Purpose
To evaluate the effects of birth weight on ocular anterior segment parameters in full-term children without low birth weight using the Galilei Dual-Scheimpflug Analyzer.
Design
Retrospective cohort study.
Methods
The right eyes from 110 healthy children, 3–6 years of age, were scanned with the Galilei Dual-Scheimpflug Analyzer. A total of 78 eyes were measured in full-term children with birth weight of >2500 g. Central, paracentral, pericentral, and the thinnest corneal thicknesses; anterior and posterior keratometry (average, steep, flat); axial curvatures; asphericity of cornea; anterior chamber depth and volume; and iridocorneal angle values were measured. Axial length, lens thickness, and vitreous length were obtained by ultrasound biometry.
Results
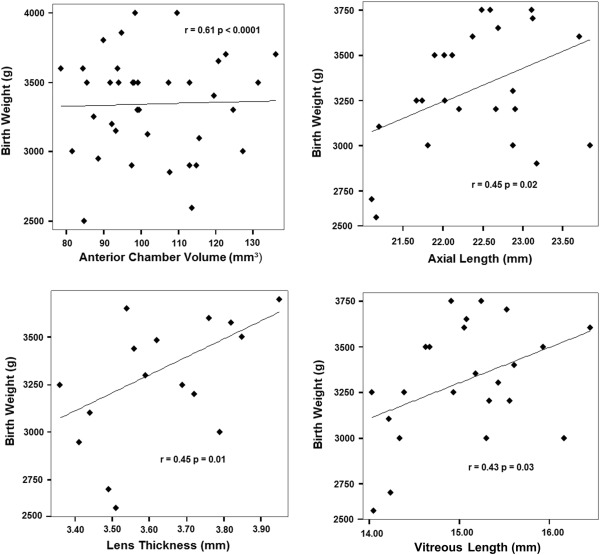
The mean age of children was 55.86 ± 12.52 (mean ± SD) months. Mean birth weight and gestational age were 3426.3 ± 545 g and 39.4 ± 1.2 weeks, respectively. Although lens thickness, vitreous length, axial length, and anterior chamber volume were moderately correlated with birth weight ( P < .05), there was no relationship between birth weight and anterior chamber depth. With the exception of pericentral corneal thickness, all regions of corneal thicknesses were correlated with birth weight ( P < .05). Birth weight was negatively correlated with anterior curvature ( P < .05) and had no relationship to posterior curvature. While central and paracentral axial curvatures correlated with birth weight ( P < .05), pericentral axial curvature did not.
Conclusion
Preschoolers who were born heavier had thicker cornea and lens, longer axial length, and flatter corneal curve. The thicknesses and axial curves of central cornea within 7 mm may be particularly associated with birth weight.
Prematurity and low birth weight are associated with many later-in-life maladies, including metabolic syndrome, high blood pressure, type 2 diabetes mellitus, and neurodevelopmental disorders. Preterm babies with low birth weight may experience retinopathy of prematurity (ROP), myopia, strabismus, amblyopia, and cerebral visual impairment. Although the association of birth weight with anterior segment parameters has been investigated, those studies had usually been performed on preterm, low-birth-weight (especially those with ROP) infants in the early weeks of life or on multiethnic children. Ethnicity is a factor with a potentially great impact on both birth and anterior segment parameters. In order to determine the effect on anterior segment structures of birth weight alone, healthy children, born at term, of the same ethnicity need to be evaluated.
Although it is widely accepted that birth weight and gestational age have effects on the globe’s axial length, anterior corneal curvature, and anterior chamber depth, no study has specifically investigated the association of birth weight with anterior segment parameters, including central, paracentral, pericentral, and thinnest corneal thicknesses; posterior corneal curvature; total corneal power; central, paracentral, and pericentral axial curvature; corneal asphericity; anterior chamber volume; lens thickness; and angles of the 4 cardinal quadrants. With the exception of lens thickness, all of the aforementioned parameters are measurable with the Galilei Dual-Scheimpflug Analyzer, a device that is able to precisely detect the posterior corneal surface. The Galilei Dual-Scheimpflug Analyzer is equipped with 2 Scheimpflug cameras, both of which provide accurate pachymetry across the entire cornea by delivering precise measurements of the anterior chamber, even when the camera is de-centered owing to micro-movements of the eye. The Galilei Dual-Scheimpflug Analyzer is also integrated with placido disc–based topography, which conveys accurate topographic data not only from the entire cornea but also from >122 000 data points/scan.
In the present study, we investigated the impact of birth weight on anterior segment structures measured by Galilei Dual-Scheimpflug Analyzer in healthy full-term preschoolers without low birth weight.
