INFECTIONS OF THE EXTERNAL EAR
Otolaryngologists see many patients with infections of the external ear. The infections may be classified by location, cause, and time course as acute, subacute, and chronic. Before discussing the individual disease processes, we review the normal anatomy and physiology of the external ear.
ANATOMY AND PHYSIOLOGY
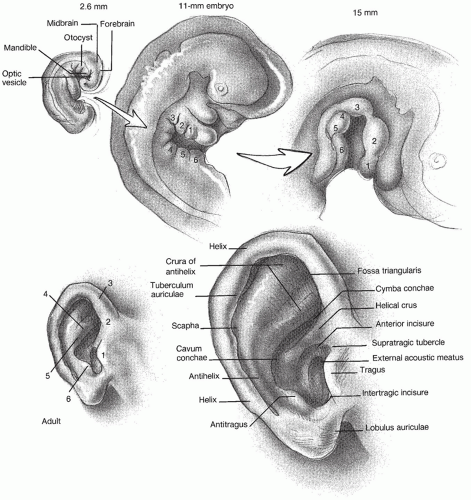
The external ear is composed of the auricle and external auditory canal (EAC). Both contain elastic cartilage derived from mesoderm and a small amount of subcutaneous tissue, covered by skin with its adnexal appendages (
1,
2). There is fat but no cartilage in the lobule. The auricle is derived from six hillocks, three each from branchial arches I and II (
Fig. 146.1). During normal gestation, the cartilaginous hillocks merge to form the auricle, and with selective growth of the mandible, the auricle rises from its original position near the lateral commissure of the mouth to the temporal area. The tragus and antitragus form a partial barrier to the entrance of macroscopic foreign bodies.
The EAC is derived from the first ectodermal branchial groove between the mandibular (I) and hyoid (II) arches (
2,
3). The epithelium lining this groove contacts the endoderm of the first pharyngeal pouch, thus forming the tympanic membrane (TM), the most medial extent of EAC. Connective tissue of mesodermal origin is found between ectoderm and endoderm and becomes the fibrous layer of the TM (
2). Because of its origin, the EAC, including the lateral surface of the TM, is derived from ectoderm and is lined by squamous epithelium.
The process of canalization is complete by about week 12 of gestation, at which time the canal fills with epithelial tissue. The canal ordinarily recanalizes by about week 28 of fetal life (
3).
The EAC may be thought of in two sections. The outer 40% of the canal in its anterior and inferior aspect is cartilaginous and contains a thin layer of subcutaneous tissue between the skin and cartilage. The inner 60% is osseous, is formed primarily by the tympanic ring, and contains very scant soft tissue between the skin, periosteum, and bone. The average length of the adult EAC is 2.5 cm. Because of the oblique position of the TM, the posterosuperior part of the canal is about 6 mm shorter than the anteroinferior portion (
1). The junction of the cartilaginous and bony portions of the canal is a narrowed section termed the isthmus.
Laterally to medially, the canal curves slightly superiorly and posteriorly in a gentle S shape. The canal can be thought of as pointing toward the nose; thus, pull the auricle gently upward, outward, and backward to straighten the canal for examination. Three macroscopic defense mechanisms protect the EAC and lateral surface of the TM: the tragus and antitragus, the skin with its cerumen coat, and the isthmus of the canal.
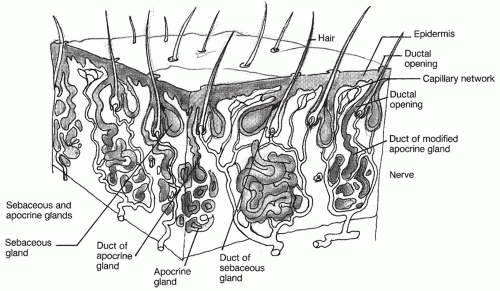
The skin of the cartilaginous canal contains many hair cells and sebaceous and apocrine glands such as cerumen glands (
Fig. 146.2). Together, these three adnexal structures provide a protective function and are termed the
apopilosebaceous unit. Glandular secretions combine with sloughed squamous epithelium to form an acidic coat of cerumen, one of the primary barriers to infection of the canal. An invagination of the epidermis forms the outer wall of the hair follicle, and the hair shaft forms the inner wall. The follicular canal is the space between these two structures. The alveoli of the sebaceous and apocrine glands empty into short straight excretory ducts, which drain into follicular canals. Obstruction of any part of the ductal system predisposes to infection.
