Objective
To define morphologic features of polypoidal choroidal vasculopathy (PCV) using en face images from swept-source optical coherence tomography (SS OCT).
Design
Prospective cross-sectional study.
Methods
The study included 10 eyes from 6 patients with PCV and 10 eyes from 5 age-matched normal subjects. All subjects were prospectively scanned with a prototype SS OCT system. A motion correction algorithm was applied to correct and merge scans into a single volumetric dataset. En face images were generated at intervals of 4.13 μm (1 pixel) relative to the Bruch membrane.
Results
Age ± standard deviation for the normal group was 62.4 (±12.1) years and for the PCV group was 68.3 (±5.2) years. En face SS OCT imaging of PCV eyes demonstrated the relationship between larger pigment epithelial detachments (PEDs) and small adjoining PEDs that correlated with the polypoidal lesions seen on indocyanine green angiography in all PCV eyes. En face SS OCT demonstrated choroidal vascular abnormalities in 7 out of 7 eyes with PCV, and in 2 out of 3 enrolled fellow eyes in patients with unilateral PCV. Out of 7 PCV eyes, focal choroidal vascular dilation was noted in 3 eyes and diffuse choroidal vascular dilation was noted in 1 eye. In addition, a branching vascular network was noted above the Bruch membrane in 1 eye, below the Bruch membrane within the choriocapillaris in 1 eye, and in the larger choroidal vascular layer in 1 eye.
Conclusions
En face SS OCT provides an in vivo tool to visualize the pathologic features and the choroidal vasculature in PCV.
Polypoidal choroidal vasculopathy (PCV) is a term coined by Yannuzzi in 1982 (Yannuzzi LA. Idiopathic polypoidal choroidal vasculopathy. Presented at Macula Society Meeting 1982; Miami, Florida, USA). PCV is a disease characterized by multiple, recurrent serosanguineous detachments of the retinal pigment epithelium (RPE) and neurosensory retina, associated with secondary bleeding or leakage from a branching vascular network. PCV likely comes in 2 varieties: a subset of choroidal neovascularization from a variety of causes, but most commonly attributable to neovascular age-related macular degeneration (AMD); or a distinct disease from AMD that is typically found in mostly darkly pigmented younger individuals, and without other fundus findings typical of AMD.
Indocyanine green angiography (ICGA) is important in securing the diagnosis of PCV, because it has the ability to demonstrate polypoidal lesions and branching vascular networks beneath the RPE that cannot be visualized using standard fluorescein angiography. The use of optical coherence tomography (OCT) has become ubiquitous among retina specialists and is quite useful in the setting of PCV. The OCT features of PCV have been described and correlated with ICG findings in multiple studies. A topic of interest has become the identification and characterization of polypoidal lesions and abnormal vascular networks using en face OCT images. Utilizing a spectral-domain OCT (SD OCT) ophthalmoscope (C7; Nidek, Gamagori, Japan), investigators had reported the en face findings in PCV eyes. However, the choroid was not clearly visualized because of the signal loss beyond the RPE–Bruch membrane complex, which is typical of SD OCT. Swept-source OCT (SS OCT) provides long wavelength, reduced attenuation, and longer imaging range when compared with SD OCT. These properties of SS OCT allow better penetration and improved signal strength in the choroid. Hong and associates used a prototype swept-source-based high-penetration Doppler OCT to present comprehensive visualization of the 3D structure of PCV, including feeder vessels and branching vascular networks; however, the underlying anatomy of the choroidal vasculature was not analyzed in detail. Our study presents the en face imaging features of the retina and choroid in PCV patients using a prototype 1050-nm-wavelength SS OCT instrument.
Methods
We prospectively enrolled patients with PCV at the New England Eye Center at Tufts Medical Center between December 1, 2013 and May 31, 2014, and age-matched healthy volunteers. Study protocols were approved by the Institutional Review Board of Tufts Medical Center and Massachusetts Institute of Technology and were in accordance with the Health Insurance Portability and Accountability Act. The research adhered to the tenets of the Declaration of Helsinki for research involving human subjects.
Signed informed consent was obtained prior to SS OCT image acquisition. Subjects were examined with a prototype SS OCT system operating at 1050 nm for enhanced choroidal penetration. The details of this prototype machine have been previously reported and validated. Briefly, the system employs a commercially available 100 kHz wavelength-swept semiconductor laser (Axsun Technologies Inc, Billerica, Massachusetts, USA) with a sweep bandwidth of ∼100 nm, providing a tissue axial resolution of 6 μm. The light incident on the eye was 1.9 mW, which is consistent with the American National Standard Institute (ANSI) standards for safe ocular exposure. Three-dimensional 6 × 6-mm scans with 400 × 400 axial scans or 12 × 12-mm scans with 500 × 500 axial scans were obtained, each acquired within 1.7 seconds. For each subject, at least 2 orthogonal volumetric scans were acquired, processed with software for motion correction, and merged into a single volumetric dataset to enhance the signal.
