Purpose
To determine the relationship between delayed patchy choroidal filling and morphologic and functional outcomes among eyes treated with ranibizumab or bevacizumab.
Design
Cohort study.
Methods
Comparison of Age-related Macular Degeneration Treatment Trials participants were assigned randomly to ranibizumab or bevacizumab on a monthly or as-needed schedule. Presence of delayed patchy choroidal filling and morphologic and functional outcomes were evaluated among eyes with gradable fluorescein angiography at baseline (n = 973) and at 1 year (n = 860) eyes.
Results
Delayed filling was present in 75 (7.7%) of 973 eyes at baseline. Eyes with incident delayed filling at 1 year (23 [2.9%] of 798) showed a mean decrease of 1.7 letters in visual acuity, whereas eyes without incident delayed filling had a mean improvement of 8.1 letters (difference [Δ], −9.8; 95% confidence interval [CI] , −15.8 to −3.9; P < .01). Eyes with incident delayed filling had a larger increase in mean total lesion area of choroidal neovascularization (3.00 mm 2 ) than eyes without incident delayed filling (0.56 mm 2 ; Δ , 2.4; 95% CI, 0.4 to 4.4; P = .02). The proportion with incident delayed filling at 1 year was similar among eyes treated with ranibizumab (10 [2.4%] of 413) or bevacizumab (13 [3.3%] of 385; P = .53) and among eyes treated monthly (12 [3.1%] of 388) or as needed (11 [2.7%] of 410; P = .83).
Conclusions
Delayed patchy choroidal filling was uncommon at baseline. Although only a small percentage of eyes demonstrated delayed filling during the first year of anti–vascular endothelial growth factor treatment, these eyes had worse visual acuity and a larger increase in total lesion area of choroidal neovascularization.
The pathogenesis of age-related macular degeneration is believed to be a multifactorial process involving genetic predisposition, inflammatory mediators, oxidative stress, and hypoxia-induced angiogenesis. Although the interplay of these factors remains elusive, increasing evidence points toward an underlying ischemic process as a major contributing factor to age-related macular degeneration (AMD) pathogenesis. Abnormalities of the choroidal circulation are associated with the development of choroidal neovascularization (CNV) in patients with AMD by a process that may involve ischemia, hypoxia, and resultant vascular endothelial growth factor (VEGF) production.
Choroidal circulation abnormalities, as displayed by fluorescein and indocyanine green angiography, color Doppler flowmetry, laser Doppler flowmetry, pulsatile ocular blood flow, and histopathologic analyses, have been associated with progression of AMD. The choroidal circulation plays a vital role in both the provision of oxygen and nutrients to the outer retina, as well as removal of metabolic waste products. Impairment of these functions may contribute to accumulation of Bruch’s membrane deposits, retinal atrophy, and CNV.
The results from several previous studies suggested a role for decreased choroidal blood flow in the development of CNV in AMD. Decreased choroidal blood flow is associated with many AMD risk factors, including extent of drusen, retinal hyperpigmentary changes, hypertension, and hyperopia. Furthermore, in a longitudinal study of choroidal blood flow in AMD patients, Metelitsina and associates reported that patients who demonstrated CNV during the study had lower choroidal blood flow at baseline than those who did not demonstrate CNV. In addition, choroidal blood flow decreased before CNV formation, suggesting a role for ischemia in the development of CNV. Finally, patients with lower choroidal blood flow at baseline were 3 times more likely to experience decreases in visual acuity during the study.
A different line of evidence pointing to a role of decreased choroidal blood flow and ischemia in the pathogenesis of CNV can be derived from reports suggesting that in exudative AMD patients, there is a tendency for CNV to form in proximity to angiographically documented watershed areas where blood flow may be diminished. In addition, Stefánsson and associates recently noted features of AMD that may contribute to abnormal retinal oxygen metabolism and potentially ischemia, including the presence of confluent drusen, retinal elevation, retinal edema, and vitreoretinal adhesion.
Delayed filling of the choroidal lobules during the transit phase of fluorescein angiography, or delayed patchy choroidal filling, may be related to decreased choroidal circulation, as seen in choroidal ischemia secondary to vascular diseases. In this study, we investigated the association of delayed patchy choroidal filling with morphologic and functional outcomes after anti-VEGF treatment in participants in the Comparison of Age-Related Macular Degeneration Treatments Trials (CATT).