The canal is normally a self-protecting and self-cleansing structure. The cerumen coat gradually works its way past the isthmus to the lateral part of the canal and
sloughs externally. Instrumentation and excessive cleansing of the canal disturb this primary protective barrier and may lead to infection. Individual variations in the anatomy of the canal may predispose some people to wax accumulation.
The canal interfaces on all but its lateral surface. Medially, it is bound by the TM, which when intact is a good barrier to the spread of infection. In the presence of a TM perforation, infection may spread back and forth from the middle ear cleft to the EAC. The horseshoe-shaped tympanic ring separates the canal from the middle cranial fossa. Rarely is this the direct mechanism of intracranial extension of infection. The posterior bony canal serves as the anterior boundary of the mastoid cavity. Several vessels penetrate the canal, especially along the tympanomastoid suture. These may be involved in the hematogenous extension of infection from the canal to the mastoid segment. Posterior to the cartilaginous canal lies dense connective tissue overlying the mastoid, which may become secondarily infected.
Superiorly, the canal is bound by the middle cranial fossa and inferiorly by the infratemporal fossa and base of the skull. Infections extending through the roof of the canal may extend into these structures. Anteriorly, the canal is bordered by the temporomandibular joint and the parotid gland.
The lymphatic drainage of the canal is an important channel for the spread of infection. Anteriorly and superiorly, the canal drains to the preauricular lymphatics in the parotid gland and the superior deep cervical nodes. The inferior portion of the canal drains into the infra-auricular nodes near the angle of the mandible. Posteriorly, the lymphatics drain into the postauricular nodes and the superior deep cervical nodes. The auricle and EAC receive their arterial supply from the superficial temporal and posterior auricular branches of the external carotid artery (
1). Venous drainage from the auricle and meatus is via the superficial temporal and posterior auricular veins. The former joins the retromandibular vein, which usually divides and joins both jugular veins; the latter joins the external jugular but may also drain to the sigmoid sinus through the mastoid emissary vein (
1).
Sensation to the auricle and EAC is supplied from cutaneous and cranial nerves, with contributions from the auriculotemporal branches of the trigeminal (V), facial (VII), glossopharyngeal (IX), and vagus (X) nerves and the greater auricular nerve from the cervical plexus (C2 to C3). The vestigial extrinsic muscles of the ear, anterior, superior, and posterior auricular, are supplied by the facial nerve (VII) (
1).
OTITIS EXTERNA
Otitis externa is a spectrum of infection of the EAC. The appearance of the canal varies according to the time course of infection: acute, subacute, or chronic.
Acute otitis externa (AOE) is a bacterial infection of the canal caused by a break in the normal skin/cerumen protective barrier in the milieu of elevated humidity and temperature. Although commonly called “swimmer’s ear,” AOE may be caused by anything that results in the removal of the protective lipid film from the canal, allowing bacteria to enter the apopilosebaceous unit. It usually begins with itching in the canal and is commonly caused by instrumenting the canal with a cotton swab or fingernail. This temporarily relieves itching but allows proliferation of bacteria in locally macerated skin and sets up an itch-scratch cycle. The warm, dark, moist setting of the canal is now a perfect medium for rapid bacterial growth. Later, pain ensues as the swollen soft tissues of the canal distract the periosteal lining of the bony canal. As the disease progresses, purulent discharge begins, and the auricle and periauricular soft tissues may become involved.
In patients in whom the disease does not resolve after treatment, a subacute or chronic form may occur. This condition may be likened to eczema and is a spectrum of disease ranging from mild drying and scaling of the canal skin to complete obliteration of the canal by chronically infected hypertrophic skin.
HISTORY
The history and functional inquiry should include information regarding the length of time, the number of occurrences, the nature and severity of pain, antecedent otologic disease, previous auricular instrumentation or trauma (especially the use of cotton-tipped applicators), and any predisposing factors such as diabetes or radiotherapy or any condition causing immunosuppression. Any previous otologic or head and neck surgery is noted.
Pain, fullness, itching, and hearing loss are the four major symptoms of external otitis, although not every patient has each symptom (
4). Throughout the examination, the examiner should remember the innervation of the EAC and recall that pain from other areas of the upper aerodigestive tract may be referred to the ear.
BACTERIOLOGY
The usual pathogens responsible for AOE are Pseudomonas aeruginosa, Proteus mirabilis, staphylococci, streptococci, and various gram-negative bacilli. For the mild or uncomplicated infection, culture of the canal is ordinarily not taken, because it will usually demonstrate a mixed pattern of growth. For recalcitrant infections, culture may identify a predominant organism and assist in the choice of antibiotic therapy.
