8
Electrophysiology and Device Telemetry
Paul J. Abbas, Carolyn J. Brown, and Christine P. Etler
The performance of individuals receiving cochlear implants has improved significantly over the course of the past two decades. This improvement is likely the result of several factors including advances in cochlear implant technology, changes in patient criteria (both age and degree of hearing loss), and advances in fitting procedures. However, despite these advances, the range of postimplant outcomes is still quite large. The variability in performance among cochlear implant users undoubtedly reflects several factors including the status of the auditory nerve and central auditory system, electrode placement, and the integrity of the implanted electronics. Although imaging techniques have improved significantly in the past decade and can be used to assess electrode placement, such techniques have not proven to be effective in determining neural survival, assessing device function, or predicting postimplant performance (Skinner et al, 2002; Wang et al, 1996, 2005). It is possible, however, to use the telemetry systems available with present-day cochlear implants to assess the function of the implanted components of the cochlear implant. Additionally, both the Nucleus (Cochlear Corporation; Sydney, Australia) and Advanced Bionics (Valencia, CA) devices have neural telemetry systems that allow the user to record the response of the auditory nerve to electrical stimulation. This chapter reviews the information about how both devices function as well as the status of the peripheral auditory nervous system that can be obtained using these telemetry systems. Specifically, this chapter discusses how telemetry systems can be used to assess device function, the specific methodology used to measure electrically evoked compound action potentials (ECAPs), and the application of these measures both in terms of direct clinical applications and as a research tool.
♦ Techniques to Assess Device Function
Cochlear implants are designed to last for the life of the recipient. Unfortunately, the internal components of the cochlear implant can and do fail. Consequently, clinicians need to assess and monitor the function of the implanted electronics on a regular basis. Today, all three of the major cochlear implant manufacturers (Cochlear Corporation; Advanced Bionics Corporation; and Med-El Corporation, Innsbruck, Austria) have incorporated telemetry systems that can be used to measure the impedance of the intracochlear electrodes. Additionally, surface electrodes and standard evoked potential recording techniques can be used to evaluate the function of the internal electronics for a range of different stimulation conditions. The following sections discuss the information about device function that can be obtained and how that information can be used in clinical practice.
Telemetry Systems
All of the cochlear implants that are available today have built-in telemetry systems that allow limited information about the function of the internal device to be assessed directly. In general, these telemetry systems work by sampling and digitizing the voltage generated on the internal electrodes during stimulation. This information is then transmitted back through the computer interface to the programming software. No external recording electrodes are needed and the stimulation levels used to assess device function are typically low enough that the patient does not hear the stimulus.
It is possible, using the standard programming software, to measure the impedance of the implanted electrodes in the Nucleus, Advanced Bionics, and Med-El implants in a monopolar stimulation mode, that is, in each intracochlear electrode relative to an external reference. Monopolar impedance measures can be used to identify open circuit electrodes but do not allow identification of a malfunction where two intracochlear electrodes are shorted. To identify electrodes that are shorted to each other, either the impedance must be measured in a common ground stimulation mode, or testing must be conducted for all possible bipolar electrode pairs. The clinical programming software used with both the Nucleus and Med-El devices does flag short-circuited electrodes. Identifying shorts between intracochlear electrodes for the Advanced Bionics implant, however, requires access to specialized electrical field imaging (EFI) software that allows the user to sample the electrical field across the length of the cochlea during stimulation.
It is also possible to use the telemetry systems of the cochlear implant to assess whether or not compliance voltage has been reached. Every current source has some limitation in maximum output that is dependent, in part, on the impedance of the electrodes and, in part, on battery capacity. That maximum level is called the compliance voltage. In order for the cochlear implant user to have adequate loudness growth, it is important that all stimulation provided by the speech processor stays below this compliance voltage. The software used to program the Nucleus, Advanced Bionics, and Med-El speech processors includes flags that can alert the user that compliance voltage has been reached so appropriate adjustments in the stimulation parameters (e.g., increasing pulse duration) can be made for the individual patient.
Average Electrode Voltages
A second technique that can be used to assess function of the internal components of the cochlear implant is to use surface electrodes to record the voltages generated when one of the intracochlear electrodes is stimulated. These potentials have been referred to in the literature as averaged electrode voltages (AEVs). When the intracochlear electrodes are activated, the voltages associated with stimulation that are measured at the scalp are large and can be recorded relatively easily and quickly using standard evoked potential recording systems. These are not physiologic potentials; rather, they are far-field recordings of the stimulus artifact that accompanies stimulation.