Methods
Subjects
In this retrospective cohort study, we examined 133 healthy white children, aged 3–6 years, who visited (with their parents) our clinic for routine eye examinations between January 1, 2011 and March 28, 2013. The investigational purposes of all measurements were explained to parents, after which written informed consents were obtained from both parents of each participating child. The study was approved by the Human Research Ethics Committee of Ondokuz Mayıs University, Samsun, Turkey (Permission no: 2010-171), and was conducted in accordance with the tenets of the Declaration of Helsinki and was registered at clinicaltrials.gov (registration number NCT02466932 ).
After taking extensive histories, we performed a comprehensive ophthalmologic examination, including autorefractometry (KR-3500 Autokeratorefractometer; Topcon, Tokyo, Japan) before and after pupil dilation; visual acuity measurement (tumbling E-test); digital intraocular pressure measurements; slit-lamp biomicroscopy; and funduscopy on all subjects. Inclusion criteria comprised the following: age 3–6 years; birth weight >2500 g; gestational age (ie, the period of time between the first day of the mother’s last menstrual period and birth) >37 weeks; visual acuity >0.8 (in decimals); no history of any systemic or ocular diseases; no history of previous ocular surgeries; no history of ROP; no current medications; and no retinal or optic disc abnormalities on funduscopy. Birth history data, including birth weight and gestational age, were obtained from documented medical records (hospital, physician, or nurse), copies of which were provided by the parents. Prematurity and low birth weight were defined as gestational age <37 weeks and birth weight <2500 g, respectively. As 13 subjects had no documented records, we obtained values of the various birth parameters from the parents; 4 of these subjects were ultimately excluded because of prematurity or low birth weight. Thus, 9 of 78 children (11.5%) with only verbal documentation were included in the study. In order to prevent any bias, we reanalyzed the data after these children (n = 9) were excluded, and we found statistically significant and similar results. Because the extraction of their data had not changed the statistical results, we did not need to exclude them from the study.
The parents of 23 children did not accept study guidelines and, thus, these children were excluded. Twenty-eight children whose measurements were unreliable for analysis because of involuntary eye movements were also excluded from the study. Two children had gestational age >37 weeks but birth weight <2500 g, and 2 had birth weight >2500 g but gestational age <37 weeks. Consequently, 55 subjects were totally excluded from the study (55 of 133 children; [41.3%]). Data regarding exclusion and inclusion of the children are shown in Table 1 . Because results from separate analyses of right and left eyes were similar, only results of 1 eye (right) are reported.
| Number of Children | |
|---|---|
| All examined | 133 |
| No parental permission | 23 a |
| Measured by Galilei Dual-Scheimpflug Analyzer | 110 |
| With unreliable measurements | 28 a |
| Birth weight >2500 g, gestational age <37 wk | 2 a |
| Gestational age >37 wk, birth weight <2500 g | 2 a |
| Totally excluded | 55 a |
| Totally included | 78 |
Anterior Segment Measurements
Measurements were taken consecutively using the Galilei Dual-Scheimpflug Analyzer (Ziemer Group, Port, Switzerland). Conventional ultrasound biometry was performed by the same experienced ophthalmic technician. All measurements were accomplished on undilated pupils. Since the direct contact of the ultrasound probe on the cornea could influence Scheimpflug measurements, we took the measurements by Galilei Dual-Scheimpflug Analyzer prior to ultrasound biometry. The child was comfortably seated next to his or her parents, with his or her chin on the chin rest and forehead against the strap. While the child looked straight ahead at the fixation target of the device, the operator manually focused and aligned the image. One of the device’s indicators requires manual placement of the Scheimpflug slit-image at the corneal apex by manipulating the joystick with one hand, while appropriate alignment in the horizontal, vertical, and anteroposterior axes is accomplished as the other hand positions the center of the pupil on the screen. When all device indicators were appropriately positioned according to manufacturer guidelines, the child was told to blink once and then open his or her eye wide, with no further eye movement. When this was done, the entire scan was performed. The time taken for each scan was ∼2 s. Scans with overall qualities of >85% based on the quality checker of Galilei software were considered “reliable.” If a scan was deemed unreliable, the process was repeated until a reliable scan was obtained.
