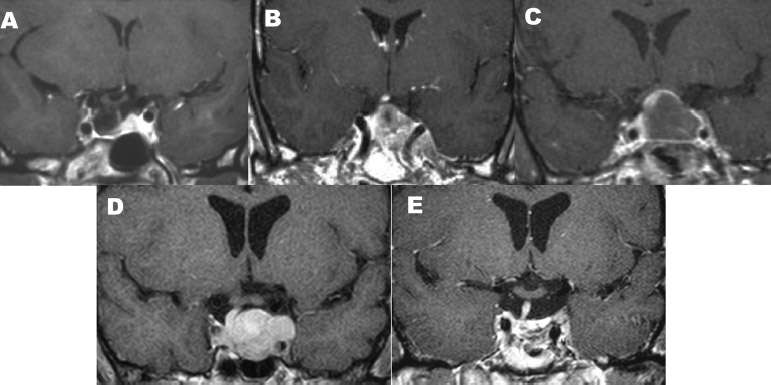
Fig. 15.5 Anatomic considerations as demonstrated by preoperative magnetic resonance imaging (MRI). (A) Empty sella syndrome can mimic a Rathke cleft cyst or cystic pituitary adenoma. (B) True cavernous sinus invasion, in which the tumor encircles the cavernous sinus. (C) This macroadenoma compresses the cavernous sinus but does not actually invade. Note the normal pituitary gland compressed superiorly. (D) Recurrent tumor following radiosurgery shows the possibility of cavernous sinus invasion on the left. (E) Postoperative films reveal that the entire tumor was removed via an endonasal, endoscopic transsellar approach. (From Anand VK, Schwartz TH. Practical Endoscopic Skull Base Surgery. San Diego: Plural Publishing; 2007. Reprinted with permission.)
 Preoperative Considerations
Preoperative Considerations
Other lesions such as meningiomas, germinomas, hypothalamic hamartomas, and basilar tip aneurysms can be mistaken for pituitary macroadenomas. Such lesions must be ruled out preoperatively. In comparison to meningiomas, pituitary tumors typically enlarge the sella and lack a dural tail. Furthermore, meningiomas with cavernous sinus extension tend to constrict the carotid artery, whereas pituitary tumors do not. Calcifications that are more commonly present in either craniopharyngiomas or meningiomas can be identified with computed tomography (CT) scanning (Fig. 15.4D). If an aneurysm is suspected, an angiogram should be performed.
All patients with pituitary adenomas should be evaluated for endocrine abnormalities. Patients should be questioned about symptoms of endocrine abnormalities, which include amenorrhea; galactorrhea; impotence or decreased libido; arthropathy; hyperhidrosis; changes in facial features; changes in foot, hand, and hat size; weight gain or loss; hypertension; polyuria; polydipsia; hypotension; hirsutism; bruising; stretch marks; and development of adipose deposits. A comprehensive endocrinologic evaluation of the hypothalamic-pituitary axis should be undertaken with each patient. Patients with chiasmal compression are evaluated with formal visual field testing. Previously instituted medical therapy such as dopaminergic or somatostatin analogues do not need to be stopped before surgery, as these agents do not appear to decrease the efficacy of surgery.44 In the case of stereotactic radiosurgery, medical therapy should be stopped before treatment as it appears to be more effective if the tumor is more metabolically active.45,46 Identification of the pituitary microadenoma on preoperative imaging is critical. One clue is that the stalk is generally shifted away from the tumor, although this is neither 100% sensitive nor specific.47 Pituitary adenomas are typically hypointense on T1-weighted MRI (Fig. 15.3) and variable in appearance on T2-weighted imaging. Because the normal pituitary gland takes up gadolinium rapidly, microadenomas appear non-enhancing on early postcontrast images and isointense at later time points. If imaging does not demonstrate a tumor in a patient with possible Cushing’s disease, bilateral petrosal sinus sampling after administration of corticotropin-releasing hormone can be performed to measure adrenocorticotropic hormone (ACTH) levels to confirm the diagnosis. However, the reliability of this test in lateralizing the tumor is questionable.
Rathke cleft cysts are benign epithelium-lined cysts, probably arising from the remnants of a Rathke pouch (Fig. 15.3D). Preoperatively, mucoid Rathke cleft cysts are hyperintense on T1- and hypointense on T2-weighted MRI. Serous cysts are isointense to cerebrospinal fluid (CSF), as compared with cysts that contain cellular remnants, which can resemble more solid tumors.47 Craniopharyngiomas arise from squamous epithelial remnants of a Rathke pouch in the region of the pars tuberalis. These tumors commonly extend into the sella. Adamantinomatous craniopharyngiomas, often found in children, generally contain cysts that are hypointense on T1- and hyperintense on T2-weighted MRI with rim enhancement (Fig. 15.3E). Characteristically, these lesions have a calcified ring and a hyperintense nodular component (Fig. 15.4D). Squamopapillary craniopharyngiomas, more common in adolescents and young adults, can extend further into the third ventricle and are both cystic and solid.