AEVs can be measured with any of the available cochlear implant systems but have proven to be particularly useful with older implants, like the Nucleus CI22 device, where it is not possible to measure intracochlear electrode impedance directly. The primary clinical application for AEVs is to monitor device function in cochlear implant users (Heller et al, 1991; Kileny et al, 1995; Mens et al, 1994, 1995). Several investigators have published reports demonstrating how abnormal electrode impedance or function can be inferred from examination of AEV recordings (Kileny et al, 1995; Mahoney & Proctor, 1994; Mens et al, 1994). A recent study by Hughes et al (2004) demonstrated that AEVs recorded in response to stimulation in a common ground electrode configuration could consistently detect both open circuits and short circuits among electrodes. At a simple level, these results suggest that AEVs could serve as an efficient test of electrode integrity for implants without impedance telemetry systems.
It should be understood, however, that the morphology of the AEV recording is affected by more than simply the impedance of the stimulated electrode. The size and general shape of the recording that is made from surface electrodes is affected by many different factors including current level, mode of stimulation, spatial orientation of the stimulating and recording electrodes, the current path within the tissue, as well as changes in position and tissue growth over time. It is precisely because the AEV is a reflection of more than simply the electrode impedance that recording this potential may be useful even in patients who use implants that are equipped with impedance telemetry systems. Although electrode impedance may be normal for a given electrode, other factors, such as unusual tissue growth or electrode position, may influence the ability of an implant to effectively stimulate the neural population. Malfunction of the internal components of the device can also result in a broad range of percepts ranging from complete lack of stimulation to intermittent overstimulation or a subtle change in the level or quality of stimulation provided by the implant. Such failures may not be common but are certainly not insignificant. Two recent studies (Franck and Shah, 2004; Hughes et al, 2004) also demonstrated that abnormally functioning electrodes can be missed with clinical impedance measures, but correctly identified with AEVs. Such data suggest that in addition to monitoring electrode impedance using the standard telemetry systems available with the clinical software, baseline AEV measures should be obtained some time during the first few months following implantation. For pediatric patients or other cochlear implant recipients who may not be reliable reporters of changes in sound quality over time, it might be advisable to measure AEVs on a routine basis to monitor electrode function over time.
♦ Techniques to Assess the Response of the Auditory System
Over the course of the past several decades, several electrophysiologic tools have been developed and used with acoustic stimulation to assess the response of the auditory system at various levels of neural processing. These include the whole nerve action potential, the auditory brainstem response, middle and long latency responses, as well as more cognitively related brain potentials, such as the mismatch negativity (MMN) response and event-related cortical responses such as the P-300 response. Counterparts to each of these potentials have been recorded in response to electrical stimulation. Studies using the electrically evoked compound action potential (ECAP) (Brown et al, 1990), the electrically evoked auditory brainstem response, (EABR) (van den Honert & Stypulkowski, 1986), the electrically evoked middle latency response (EMLR) (Kileny & Kemink, 1987), MMN (Kraus et al, 1993), the N1-P2 complex (Sharma et al, 2002), and the P-300 (Kaga et al, 1991) have been reported. In addition, there have been several studies published describing techniques for using electrical stimulation to evoke the stapedius reflex (Battmer et al, 1990).
In general, all of these electrically evoked potentials have characteristics that are very similar to their acoustically evoked counterparts. On a global level this suggests that the basic organization of the auditory system in implant users is intact. Electrical stimulation, however, bypasses the normal cochlear processes including the compressive effects of the basilar membrane mechanics and the hair cell and synaptic process. The resulting excitation of the auditory nerve is known to have shorter latency, greater synchrony, and steeper response growth in response to electrical stimulation as compared with acoustic stimulation (Kiang & Moxon, 1972). The characteristics of evoked potentials measured with implant patients reflect these differences in the mode of stimulation. Response latencies of early potentials (ECAP and EABR) tend to be shorter, response amplitudes tend to be greater, and the growth of response with the level tends to be faster than is observed with acoustic stimulation (Brown et al, 1990; van den Honert & Stypulkowski, 1986).
Initially, most of the research that was published exploring ways in which electrophysiologic techniques could be adapted for use with patients with cochlear implants focused on the EABR and the EMLR. Recording either of these potentials requires the application of surface recording electrodes, the use of sophisticated recording instrumentation, and a quiet subject. Although several investigators described clinical applications for these electrically evoked responses, many clinical settings did not routinely record these responses because of these practical limitations. In the mid-1990s, Cochlear Corporation introduced a telemetry system that allows the user to record a response from the auditory nerve by using one of the electrodes in the intracochlear array. The telemetry system used to record the response of the auditory nerve that was introduced with the Nucleus 24M and 24R implants has been termed neural response telemetry (NRT). A similar system was later introduced by Advanced Bionics Corporation with the Clarion CII and HiResolution 90k implants and is referred to as neural response imaging (NRI). Although the current generation of Med-El cochlear implants (the Combi 40+) are not equipped with neural response telemetry systems, the next generation Med-El implant (the PULSARci) will have a comparable recording system that they refer to as auditory nerve response telemetry (ART). All of these telemetry systems include software to control the stimulation and recording parameters as well as methods for averaging, filtering, and analyzing the recorded waveforms. The response that is recorded by each of these systems has been called the electrically evoked compound action potential (ECAP or EAP) and compared with the electrically evoked auditory responses recorded previously (the EABR or EMLR), the ECAP is a very peripheral measure of the response of the auditory system to electrical stimulation.