Methods
Institutional review board approval was obtained from the University of Pennsylvania and all participating CATT clinical centers. Each participant provided written informed consent that complied with the Health Insurance Portability and Accountability Act before entry into the study. Details regarding the methodology of the CATT study, an interventional double-masked trial, have been reported previously and also can be reviewed at ClinicalTrials.gov (identifier, NCT00593450). Below is a description of the methodology involved in this cohort study within the CATT pertaining to the fluorescein angiography evaluation of choroidal filling in study participants.
Study Participants
A total of 1185 participants from 43 clinical centers in the United States were enrolled in the CATT between 2008 and 2009. After written consent was obtained, study participants were randomized to 1 of the 4 treatment groups: (1) ranibizumab monthly, (2) bevacizumab monthly, (3) ranibizumab pro re nata (PRN), and (4) bevacizumab PRN. Inclusion criteria for the study included the presence of active CNV, subfoveal involvement by CNV or sequelae of CNV, fibrosis of less than 50% of total lesion area, visual acuity of 20/25 to 20/320, age of 50 years or older, and at least 1 drusen (>63 μm) in either eye or late AMD in the fellow eye. Exclusion criteria included previous treatment for CNV in the study eye, other progressive retinal disease likely to compromise visual acuity, as well as any contraindications to injections with ranibizumab or bevacizumab.
Study Procedures
During the initial visit to the participating centers, several baseline characteristics were recorded including age, gender, race, smoking status, systolic and diastolic blood pressures, and use of antihypertensive medications. Visual acuity was measured in both eyes after refraction using the Electronic Visual Acuity Tester following the protocol used in the Diabetic Retinopathy Clinical Research Network. Systemic hypertension was defined as a systolic blood pressure of 160 mm Hg, diastolic blood pressure of 95 mm Hg, or current use of antihypertensive medications.
Trained photographers followed a standard protocol to obtain color fundus photographs and fluorescein angiograms before initiation of anti-VEGF therapy and at 1 year of follow-up. All photographs were digital with the exception of 1 site that carried out film-based imaging.
Graders at the CATT fundus photograph reading center reviewed color photographs and fluorescein angiograms of study eyes (1 study eye per subject) and recorded the presence or absence of CNV, CNV lesion type (ie, predominantly classic, minimally classic, occult only, and cannot grade or no lesion), and retinal angiomatous proliferation. The total CNV lesion area was measured at baseline by taking into consideration the area of CNV plus any associated hemorrhage, scarring, blocked fluorescence, or pigment epithelial detachment associated with neovascularization. At follow-up, areas of geographic atrophy in areas previously occupied by the CNV lesion were included in the measurement to the total CNV lesion area. Graders at the CATT optical coherence tomography (OCT) reading center reviewed time-domain OCT images and recorded the presence of intraretinal fluid, subretinal fluid, or subretinal pigment epithelial fluid. Total retinal thickness in micrometers was measured at the foveal center based on the average of 6 fast macular OCT scans.
For the evaluation of the choroidal filling pattern, 2 trained readers (D.Y.G., C.P.O.) in the CATT photograph reading center initially determined whether delayed patchy choroidal filling was present at baseline and 1 year after anti-VEGF treatment. Nonphysiologic delayed patchy choroidal filling was deemed present if at least half a disc diameter of patchy choroidal filling was present beyond the early venous transit phase. The early venous transit phase was defined as lamellar filling with fluorescein that comprised less than half of the diameter of the larger retinal venules. At least 2 clear transit frames were required to evaluate a series. In equivocal cases, a third senior grader (J.E.G.) adjudicated. Photographs from the first 5% of patients were graded by both trained readers to assess intergrader agreement with respect to presence of delayed patchy choroidal filling (yes/no/can’t grade; n = 118 photographs; 94.1% agreement; κ, 0.89; 95% confidence interval [CI], 0.82 to 0.97), and subsequent to the determination of acceptable agreement, all patient photographs were assigned randomly to only 1 grader. Only the 1118 eyes that had undergone angiography at both baseline and 1 year were graded for delayed patchy choroidal filling. All assessments were performed without access to demographic data, treatment allocation, and clinical outcomes.