A semiautomatic algorithm with manual correction capabilities was developed and was used to flatten the scans using the Bruch membrane as a reference, in order to generate a reference surface for en face display. En face images through the retina, RPE, and choroid were extracted at varying depths every 4.13 μm (1 pixel, corresponding to the digital resolution of OCT images) using the Bruch membrane as a reference surface. Image J Software (National Institutes of Health, Bethesda, Maryland, USA; available at http://rsb.info.nih.gov/ij/index.html ) was used to facilitate an en face image view in an orthogonal 3-dimensional fashion, where B-scans and C-scans could be precisely correlated.
Using en face SS OCT imaging, the transition from the fine homogeneous choriocapillaris (CC) into the choroidal vascular layer (CV), where the choroidal vessels became distinct, was apparent. The most representative C-scan for the transition between CC and CV layers was identified. Using this, an estimate of the CC thickness was performed by counting the number of axial pixels between the RPE and the transition zone and then multiplying by the pixel dimension (4.13 μm). The lumen of the choroidal vessels started to enlarge from the transition zone going through the inner choroid toward the outer choroid. The choroidal-scleral interface was easily identified on en face display by displacement of the choroidal vasculature with the white sclera. Similarly, the CV thickness was calculated by counting the axial pixels in between the transition zone and the choroidal-scleral interface and performing the above-mentioned calculation. Subsequently, the total choroidal thickness was calculated by adding the CC and CV thicknesses.
Morphologic features of PCV were identified by 2 independent authors (T.A. and D.F.) using a standard protocol to analyze color photographs, fundus autofluorescence, fluorescein angiography, ICGA, and SS OCT images. Disagreement between the 2 observers was resolved by open adjudication.
Results
Ten eyes from 6 Asian patients with PCV and 10 eyes from 5 age-matched normal subjects (1 Asian and 4 white subjects) were enrolled in a prospective cross-sectional study. Age ± standard deviation was 68.3 ± 5.2 years in the PCV group and 62.4 ± 12.1 years in the normal group. Visual acuity ranged between 20/30 and 20/200 in the PCV group, and was 20/30 or better in the normal group. One PCV patient had history of enucleation of the contralateral eye because of massive hemorrhage. One patient with unilateral peripheral PCV lesion refused to have the fellow eye scanned. Five PCV eyes had received no treatment and 2 eyes underwent intravitreal injections with anti–vascular endothelial growth factor agent in the past. ICGA identified polypoidal lesions in all 7 PCV eyes. The fellow eyes in 3 patients with unilateral PCV were scanned with SS OCT as well. Table 1 further demonstrates the demographics of patients with PCV.
| Subject | Orientation | Age | VA | Treatment | Location of PED | Choroidal Vasculature |
|---|---|---|---|---|---|---|
| 1 | OD | 63 | 20/30 | None | Superior periphery | Branching vascular network above Bruch membrane |
| 2 | OS | 69 | 20/200 | None | Juxtafoveal | Focal dilation |
| 3 | OS | 61 | 20/200 | None | Peripapillary | Branching vascular network in CV |
| 4 | OD | 74 | 20/70 | None | Extrafoveal | Focal dilation |
| OS | 20/50 | None | Juxtafoveal | Branching vascular network in CC | ||
| 5 | OS | 71 | 20/50 | Anti-VEGF | Extrafoveal | Diffuse dilation |
| 6 | OD | 72 | 20/30 | Anti-VEGF | Subfoveal | Focal dilation |
| Fellow eyes in patients with unilateral PCV | ||||||
| 2 | OD | 69 | 20/50 | NA | NA | Focal dilation |
| 5 | OD | 71 | 20/50 | NA | NA | Diffuse dilation |
| 6 | OS | 72 | 20/20 | NA | NA | No abnormalities |
Qualitative Analysis
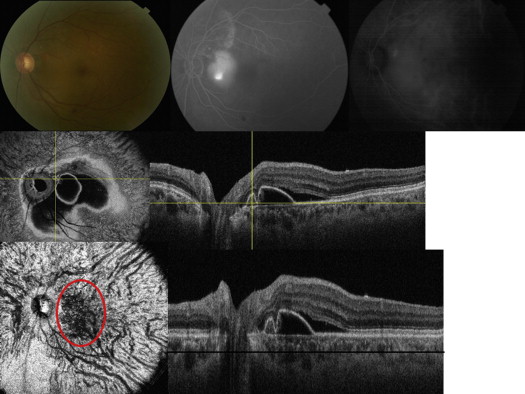
En face SS OCT imaging allowed virtual sectioning of the pathologic area of interest in consecutive scans through the retina, RPE, and choroid. One or more dome-shaped pigment epithelial detachments (PEDs) were visualized on SS OCT cross-sectional scans in all 7 PCV eyes. On en face SS OCT scans anterior to the Bruch membrane, PEDs were visualized as a highly reflective oval or circular line of RPE, which corresponded to the outline of the PED. Irregularities in the outline of larger PEDs were identified as small adjoining PEDs and correlated with polypoidal lesions seen on ICGA in all 7 PCV eyes ( Figures 1–4 ). The material within the smaller PEDs representing polypoidal lesions was typically more highly reflective than the material in the larger adjoining PED ( Figure 1 ).
