Cranial Nerve Palsies
Paul R. Mitchell
Marshall M. Parks*
*Marshall M. Parks, M.D. (1916–2005), was the original author of this chapter first published in Duane’s Clinical Ophthalmology in 1976 and revised several times under his guidance. His name is retained as a coauthor on this revision out of respect for his work with this chapter and his enormous contributions to the field of strabismus and amblyopia.
The Editors
A palsy of any one of three motor cranial nerves (CNs) that supply the extraocular muscles presents with characteristic findings affecting ocular motility. A single nerve may be involved; there may be a degree of bilateral involvement in either the third, fourth, or sixth CNs; or various combinations of them may be involved in the lesion. The palsy may be congenital, caused by some defect in the development of the nucleus or motor nerve fibers, or acquired. The lesion is located in or beyond the nucleus. If the motor fibers are affected, they may be interrupted either within or without the medulla; if outside the medulla, involvement may be intracranial (within the foramen through which the cranial nerve exits from the cranium) or extracranial (intraorbital).
The distribution and etiology of third, fourth, and sixth CN palsy at the Mayo Clinic have been reported by Rucker in 19581 and 19662 and Rush and Younge in 19813 (Table 19-1). Sixth CN palsy was noted in 45% of the cases, third CN palsy in 30%, fourth CN palsy in 11%, and involvement of multiple CNs in 14%. Each series of 1,000 patients, of all ages, was classified into six broad categories of CN-palsy etiology: undetermined, head trauma, neoplasm, vascular, aneurysm, and other. In the most recent series, the etiology remained undetermined in 26% of the cases, or was a result of head trauma in 20%, vascular in 17%, neoplastic in 14%, other sources in 15%, and aneurysm in 7%. These series are not typical of a general ophthalmology experience because they review cases of patients treated at a large referral center by ophthalmologists, neurologists, and neurosurgeons. Nevertheless, the scope of these series provides important data in the etiology and classification of CN palsies.
TABLE 19-1. Patients with Cranial Nerve Paralysis | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
A study at the same institution by Holmes et al4 evaluated the population-based incidence of pediatric third, fourth, and sixth CN palsies. In a 15-year period, 36 cases of CN palsies were identified in 35 children younger than 18 years of age. In contrast with the three previous series of 1.000 patients each, of all ages, this pediatric series noted the most commonly affected CN was the fourth, in 36%, followed by the sixth in 33%, third in 22%, and multiple nerve involvement in 9%. The most common cause for third and fourth CN palsy was congenital, for sixth CN palsy was undetermined, and for multiple CN palsies was trauma (Table 19-1).
Third, fourth, and sixth CN palsies following closed head injury were reviewed in a consecutive series of 210 patients by Dhaliwal et al.5 The control group sustained closed head injury without traumatic third, fourth, or sixth CN palsy. Those patients with closed head injuries and traumatic third, fourth, or sixth CN palsy had significantly higher severity of head injury, more abnormalities on CT, and worse short-term neurologic outcomes as compared with the control group. In comparing the CN injury subgroups, those with third CN palsy sustained the most severe head injury, those with fourth CN palsy had an intermediate level of head injury, and those with sixth CN palsy had the lowest level of head injury. There was not a consistent association of the location of the imaging abnormalities and which cranial nerve was damaged.
A series of 31 consecutive patients6 with ocular motor nerve palsies revealed sixth nerve paralysis as the most common at 38.4% and third nerve at 35.3%. Of the third nerve cases, 63.2% had pupil sparing. The most common etiologies were vascular, 25.8%; otorhinolaryngologic diseases, 19.7%; and trauma, 12.8%. MRI was not available, but CT scans did not reveal any abnormality in 54.8% of patients.