Data Analysis
The comparisons between groups of eyes based on presence of delayed patchy choroidal filling were performed for baseline characteristics and morphologic and functional outcomes at 1 year using the Fisher exact test for proportions and the independent t test for means. Similar comparisons were made between groups of eyes based on development of delayed patchy choroidal filling within 1 year. The McNemar test was used for comparisons of delayed patchy choroidal filling in the same eyes over time. All data analyses were performed using Statistical Analysis Software version 9.3 (SAS, Inc, Cary, North Carolina, USA).
Results
Baseline Characteristics of Study Participants
Among the 1118 graded eyes, 973 (87.0%) were gradable for delayed patchy choroidal filling, and 75 (7.7%) of these had evidence of delayed patchy choroidal filling at baseline. Figure shows a typical case of delayed patchy choroidal filling.
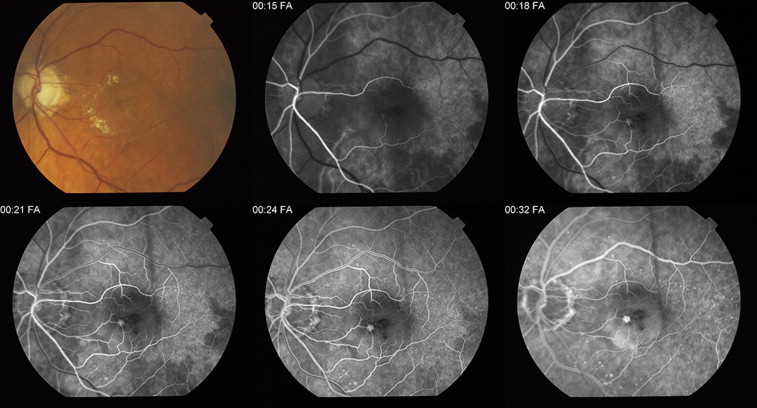
The mean age was 79.3 years in patients with baseline delayed patchy choroidal filling and 78.8 years in patients with normal baseline choroidal filling; this difference was not statistically significant (difference [Δ], 0.5; 95% CI, −1.3 to 2.2; P = .62; Table 1 ). The mean baseline visual acuity was 62.1 letters in those with baseline delayed patchy choroidal filling and 61.3 letters in those without baseline delayed patchy choroidal filling; this difference was not significant (Δ, 0.8; 95% CI, −1.3 to 2.2; P = .61). There were no statistically significant differences in gender, race, smoking status, hypertension, CNV area, CNV lesion type, or retinal angiomatous proliferation lesion type and OCT total retinal thickness between patients with and those without baseline delayed patchy choroidal filling.
| Characteristics | Baseline Delayed Patchy Choroidal Filling | P Value | ||
|---|---|---|---|---|
| No (n = 898) | Yes (n = 75) | Difference a : Yes vs No (95% CI) | ||
| Age (y) | ||||
| Mean (95% CI) | 78.8 (78.3 to 79.3) | 79.3 (77.6 to 81.0) | +0.5 (−1.3 to 2.2) | .62 |
| Sex, no. (%) | ||||
| Male | 344 (38.3) | 29 (38.7) | 1.00 | |
| Female | 554 (61.7) | 46 (61.3) | −0.4% (−11.8% to 11.1%) | |
| Race, no. (%) | ||||
| White | 886 (98.7) | 74 (98.7) | 0.0% (−2.7% to 2.7%) | 1.00 |
| Other | 12 (1.3) | 1 (1.3) | ||
| Smoking, no. (%) | ||||
| Never | 384 (42.8) | 26 (34.7) | .09 | |
| Past | 435 (48.4) | 37 (49.3) | ||
| Current | 79 (8.8) | 12 (16.0) | +7.2% (−0.1% to 15.7%) | |
| Mean blood pressure (95% CI), mm Hg | ||||
| Diastolic | 75.5 (74.9 to 76.2) | 74.0 (71.8 to 76.2) | −1.5 (−0.8 to 3.9) | .20 |
| Systolic | 135 (134 to 136) | 134 (130 to 138) | −1.0 (−3.2 to 5.2) | .65 |
| Hypertension, no. (%) | ||||
| No | 267 (29.7) | 28 (37.3) | .19 | |