The values of central (0–4 mm central cornea), paracentral (4–7 mm central cornea), pericentral (7–10 mm central cornea), and thinnest corneal thicknesses; posterior corneal curvature; total corneal power; central (0–4 mm central cornea), paracentral (4–7 mm central cornea), and pericentral (7–8 mm central cornea) axial curvatures; anterior and posterior corneal asphericities; anterior chamber depth and volume; and superior, temporal, inferior, and nasal iridocorneal angles (automatically determined by Galilei software) were used for analyses. As the 4 parameters used to describe the shape of a conic section—eccentricity (ε), reported as its square, ε 2 ; Q (asphericity); P value; and corneal shape factor (E)—are mathematically expressed by the following equation: ε 2 = E = 1 − p = −Q , we converted ε 2 to Q by multiplying by −1. Galilei software calculates the eccentricity (ε 2 ) of the surface within a central diameter of 8 mm, averaged over all meridians, and was calculated for the anterior and posterior surfaces. Total corneal power was calculated by ray-tracing through the anterior and posterior surfaces using Snell’s law and thin-lens equations. The indices of refraction used were n = 1.0, n = 1.376, and n = 1.336 for air, cornea, and aqueous, respectively. Pachymetry data were used for thicknesses in ray-tracing. The parameters of posterior keratometry (K-avg, K-flat, and K-steep) are basically the same as those for the anterior surface, the only differences being that these K-values are calculated, not simulated, using the real indices of refraction of the cornea and aqueous humor.
For A-scan ultrasonography (Pacscan 300 A A-scan ultrasound; Sonomed Inc, Lake Success, New York, USA), the cornea was anesthetized with 1 drop of topical Alcaine (proparacaine hydrochloride 0.5%; Alcon, Fort Worth, Texas, USA). Following (5 min) application of the topical anesthetic, complete ocular biometric measurements were performed, with the child seated upright and looking at a fingertip located 30 cm from his or her eye (in order to provide an accommodative condition similar to that of the Galilei). Ultrasound velocity was set at 1532 m/s, with the frequency of the ultrasound probe set at 20 MHz. At the same time, a handheld straight probe was aligned (as perpendicularly as possible) onto the central cornea by the technician taking measurements. The means of 3 (minimum) consecutive measurements of axial length, anterior chamber depth, lens thickness, and vitreous length—all with the lowest standard deviations (SD <0.05)—were used for analyses.
Statistical Analyses
Data were statistically analyzed using version 16 SPSS software for Windows (SPSS Inc, Chicago, Illinois, USA). The Pearson correlation coefficient ( r ) was used to measure the strength of the relationship between 2 variables, as all parameters in our study showed parametric distributions. The Mann-Whitney U test was performed for comparison of groups. A P value <.05 indicated statistical significance.
Results
Descriptive statistics of all subjects for age; spherical equivalant; central, paracentral, pericentral, and thinnest corneal thicknesses; posterior corneal curvature; total corneal power; central, paracentral, and pericentral axial curvatures; asphericities (anterior and posterior); anterior chamber depth; anterior chamber volume; superior, temporal, inferior, and nasal iridocorneal angles; lens thickness; vitreous length; and axial length are shown in Table 2 .
| Parameter | Mean ± SD | Median (Min, Max) | Birth Weight | Gestational Age | ||
|---|---|---|---|---|---|---|
| r | P | r | P | |||
| Age (m) | 55.86 ± 12.52 | 57 (34, 70) | −0.47 | .70 | 0.03 | .77 |
| Spherical equivalent (D) | −0.01 ± 0.47 | 0.25 (−1.00, 1.00) | 0.10 | .38 | 0.04 | .72 |
| Central corneal thickness (μm) | 574.3 ± 24.3 | 575.0 (510, 627) | 0.30 | .01 a | 0.11 | .40 |
| Paracentral corneal thickness (μm) | 610.8 ± 23.3 | 611.0 (564, 666) | 0.31 | .01 a | 0.18 | .18 |