It is often difficult and time-consuming to program the speech processor for very young cochlear implant recipients, and it is for these patients in particular that having the ability to measure the response of the auditory nerve to electrical stimulation has proven useful. There are several reasons that this response has gained popularity during the past decade. First, recording the response does not require the application of external recording electrodes. Also, because the internal electrodes are placed close to the neural elements (in contrast to surface electrodes), the amplitude of the response is relatively large compared with other biologic activity, such as that originating in superficial muscle. As a result, subjects can move around without negatively affecting the quality of the recording. It is not necessary for the child to sleep during the recording procedure. Finally, because of this favorable signal-to-noise ratio, fewer sweeps are needed per average and data collection time is reduced considerably relative to that typically required to record the EABR.
It is largely because of these advantages that there has been a great increase in experimental studies related to ECAP measures made through telemetry systems, and those studies are the focus of the rest of this chapter. Although we are convinced that the ECAP can prove to be a powerful tool in the management of patients with cochlear implants, we also recognize that that there are several very important issues that cannot be addressed with the ECAP. For example, there are changes that are known to take place in the central nervous system as a result of peripheral hearing loss (Miller et al, 1996) and as the result of subsequent electrical stimulation. The ECAP is a measure of the response of the auditory nerve and as such clearly is not sensitive to changes in structures central to the auditory nerve. Studies involving both EABR and late potentials (Gordon et al, 2003; Ponton and Eggermont, 2001; Sharma et al, 2002) have demonstrated plastic changes in the central auditory system in young children receiving cochlear implants. Such measures show considerable promise in characterizing the ability of the central auditory system to adapt to a relatively novel input provided by the cochlear implant. Degenerative changes in structures central to the auditory nerve are likely to affect the amplitude, sensitivity, tuning, and temporal response properties of the auditory system to electrical stimulation. The EABR and/or other more centrally evoked auditory potentials could potentially be used as comparative measures to assess such affects. Because the ECAP and EABR are related and share many of the same properties, the following sections describe experimental methods that can be used to record these responses and provide the reader with an overview of their potential clinical applications. This chapter does not discuss how electrically evoked potentials might be used to assess higher level processes such as learning, adaptation, and cortical plasticity.
♦ Experimental Methods
Electrically Evoked Auditory Brainstem Response
The EABR is typically recorded by stimulating the auditory nerve with a series of pulses applied to a single electrode at a relatively slow rate (i.e., under 50 Hz). The response of the auditory system to this electrical stimulation is then recorded using a differential amplifier and standard averaging techniques. Stimulation is typically achieved using software that is provided by the manufacturer. This software allows for discrete stimulation of individual electrodes with single pulses or trains of pulses and provides an external trigger pulse that can be used to initiate sampling on a separate evoked potential system. Any one of several commercially available evoked potential recording systems that has the capability of being externally triggered can be used to record the EABR. Because stimulus artifact can be problematic, radiofrequency (RF) shielding at the electrode interface and contralateral rather than ipsilateral recording electrode montages are typically used. Despite the use of both RF shielding and a contralateral recording electrode montage, the stimulus artifact associated with the electrical stimulus provided by the cochlear implant can be large enough to saturate the recording amplifier and significantly distort the recordings that are made. Generally, this problem can be managed by using very short duration pulsatile stimuli, by decreasing the gain in the recording amplifier, by increasing the width of the analog filter pass-band that is used to record the EABR (e.g., using 1–3000 Hz rather than 100–3000 Hz), or by alternating the polarity of stimulation in the recording. Some investigators have also used blanking circuits to minimize the effects of stimulus artifact on the EABR (Black et al, 1983). Finding a way to successfully manage stimulus artifact is the key to recording EABRs successfully, and most of the major implant companies and evoked potential manufacturers can provide information to the clinician about how to achieve this goal.
Electrically Evoked Compound Action Potential
The first direct recordings of the ECAP in humans were made in Ineraid cochlear implant users (Brown et al, 1990). With the Ineraid cochlear implant system (Symbion Inc., Utah), all of the implanted electrodes were accessible via a percutaneous plug that could be directly connected to external stimulation and recording equipment (with appropriate ground isolation). The relative simplicity of this design allowed us to experiment with techniques for using one of the intracochlear electrodes to record a response directly from the auditory nerve. Although the proximity of the recording electrode to the auditory nerve was advantageous, the use of an intracochlear electrode to record the ECAP also results in extremely large stimulus artifact recordings. Typically, the electrical stimulus artifact that is recorded from an intracochlear electrode is several orders of magnitude larger than the associated neural response. Additionally, the relatively short latency of the ECAP also made separation of the stimulus artifact from the neural response problematic. Most investigators have used either a two-pulse subtraction technique (Brown et al, 1990) or a polarity alternation technique (Wilson et al, 1997) to reduce the effects of stimulus artifact. Additionally, use of low gain, fast sampling rates (50–100 kHz) and good sampling resolution (≥16 bits) allowed for minimization of stimulus artifact and amplifier saturation effects.