STAGING
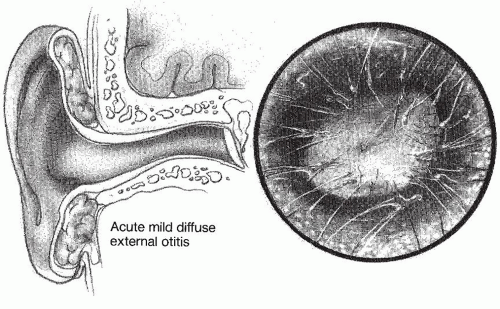
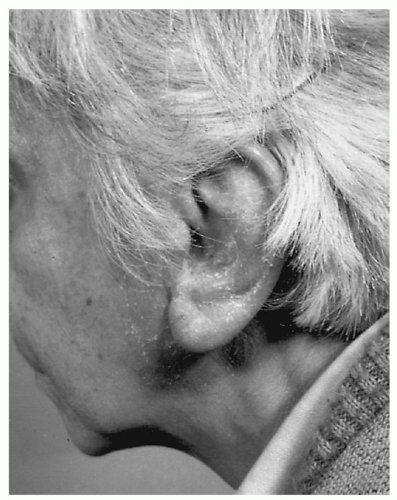
The clinical course of external otitis may be divided into the following stages: preinflammatory; acute inflammatory, which can be mild, moderate, or severe; and chronic inflammatory. Typically, the preinflammatory stage begins when the stratum corneum becomes edematous due to the removal of the protective lipid layer and acid mantle from the canal, resulting in plugging of the apopilosebaceous unit. As obstruction continues, a sense of fullness and itching begins. The disruption of the epithelial layer allows invasion of bacteria that either reside in the canal or are introduced on foreign objects inserted into the canal, such as a cotton swab or a dirty fingernail. This produces the acute inflammatory stage, which is accompanied by pain and tenderness of the auricle. In the earliest stage, the skin of the EAC shows mild erythema and minimal edema (
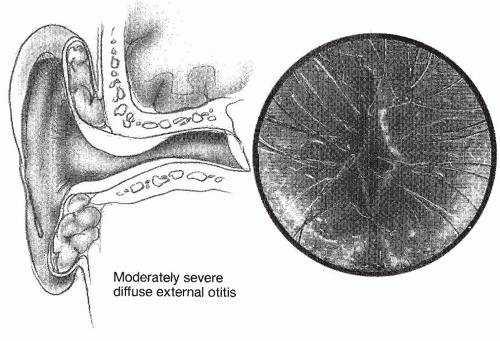
Fig. 146.3). A small amount of clear or slightly cloudy secretion may be seen in the canal. As pain and itching increase, the patient progresses to the moderate stage, in which the canal shows more edema and a thicker more profuse exudate (
Fig. 146.4). Further progression of the inflammation in the absence of treatment produces the severe inflammatory stage, characterized by increased pain and obliteration of the lumen of the canal. A profuse, purulent exudate and edema of the canal skin may obscure the TM. In addition, small white papules are often visible on the surface of the canal skin.
P. aeruginosa or another gram-negative bacillus can almost always be cultured at this stage. In the severe stage, the physician often sees evidence of extension of infection beyond the canal to involve the adjacent soft tissues and cervical lymph nodes (
Fig. 146.5).
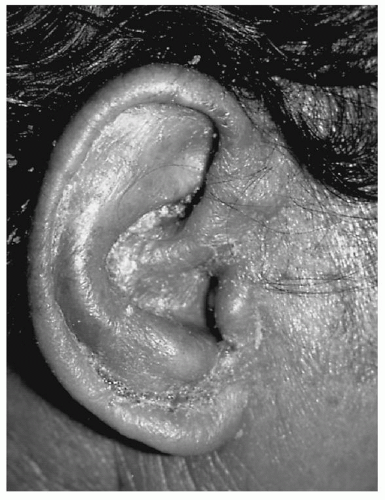
In the chronic inflammatory stage, the patient experiences less pain but more profound itching. The skin of the external canal is thickened, and superficial flaking may be seen (
Fig. 146.6). The auricle and concha often show secondary changes such as eczematization, lichenification, and superficial ulceration. This condition is likened to eczema and may range from mild drying and thickening of the canal to complete obliteration of the external canal by chronically infected, hypertrophic skin (
Table 146.1).