Improvements in MRI now provide the opportunity for detailed study of the functional anatomy of extraocular muscles and cranial nerves in the orbits of living subjects, and it is possible to image cranial nerves against the surrounding cerebrospinal fluid as they exit the brainstem.7,8,9,10,11 Demer et al11 in a prospective study evaluated cranial nerves in 83 orthotropic volunteers and 96 patients with strabismus. In all normal subjects, the third and sixth cranial nerves, but not the fourth cranial nerve, were demonstrable in the orbit and skull base. Patients with congenital third nerve palsies had hypoplastic nerves both in the orbit and skull base, along with hypoplasia of the involved extraocular muscles. The patients with chronic fourth and sixth CN palsies demonstrated atrophy of the involved extraocular muscles. With Duane syndrome, patients demonstrated absence or hypoplasia of the sixth cranial nerve in both orbit and brainstem areas, often with mild hypoplasia and apparent misdirection of the third cranial nerve to the lateral rectus muscle. In contrast to patients with sixth nerve palsy, there was no extraocular muscle hypoplasia in Duane syndrome. With congenital fibrosis syndrome, there was severe hypoplasia of the third cranial nerve, moderate hypoplasia of the sixth cranial nerve, and extraocular muscle hypoplasia, especially severe for the superior rectus and levator muscles. They concluded that direct imaging of cranial nerves and extraocular muscles by high resolution magnetic resonance imaging is feasible and useful in the differential diagnosis of complex strabismus. A subsequent report by Demer et al10 was a prospective study effort to learn the effects of cranial nerve abnormalities on the functional anatomy of the extraocular muscles and to directly image these cranial nerves. They confirmed that modern MRI techniques can directly demonstrate cranial nerve pathology as a cause of strabismus. In some cases, MRI defined bilateral but asymmetric involvement, which was more severe on the clinically apparent side. Hence, clinically normal versions do not rule out subtle cranial nerve anomalies innervating extraocular muscles.
Multiple cranial nerve palsies have been reported in association with idiopathic intracranial hypertension in primary antiphospholipid syndrome with bilateral sixth, left third and fourth palsies,12 unilateral third, fourth, and sixth palsy in association with type 2 diabetes,13 and right third and fourth palsy and bilateral sixth nerve palsy in association with hepatocellular carcinoma metastasizing to the base of the skull.14
THIRD CRANIAL NERVE PALSY
Third CN palsy in children is more frequently a congenital disorder than an acquired disorder, whereas acquired third CN palsies appear more frequently in adults than in children.
However, in a retrospective study, Ing et al15 evaluated 54 children with oculomotor nerve (CNIII) palsy, presenting over a period of 21 years, and found 38 isolated third CN lesions and 16 with additional CNs involved. Only 11 cases were congenital, and of the 43 that were acquired, 31 were traumatic, 7 related to infection, 2 ophthalmoplegic migraine, 2 neoplastic, and 1 vascular-hypertensive. In contrast to studies with adults, this series did not include any third CN palsy secondary to aneurysm, diabetes, metastatic tumors, or pituitary lesions.
CONGENITAL
Congenital third CN palsy presents with varied degrees of extraocular involvement. Intraocular musculature is not usually affected in congenital third CN palsy, although pupil constriction may occur on attempted adduction in some cases of aberrant regeneration.16,17
The involved pupil may even be smaller than that in the uninvolved eye in congenital third CN misdirection syndrome.18 Although congenital third CN palsies are considered to be benign and isolated, Balkan and Hoyt19 reported other signs of focal neurologic damage, including pupillary involvement, oculomotor synkinesis, hemiplegia, seizures, and developmental delay. These congenital third CN palsies probably occur as a result of damage to both the peripheral nerve and the brainstem.19
Balkan and Hoyt found neurologic involvement or developmental delay in 7 of 10 patients studied. Hamed20 described neurologic involvement in 10 of 14 cases with congenital oculomotor palsy, and Tsaloumas and Willshaw21 reported 5 of 14 patients with congenital oculomotor palsy with significant neurologic abnormalities, including 2 patients using digital lid elevation to allow fixation with their affected eye.