Although much was learned about how to record the ECAP from Ineraid cochlear implant users, the development of telemetry systems such as NRT for the Nucleus implant and NRI for Advanced Bionic implants has provided the means to record the ECAP in patients with cochlear implants that used transcutaneous transmission. With these systems, special software is used to control both stimulation and recording parameters. A stimulus pulse or series of pulses is applied to a specific intracochlear electrode, and the voltages on a nearby electrode are measured for a period of time following the presentation of that stimulus pulse. These voltages are then transmitted to an external receiver through the reverse telemetry system. The neural response telemetry system that was first introduced by Cochlear Corporation in 1995 was not perfect. In fact, it has several important limitations, many of which are directly related to how the recording amplifier responds to large levels of stimulus artifact. We have developed a procedure for adjusting stimulus parameters to minimize the effects of these complications and to assist with recording of responses that are not excessively contaminated by artifact (Abbas et al, 1999). Many of these same techniques for minimizing the impact of excessive stimulus artifact are also useful when the NRI system is used to record the ECAP from Advanced Bionics cochlear implant recipients.
Fig. 8–1 shows examples of the ECAP waveforms that can be recorded using the NRI software. These recordings were obtained from the same individual using the same stimulation levels. The waveforms on the left were obtained using a subtraction procedure to minimize stimulus artifact contamination. The subtraction method takes advantage of neural refractory properties to extract a template of the artifact with no response (masker +probe), which can then be subtracted from the recorded response to the probe alone. Details regarding how this technique works have been published previously (Abbas et al, 1999). The waveforms on the right were collected using alternating polarity stimuli. With both methods, there is some residual stimulus artifact that is followed by a negative peak (N1) and, in many cases, a positive peak (P2). As illustrated in this example, our experience with both telemetry systems has been that the subtraction method is more reliable in reducing stimulus artifact and has typically been our method of choice.
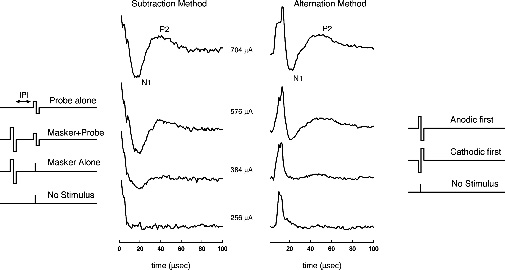
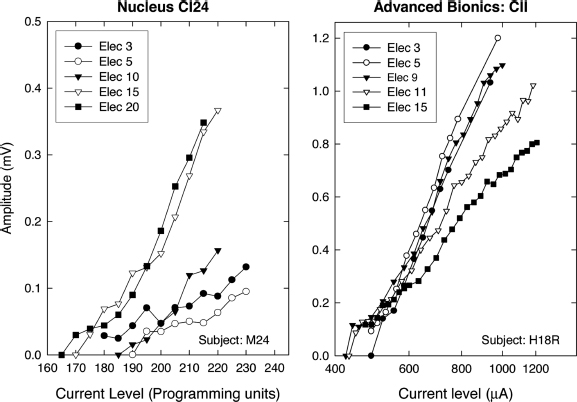
The waveforms illustrated in Fig. 8–1 show responses to four levels of probe. Unlike acoustically evoked auditory potentials, electrically evoked responses change very little in latency as stimulus level is changed. Consequently response amplitude (measured between the negative trough and the following positive peak) rather than latency is commonly used to quantify the response. To measure an ECAP growth function using the subtraction method, the interpulse interval (IPI) is fixed (typically at 500 μs), the masker pulse is fixed at a relatively high level [near the uncomfortable loudness level (UCL)], and the level of the probe is varied. A high-level masker pulse is used because the sole purpose of the masker is to put all of the neurons that may respond to the probe into a refractory state. Probe levels are then chosen that do not exceed the level of the masker. Although we cannot be certain that all of the stimulable fibers are refractory at the time the probe is presented, we would argue that the best way to ensure this is true is to use of the highest level of masker possible. Alternatively, the level of the masker pulse can be varied, keeping it at a fixed level relative to the level of the probe (typically 5 to 10 current units above the probe). Comparisons of growth functions using the two methods have yielded no significant differences (Hughes et al, 2001).