NATURAL HISTORY
The natural history of untreated AOE is one of increasing pain, swelling, and discharge from the canal. The infection may spread to the adjacent periauricular soft tissues, face, and neck. In an immunocompromised patient, what began as an isolated superficial infection of the apopilosebaceous unit of the EAC may progress to perichondritis, chondritis, cellulitis, and erysipelas. Rich lymphatic and hematogenous drainage pathways favor the spread of infection to local and regional sites in the head and neck. Few patients progress to such an advanced stage before seeking medical attention.
The natural history of chronic otitis externa is far less dramatic than its acute counterpart. The chronic scaling and itching in the canal predispose to manipulation of the canal, excoriation, and repeated episodes of AOE. With time, the canal skin may become lichenified, and ultimately the canal may become completely obliterated.
MODERATE STAGE
In the moderate stage of inflammation, edema of the canal may interfere with the instillation of drops. The physician should then insert a wick into the canal and instill drops on it. Often the canal may accommodate two or even three wicks. As the wick expands, it presses the soft tissues and periosteum toward the nondistracted position; this alone may relieve pain. All instrumentation of the ear is best done under the microscope. The wick is removed by the
physician at the time of reexamination. If the edema has not been significantly reduced, repacking is indicated. Antibiotic drops should be continued for at least 2 to 3 days after the cessation of pain, itching, and drainage so that complete eradication of infection may be ensured.
In the moderate stage, an oral analgesic is often prescribed because pain can be pronounced. Caution the patient to avoid manipulation of the canal. Swimmers should be taught to towel dry the concha and lateral canal, to shake water out of the canal, or to instill an acidifying drop after swimming. If the infection has not spread beyond the boundaries of the external canal, the use of oral antibiotics will be of little if any value. A final office visit is important to ensure that the infection has completely resolved and the canal is back to its normal state.
SEVERE STAGE
In the severe stage, infection usually extends beyond the limits of the canal. In addition to the cleaning, packing, and use of antibiotic drops as discussed previously, attend to any soft tissue involvement by using an oral antibiotic with broad-spectrum coverage. Successive generations of the cephalosporins widen gram-negative coverage at the expense of gram-positive coverage.
In addition to anti-Pseudomonas ear drops, common choices of oral antibiotics are one of the antipseudomonal fluoroquinolones such as ciprofloxacin or levofloxacin, antistaphylococcal penicillins, or cephalosporins. The fluoroquinolone antibiotics are effective against Pseudomonas species. In children under 12 years old, one should check with the pediatrician prior to starting oral fluoroquinolones.
Warm soaks (normal saline or a mild aluminum sulfate-calcium acetate solution) are also useful in the treatment of the crusting and edema involving the auricle and surrounding skin. Culture of the canal for aerobic bacteria and/or fungi is indicated only for the severe stage or for patients who have previously been treated without resolution. Treatment is generally continued for 10 to 14 days if there is a good response. In rare patients who do not respond to this regimen, hospitalization, vigorous daily local care, repeat culturing, and intravenous antibiotics are indicated.
CHRONIC STAGE
The chronic stage of external otitis is manifested by marked thickening of the skin of the EAC due to long-standing infection. Examination reveals flakes of dry scaly skin in the canal. Although removal of debris is recommended, this may be difficult due to the narrowing of the lumen of the canal. Repeated cleaning and instillation of antibiotics and steroids are indicated. Triamcinolone acetonide 0.25% cream or ointment (Kenalog), fluocinolone 0.01% oil (DermOtic), or dexamethasone sodium phosphate 0.1% ophthalmic drops (Decadron, Pred Forte 1%) may be used.
In all cases of acute or chronic external otitis, instruct the patient to avoid future infections by not instrumenting the ear. Foreign objects such as Q-tips often excoriate the canal skin and push debris further into the canal rather than remove it. Patients who have repeated infections despite adhering to these measures are best advised to use an acidifying drop composed of equal measures of vinegar and water, or ethyl alcohol and water, when exposed to high humidity. One should suspect otomycosis if all other reasonable measures have failed and should treat with drying agents, especially powders. Custom-made ear molds are useful for these patients.