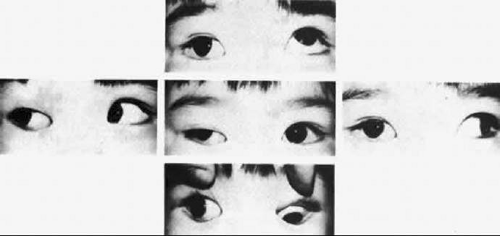
The degree of involvement of the levator muscle varies, but some function is usually retained. Therefore, ptosis is variable in this form of third CN palsy. The four extraocular muscles innervated by the third CN are also affected in various degrees. However, there is usually some trace at least of weakness of the medial, inferior, and superior rectus muscles and of the inferior oblique muscle. The fourth CN is uninvolved and, consequently, the involved eye is exotropic and hypotropic (Fig. 19-1). Therefore, the clinician should always suspect congenital third CN palsy in an exotropic patient who has one low eye, intact pupillary and accommodation responses, and minimal ptosis of the involved eye with varied degrees of limitation of both elevation and depression in addition to diminished adduction. Many of these patients are able to develop single binocular vision and to maintain a compensatory malposition of the head that allows the alignment of the eyes to serve this purpose. When the eyes are moved into a position in which fusion is not possible, these patients experience diplopia if they have binocular vision with torticollis. Amblyopia of either eye may occur if the patient does not have binocular vision and does not maintain torticollis.
Schumacher-Feero et al22 reported a series of 49 children with third CN palsy, involving 53 eyes, and observed during a follow-up for a mean of 5.5 years. Amblyopia developed in 27 eyes, and at the last follow-up visit, in 56% of affected eyes the visual acuity ranged from 20/15 to 20/40. Binocular function was difficult to restore or to preserve but was significantly improved after surgery. Horizontal alignment was initially good in 6 of 49 patients, improving to 30 with good alignment at their last evaluation. The vertical alignment improved from 24 initially to 35 with good alignment at their last evaluation. Only one child achieved fusion at distance and near after a single recession/resection procedure at 8 months of age. A complete palsy required a mean of 2.3 operations to align the eyes, and a partial third CN palsy required a mean of 1.5 operations over the 5.5-year period. In general, the surgery was a horizontal recession/resection procedure for exotropia, with graded supraplacement of the horizontal rectus insertions for the hypotropia.
The cause of congenital third CN palsy is unknown, but it is presumed to be a result of a developmental defect in either the nuclear or the motor fiber portion of the third CN complex that innervates the levator muscle and the extraocular muscles. It is not an extremely rare motility disorder. We have seen many patients having only unilateral involvement.
As in any third CN palsy, the absence of adduction of the involved eye makes it difficult to determine the intactness of the ipsilateral fourth cranial nerve. Clinically, the method used is to observe the crypts of the iris while the involved eye remains abducted and to ask the patient to look upward and downward. If the fourth CN is intact, the iris markings reveal a conspicuous intorsion as infraduction is attempted and extorsion as supraduction of the involved eye is attempted (Fig. 19-2). Dieterich and Brandt23 measured ocular torsion and subjective visual vertical tilts in acute and chronic oculomotor, trochlear, and abducens nerve palsies for each eye separately in the primary position with the head held upright. Unexpectedly, ocular torsion was abnormal in only 32% of third and fourth CN palsies involving oblique eye muscles, and normal in all abducens palsies. When measurable, pathologic ocular torsion was low, from 2° to 8°, monocular, involving either the paretic or nonparetic eye. Subjective visual vertical tilts were abnormal in 67% of third and fourth CN palsies, mostly low in amplitude, between 1° and 6°, and involving either the paretic or nonparetic eye, depending on the duration of the palsy. In contrast with acute unilateral brainstem lesions with frequent binocular and conjugate tilts, third and fourth CN palsies cause only minor and unpredictable ocular torsion and subjective visual vertical tilts.