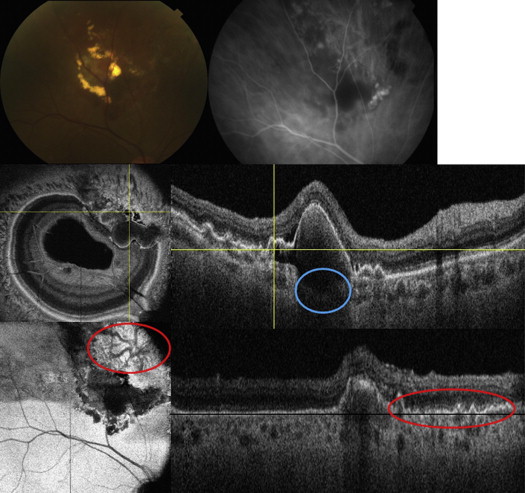
Cross-sectional SS OCT imaging demonstrated the “double layer sign” adjoining more highly elevated PEDs and polyps in 5 out of 7 PCV eyes ( Figures 1,4 , and 5 ). The “double layer sign” was identified in previous studies as 2 hyper-reflective lines representing the RPE and Bruch membrane, where an abnormal vascular network resides. Fibrotic PEDs indicating chronicity were identified in 2 out of 7 PCV eyes ( Figures 4 and 5 ). The feeder vessel was visualized in 3 out of 7 PCV eyes as a thin hypo-reflective band that started in the choroid and penetrated the Bruch membrane to reach the small PED where the polypoidal lesion was situated ( Figures 5 and 6 ).
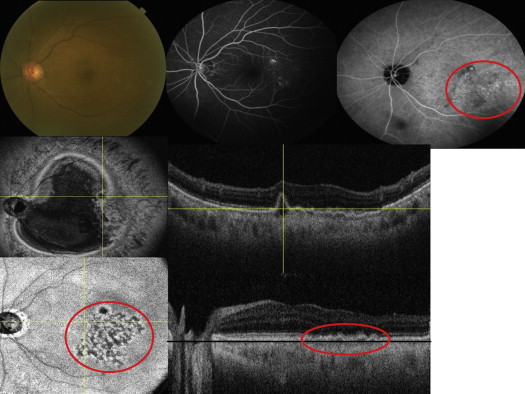
En face SS OCT demonstrated choroidal vascular abnormalities in 7 out of 7 eyes with PCV and in 2 out of 3 enrolled fellow eyes in patients with unilateral PCV. Out of 7 PCV eyes, focal choroidal vascular dilation was noted in 3 eyes while diffuse choroidal vascular dilation was noted in 1 eye. In addition, a branching vascular network was noted above the Bruch membrane in 1 eye, below the Bruch membrane (within the choriocapillaris) in 1 eye, and in the deeper choroidal vascular layer in 1 eye. The branching vascular networks above and below the Bruch membrane correlated with the double layer sign seen on cross-sectional scans ( Figures 1 and 3 ). Out of 3 fellow eyes that were examined in patients with unilateral PCV, focal choroidal vascular dilation was noted in 1 eye ( Figure 7 ), diffuse choroidal vascular dilation was noted in 1 eye, and no obvious choroidal vascular abnormalities were noted in 1 eye.
Quantitative Analysis
The CC, CV, and total choroid mean thicknesses (around the area of pathology) in PCV eyes were 41.3 μm, 274.0 μm, and 315.3 μm, respectively. The subject with superior peripheral PCV lesion was excluded from the comparison, since the choroidal thickness measurements were taken around the peripheral PCV lesion. The CC, CV, and total choroidal mean thickness in the fellow eyes were 42.7 μm, 293.2 μm, and 335.9 μm, respectively. The fellow-eye choroidal thickness measurements were not significantly different from the PCV affected eyes, and the P values for CC, CV, and total choroidal thicknesses were, respectively, .64, .74, and .73. Table 2 further demonstrates these measurements.
| Subject Number | Orientation | CC | CV | Total | Location of PED |
|---|---|---|---|---|---|
| 1 | OD | 41.3 | 210.6 | 251.9 | Superior periphery |
| 2 | OS | 41.3 | 268.5 | 309.8 | Juxtafoveal |
| 3 | OS | 45.43 | 285 | 330.4 | Peripapillary |
| 4 | OD | 37.2 | 251.9 | 289.1 | Extrafoveal |
| OS | 45.4 | 210.6 | 256.1 | Juxtafoveal | |
| 5 | OS | 41.3 | 392.4 | 433.7 | Extrafoveal |
| 6 | OD | 37.2 | 235.4 | 272.6 | Subfoveal |
| Average (excluding subject 1) | 41.3 | 274.0 | 315.3 | ||
| Fellow eyes in patients with unilateral PCV | |||||
| 2 | OD | 37.3 | 243.8 | 280.8 | NA |
| 5 | OD | 45.4 | 421.3 | 466.7 | NA |
| 6 | OS | 45.4 | 214.8 | 260.2 | NA |
| Average | 42.7 | 293.2 | 335.9 | ||
| P value | .64 | .74 | .73 | ||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