One important diagnosis to rule out preoperatively is empty sella syndrome, as these patients are likely to have previously unrecognized increased intracranial pressure and subsequently a high likelihood of postoperative CSF leak. Empty sella can be caused by a dehiscent sellar diaphragm, increased intracranial pressure, or prior pituitary surgery. It can mimic a Rathke cleft cyst (Fig. 15.5A). The sella may even be enlarged. One distinction is the absence of optic nerve compression in empty sella syndrome. In fact, the chiasm can even herniate into the sella.
Careful preoperative evaluation of the nasal and paranasal sinuses, in particular the sphenoid sinus anatomy, is critical (Fig. 15.6). We generally obtain a fine-cut CT preoperatively, which is then used for intraoperative navigation because MRI scanning alone is usually not adequate. We take note of the curvature of the middle turbinate and deviations of the septum. The endoscopic view may be obstructed by a deviated or bowed septum, precipitating a change to the side of the approach. Alternatively, a deviated septum can be repaired during the approach. Concha bullosa, or a pneumatized middle turbinate, may occur in as many as 80% of patients, depending on the definition of the term.48 The pattern of sphenoid sinus pneumatization is critically important. Three patterns of pneumatization have been described (Fig. 15.6A–C). In the most common “sellar” variant (75%), the sinus is aerated in front of and below the sella. This variation is the most amenable to the transsphenoidal approach, as the anterior and inferior walls of the sella are easily accessed. In the “presellar” variant (24%), aeration permits access only to the front of the sella. To provide adequate visualization, the clivus is drilled down below the sella. In the last “conchal” variant (<1%), there is minimal or no aeration of the sphenoid. The transsphenoidal approach requires extensive drilling of the clivus and is significantly aided by stereotactic navigation in this situation. Finally, detailed analysis of the intrasphenoidal anatomy preoperatively is important. Intrasphenoidal septa are helpful landmarks for confirming the locations of the carotid arteries and small microadenomas intraoperatively (Fig. 15.6D). Midline septa are often drilled away on the approach. However, more laterally located septa can occasionally be left in place if they do not interfere with the operative approach.
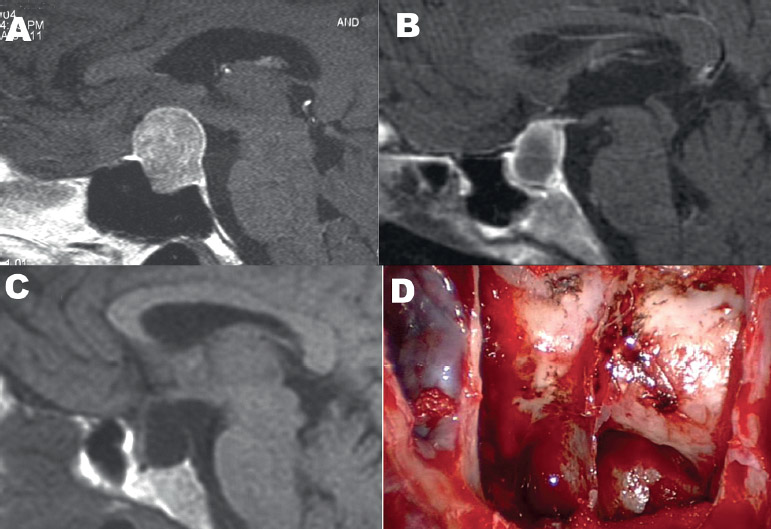
Fig. 15.6 Sphenoid sinus anatomy. (A) “Sellar” aeration of the sphenoid sinus. (B) “Presellar” aeration of the sphenoid sinus. (C) “Conchal” aeration of the sphenoid sinus. (D) Sphenoid septations can be useful landmarks in mapping out the location of the pituitary tumor, the carotid arteries, and the cavernous sinuses when compared with a preoperative MRI or computed tomography scan. (From Anand VK, Schwartz TH. Practical Endoscopic Skull Base Surgery. San Diego: Plural Publishing; 2007. Reprinted with permission.)
 Patient Positioning and Preparation
Patient Positioning and Preparation
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