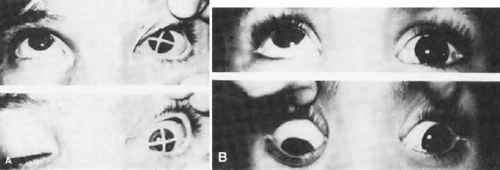 Figure 19-2. Intact fourth cranial nerve in third cranial nerve palsy. A. Litmus paper marker on cornea. B. Pigmented scleral spots demonstrate intorsion as depression is attempted. |
The forced duction test produces a negative result in third CN palsy; this rules out any adhesive phenomenon that limits motility of the eye. The degree of involvement of the third CN determines whether therapy is indicated. Involvement may be so minor and partial that no therapy is necessary, or it may affect only the elevators of an eye and is therefore known as double elevator palsy. Cadera et al24 studied pathophysiology of double elevator palsy in two patients with magnetic resonance imaging (MRI) with volume scanning technique. They found the volume of the superior rectus muscle on the affected side to be less than half that of the normal eye, with other rectus muscles normal, suggesting either congenital hypoplasia or paresis of the affected superior rectus muscle. The inferior oblique muscles could not be evaluated by MRI. After a Knapp procedure in both patients, only minimal superior displacement of the medial and lateral rectus muscles was detectable posterior to the equator of the globe in both patients with MRI.
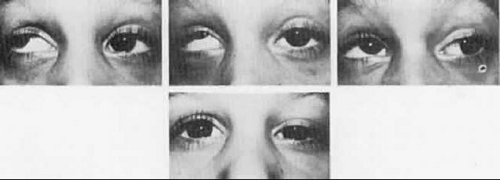
Double elevator palsy is usually rather complete, and it may also be associated with various degrees of ptosis (Fig. 19-3). The ptosis may be only pseudoptosis because of the hypotropia and because the lid position follows that of the eye. Fixating with the hypotropic eye causes the complete disappearance of the pseudoptosis; however, there may be a small degree of bona fide ptosis in addition to the pseudoptosis. The traction test result is normal in double elevator palsy. Treatment of double elevator palsy involves transposition of the insertions of the horizontal rectus muscles, placing the new insertions immediately adjacent to the insertion of the superior rectus muscle (Fig. 19-4). This does not produce normal elevation beyond the midline level, but it renders considerable improvement in, if not total elimination of, the hypotropia caused by this disorder.
After a full tendon transfer of the lateral and medial rectus muscle for double depressor or double elevator palsy, Knapp obtained an average correction of 38 diopters in the primary position and movement of 25° in the field of action of the paretic muscle group from the primary position.25 In the presence of a poor or absent Bell phenomenon, an accentuated lower lid fold of the hypotropic eye in attempted elevation, and a positive traction test result, Scott and Jackson26 recommend inferior rectus recession only as the initial procedure. With a negative forced traction test result, a full Knapp procedure is advised.
Complete congenital third CN involvement requires surgery for exotropia, hypotropia, and ptosis. Hypotropia is resolved by disinserting the tendon of the superior oblique muscle from the globe, which is tight and contracted. Maximal recession of the lateral rectus and resection of the medial rectus may be sufficient to reposition the involved eye in the horizontal plane satisfactorily. However, if this is inadequate, removing the superior oblique tendon from the trochlea, severing the reflected tendon of the superior oblique muscle from the muscular portion, and attaching the superior oblique muscle to the sclera at the insertion of the medial rectus muscle offer excellent correction of the horizontal defect created by the third CN palsy in the primary position.
This does not create normal adduction of the involved eye but is an effective technique for centering the eye.27 However, following transposition of the superior oblique muscle, when the patient depresses the involved eye, it adducts. Saunders and Rogers,28 who attempted correction of third CN palsy by superior oblique anterior transposition and advancement without trochleotomy, reported unsatisfactory results because of inadequate horizontal alignment, postoperative hyperdeviations, or paradoxical ocular movements. Superior oblique tendon transposition with trochleotomy causes the adherence syndrome, owing to violation of the Tenon capsule, which is unavoidable in removing the superior oblique tendon from the trochlea. Therefore, this procedure is no longer recommended. Scott29 and Gottlob et al30 described anterior transposition of the nasal portion of the superior oblique tendon, 2 to 3 mm anterior to the nasal border of the superior rectus muscle, without trochleotomy. In addition, Gottlob et al performed large recessions of the ipsilateral lateral rectus, and in some patients, a recess-resect procedure on the horizontal rectus muscles of the contralateral eye. Orthophoria was achieved in four patients, a 10 prism diopter (PD) residual exotropia in one patient, and two patients required reoperations because of aberrant regeneration