| Pericentral corneal thickness (μm) | 676.3 ± 29.5 | 674.5 (621, 744) | 0.21 | .1 | 0.08 | .53 |
| Thinnest corneal thickness (μm) | 562.7 ± 22.0 | 566.0 (517, 612) | 0.30 | .03 a | 0.17 | .20 |
| Sim K flat (D) | 43.00 ± 1.7 | 42.91 (38.34, 48.27) | −0.34 | .01 a | −0.14 | .30 |
| Sim K steep (D) | 44.05 ± 1.65 | 44.12 (39.98, 48.97) | −0.28 | .03 a | −0.10 | .45 |
| Sim K avg (D) | 43.52 ± 1.62 | 43.49 (39.89, 48.62) | −0.32 | .01 a | −0.13 | .34 |
| Anterior asphericity | −0.41 ± 0.30 | −0.34 (−2.13, −0.05) | 0.08 | .53 | −0.05 | .70 |
| Posterior asphericity | −0.42 ± −0.30 | −0.34 (−3.82, 0.29) | 0.19 | .18 | 0.13 | .35 |
| Anterior best-fit sphere (mm) | 7.89 ± 0.30 | 7.87 (7.08, 8.57) | 0.30 | .02 a | 0.13 | .36 |
| Posterior best-fit sphere (mm) | 6.68 ± 0.33 | 6.61 (6.05, 7.79) | 0.18 | .19 | 0.06 | .66 |
| Posterior K flat (D) | −5.99 ± 0.29 | −6.03 (−6.46, −4.78) | 0.11 | .43 | 0.02 | .88 |
| Posterior K steep (D) | −6.35 ± 0.28 | −6.39 (−7.10, −5.59) | 0.07 | .60 | −0.09 | .50 |
| Posterior K avg (D) | −6.17 ± 0.25 | −6.19 (−6.78, −5.50) | 0.10 | .46 | −0.03 | .80 |
| Total corneal power flat (D) | 42.67 ± 1.74 | 42.56 (37.9, 48.3) | −0.35 | .007 a | −0.16 | .24 |
| Total corneal power steep (D) | 43.72 ± 1.68 | 43.78 (39.9, 48.7) | −0.30 | .02 a | −0.12 | .78 |
| Total corneal power mean (D) | 43.20 ± 1.65 | 43.18 (39.8, 48.5) | −0.34 | .01 a | −0.14 | .28 |
| Axial curvature central (D) | 43.57 ± 1.62 | 43.49 (39.91, 48.70) | −0.32 | .01 a | −0.13 | .34 |
| Axial curvature paracentral (D) | 42.75 ± 1.60 | 42.72 (39.22, 47.60) | −0.32 | .01 a | −0.12 | .39 |
| Axial curvature pericentral (D) | 41.62 ± 2.13 | 41.77 (32.57, 46.33) | −0.22 | .10 | −0.09 | .48 |
| Anterior chamber depth (mm) | 3.71 ± 0.20 | 3.69 (3.38, 4.17) | 0.01 | .92 | −0.05 | .74 |
| Anterior chamber volume (mm 3 ) | 115.86 ± 60.22 | 102.45 (78.6, 405.20) | 0.61 | <.0001 a | 0.06 | .69 |
| Lens thickness (mm) | 3.60 ± 0.15 | 3.56 (3.36, 3.95) | 0.45 | .01 a | 0.28 | .15 |
| Vitreous length (mm) | 15.05 ± 0.66 | 15.08 (14.03, 16.45) | 0.43 | .03 a | 0.35 | .07 |
| Axial length (mm) | 22.37 ± 0.75 | 22.38 (21.11, 23.84) | 0.45 | .02 a | 0.37 | .06 |
| Superior – angle (degrees) | 52.32 ± 4.37 | 51.75 (44.40, 68.40) | −0.25 | .16 | −0.29 | .10 |
| Temporal – angle (degrees) | 52.56 ± 3.17 | 52.90 (45.70, 62.50) | 0.04 | .85 | 0.06 | .78 |
| Inferior – angle (degrees) | 50.78 ± 2.97 | 50.40 (43.40, 55.60) | −0.004 | .98 | 0.01 | .99 |
| Nasal – angle (degrees) | 55.10 ± 3.65 | 54.90 (48.60, 61.70) | 0.01 | .96 | −0.02 | .92 |
a Statistically significant. Pearson correlation test was used for analyses.
Means of age, birth weight, and gestational age were 55.86 ± 12.52 months (mean ± SD), 3426.3 ± 545 g, and 39.4 ± 1.2 weeks, respectively. There were 40 girls (51%) and 38 boys (49%). No statistically significant differences between sexes for any anterior segment or birth parameter ( P > .1) were found. At birth, 49% of children (n = 38) had been delivered vaginally and 51% (n = 40) by cesarean section; no significant differences were found between these groups for any parameter ( P > .05). Although lens thickness, vitreous length, axial length, and anterior chamber volume were moderately correlated with birth weight ( P < .05), no correlation was found between birth weight and anterior chamber depth ( Figure 1 ). Central, paracentral, and thinnest corneal thicknesses were only weakly correlated with birth weight ( P < .05) ( Figure 2 ). Birth weight was negatively correlated with anterior ( P < .05), but not posterior, keratometry. While central and paracentral axial curvatures were positively correlated with birth weight ( P < .05), pericentral axial curvature was not. No relationship between gestational age and any anterior segment parameter was found ( P > .05). All statistics related to birth weight and gestational age relationships are shown in Table 2 .