RECALCITRANT OTITIS EXTERNA
The physician will be able to judge very quickly which patients are responding. Prolonged antibiotic drops may suppress the normal flora of the external canal from returning and lead to a fungal superinfection. This should be suspected, especially if a grayish matted discharge is found in the ear, even if telltale hyphae are absent. A culture may have already been taken, and the result should help to direct treatment. Again, the ear must be thoroughly and meticulously cleaned, and if a fungus has been cultured such as Candida, this should be treated appropriately with drying agents such as powders and an antifungal drop or even systemically with an oral antifungal medication such as fluconazole. If no progress is made in the office, the rare patient may have to be admitted as an inpatient. Examine the ear under a microscope and carefully clean it frequently. Look for subtle signs of chronic middle ear disease—granulation tissue or the opening of a tiny perforation. The latter may be obscured by a swollen TM, giving a “fishmouth” appearance to the perforation. A “sewer cap” of crust on the drum may reveal a cholesteatoma underneath. Examine every part of the auricle carefully. Look for signs of underlying chondritis or perichondritis, especially diffuse crusting or exudative weeping. Culture the ear (for aerobes, anaerobes, and fungi). Examine the periauricular tissues carefully to look for signs of spreading infection. Computed tomography of the temporal bones may give additional information. Look for opacification of the mastoid and signs of bony erosion.
Place the patient on daily aural drops, preferably one covering P. aeruginosa, and intravenous antibiotics with gram-positive and gram-negative coverage. One may wish to try intravenous ciprofloxacin to start. In very rare cases, a cephalosporin together with an aminoglycoside is a logical combination if monotherapy does not improve the ear. Therapy should be tapered according to culture and sensitivities. Severely swollen ears may calm down with steroids.
Many recalcitrant infections occur because of noncompliance or chronic instrumentation of the canal skin. The patient should be counseled to amend bad habits. The rare patient will need to be admitted for intravenous antibiotics and daily aural toilet (
Tables 146.2 and
146.3).
SURGICAL MANAGEMENT OF HYPERTROPHIC CHRONIC OTITIS EXTERNA
When such local measures are insufficient to eradicate infection and reestablish the lumen of the canal, it is necessary to remove the involved canal skin and any adjacent involved cutaneous or cartilaginous tissue. This is very rarely required and may be performed through the canal but is better done through a postauricular incision, which allows visualization of the involved tissues. A generous amount of conchal cartilage is removed to effect a wide meatoplasty. The bony canal is enlarged with a drill. Take care to protect the facial nerve in its vertical segment; the use of an intraoperative facial nerve monitor facilitates this. The canal is resurfaced with a split-thickness (8:1,000 inch) skin graft that is temporarily held in place with stents or packing.
It is the experience of many otolaryngologists that even the most recalcitrant infection can be managed if the four basic principles—thorough cleaning, antibiotic treatment, pain control, and instruction of the patient—are meticulously followed. There is no substitute for thorough and repeated local care (
Table 146.4).
SKULL BASE OSTEOMYELITIS, FORMERLY NECROTIZING (MALIGNANT) OTITIS EXTERNA
This potentially life-threatening disease should be viewed within the larger context of osteomyelitis of the temporal bone and skull base. Thanks in large part to newer anti- Pseudomonas antibiotics, chief among them the fluoroquinolones, skull base osteomyelitis (SBO) is not as prevalent as it was even a decade ago. By no means is it a disease of the past, especially among diabetics, the immunocompromised, and the elderly. The otolaryngologist must still keep SBO within the differential diagnosis of a refractory external ear infection in the patient at risk.
Toulmouche (
5) described a case of progressive osteomyelitis of the temporal bone in 1838 that was probably the first reported case of necrotizing external otitis (NEO). In 1959, Meltzer and Kellemen (
6) described a case of progressive
Pseudomonas osteomyelitis of the temporal bone and skull base. Chandler (
7,
8,
9,
10,
11) is credited with the clinical description of what he termed malignant otitis externa in 1968 and thereafter. Although many early cases ended in the death of the patient, the entity does not ordinarily involve carcinoma, and thus, the more appropriate term used today is SBO, which is a proper description of the pathophysiology and clinical entity.
CLINICAL AND RADIOGRAPHIC FINDINGS
SBO usually begins as an acute external otitis that does not resolve despite medical therapy, as described previously. The history is significant for a long-standing infection of the external canal accompanied by aural discharge and severe deep-seated pain. The disease is usually found in elderly diabetic patients in poor metabolic control, although it may be found in any chronically ill, debilitated, or immunocompromised patient (
15). The HIV status of the patient should be known.
On physical examination, most patients with SBO have granulation tissue visible in the inferior aspect of the canal or even extruding from it. This may obscure the TM. It is rare to see granulation tissue in patients with routine otitis externa; however, granulations are common in an acute exacerbation of chronic otitis media with perforation of the TM. The skin of the canal is often erythematous, indurated, and sometimes macerated. Purulent secretions are common.