| Yes | 631 (70.3) | 47 (62.7) | −7.6% (−18.9% to 3.7%) | |
| Visual acuity (letters) | ||||
| Mean (95% CI) | 61.3 (60.4 to 62.1) | 62.1 (59.1 to 65.1) | +0.8 (−2.3 to 3.9) | .61 |
| Area of CNV (mm 2 ) | ||||
| Mean (95% CI) | 4.4 (4.1 to 4.7) | 5.3 (4.1 to 6.5) | +0.9 (−0.2 to 2.0) | .10 |
| Total area of CNV lesion (mm 2 ) | ||||
| Mean (95% CI) | 5.8 (5.5 to 6.2) | 6.1 (4.8 to 7.3) | +0.2 (−1.1 to 1.5) | .71 |
| Lesion type, no. (%) | ||||
| Predominantly classic | 204 (22.7) | 15 (20.0) | .24 | |
| Minimally classic | 144 (16.0) | 19 (25.3) | ||
| Occult only | 533 (59.4) | 40 (53.3) | −6.0% (−17.8% to 5.7%) | |
| Can’t grade/no lesion | 17 (1.9) | 1 (1.3) | ||
| RAP lesion, no. (%) | ||||
| None/questionable | 786 (88.7) | 64 (87.7) | .70 | |
| Yes | 100 (11.3) | 9 (12.3) | +1.0% (−6.8% to 8.9%) | |
| OCT total retinal thickness (μm) | ||||
| Mean (95% CI) | 456 (444 to 468) | 440 (405 to 476) | −15.1 (−57.8 to 27.6) | .49 |
a For categorical variables, difference is listed for only 1 level and is listed next to the selected level.
When we compared the 1-year outcomes between eyes without delayed patchy choroidal filling at baseline and eyes with delayed patchy choroidal filling at baseline, we also found no statistically significant differences in visual acuity, total area of CNV lesion, presence of fluorescein leakage, total OCT retinal thickness, presence of OCT fluid, presence of scar, and geographic atrophy ( Table 2 ).
| Outcomes | Baseline Delayed Patchy Choroidal Filling | P Value | ||
|---|---|---|---|---|
| No (n = 898) | Yes (n = 75) | Difference: Yes vs No (95% CI) | ||
| Visual acuity (letters), mean (95% CI) | ||||
| 1 y | 69.0 (67.8 to 70.1) | 70.4 (66.3 to 74.5) | +1.4 (−2.9 to 5.7) | .52 |
| Change from baseline | 7.6 (6.6 to 8.5) | 8.5 (5.3 to 11.7) | +0.9 (−2.7 to 4.6) | .61 |
| Total area of CNV lesion (mm 2 ), mean (95% CI) | ||||
| 1 y | 6.6 (6.1 to 7.0) | 6.5 (5.1 to 7.8) | −0.1 (−1.8 to 1.5) | .90 |
| Change from baseline | 0.75 (0.37 to 1.12) | 0.25 (−0.79 to 1.28) | −0.5 (−1.9 to 0.9) | .47 |
| FA leakage at 1 y, no. (%) | 386 (45.6) | 31 (47.0) | +1.3% (−11.2% to 13.8%) | .90 |
| OCT total retinal thickness (μm), mean (95% CI) | ||||
| 1 y | 287 (278 to 296) | 279 (249 to 309) | −8.3 (−42.1 to 25.5) | .63 |
| Change from baseline | −168 (−180 to −156) | −169 (−209 to −129) | −1.1 (−45.4 to 43.2) | .96 |
| OCT fluid at 1 y, no. (%) | 587 (69.9) | 45 (69.2) | −0.7% (−12.3% to 11.0%) | .89 |
| Fibrotic scar at 1 y, no. (%) | 165 (19.9) | 11 (16.7) | −3.2% (−12.6% to 6.2%) | .63 |
| Any scar at 1 y, no. (%) | 273 (32.9) | 20 (30.3) | −2.5% (−14.1% to 9.0%) | .79 |
| GA at 1 y, no. (%) | 45 (5.7) | 5 (8.3) | +2.7% (−4.5% to 9.8%) | .39 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