of the oculomotor nerve. In a much larger series, Maruo et al31 compiled 280 cases of exotropia secondary to oculomotor palsy, between 1971 and 1993. There were 130 congenital and 150 acquired cases of oculomotor palsy. Surgery was performed 234 times in 138 patients with paralytic exotropia, with transposition of the superior oblique tendon and resection of the medial rectus muscle, with or without recession of the ipsilateral lateral rectus muscle. With a 4-year follow-up in 35 cases, the authors found similar results when transposing the superior oblique tendon in patients with complete palsy and in resection of the medial rectus muscle in patients with incomplete palsy. There was no benefit adding a resection of the medial rectus when the superior oblique transposition was performed. However, recession of the lateral rectus muscle greatly improved the effectiveness of either the superior oblique transposition or the medial rectus resection. Mudgil and Repka32 reviewed retrospectively the ophthalmologic outcome of third CN palsy or paresis in 41 children younger than 8 years of age. Etiologies included congenital in 39%, traumatic in 37%, and neoplastic in 17%. Initial visual acuities were reduced in 71%, and the long-term outcome in the 20 who could be observed during follow-up, for a mean of 3.6 years, was reduced vision in 35% because of amblyopia, and in 25% because of nonamblyopic factors. In the congenital third CN palsy group, all patients improved to normal visual acuity. Despite improved alignment after surgery in 40% (8 of 20) of children with long-term follow-up, none obtained measurable stereopsis. Aberrant reinnervation occurred in 45% (9 of 20). Only 3 patients fully recovered and regained measurable stereopsis, with the etiologies of congenital, neoplastic, and traumatic third CN palsies. Kose et al33 described an approach to surgical management of total third nerve palsy by a hemi-hangback recession of the lateral rectus muscle and a resection of the medial rectus muscle for exotropia. Depending on the amount of vertical deviation, the horizontal rectus muscle insertions were supraplaced, either alone or in addition to a hemi-hangback recession of the inferior rectus. This technique did not affect the normally functioning superior oblique and lateral rectus muscles.
Because of disruption in sensory fusion mechanisms, Elston34 has recommended a one-stage procedure of maximal lateral rectus recession, medial rectus resection, and simultaneous transposition of insertions to that of the superior rectus. The simplest procedure providing predictable cosmetic improvement has been recommended. Ptosis, however, was not addressed. A frontalis suspension of the ptotic lid is the indicated procedure for the associated ptosis. A synthetic material (e.g., 4-0 Supramid) is ideal because if the cornea cannot tolerate the relatively dry state after the lid is elevated, these synthetic sutures can easily be removed and the cornea not harmed permanently. The surgeon should be aware of the absence of the Bell phenomenon in these patients and be alert to postoperative corneal problems associated with this deficiency.
ACQUIRED
Acquired third CN palsy may be partial or complete and may involve only the extraocular muscles or both intraocular and extraocular muscles. The pupil is usually spared in third CN palsy associated with diabetes.
Acquired third CN palsy usually occurs rather precipitously with maximal involvement. Within days to weeks, there may be an indication of restoration of third CN function manifest by only partial involvement. Recovery is usually complete by 6 months following onset, and, consequently, no judgment should be rendered regarding the necessity of treatment until after the 6-month interval. Partial or complete recovery can be expected, depending on the cause of the third CN palsy, in 48% of the patients.3 Many times such palsy results from relatively serious intracranial involvement, and this may determine whether therapy is indicated. Minor head trauma may produce isolated oculomotor nerve palsy in the presence of a normal CT, normal MRI, and normal magnetic resonance angiography (MRA).35 A case of mild head trauma created a complete left third nerve palsy, with normal CT and MRI of the brain and a normal CT of the orbit. Because of only partial recovery with time and with botulinum toxin, transposition surgery was required to reduce the strabismus.36 Mark37 recommends MRI evaluation of patients with third CN palsy to include proton density and T2-weighted images through the brain in axial section, to study the brainstem for nuclear lesions, along with thin section T1-weighted images in the coronal and axial planes to evaluate cisternal, cavernous, and orbital segments of the third nerve. Gadolinium diethylenepentaminetetraacetic acid has also been found helpful in evaluation of third CN palsy. In posttraumatic third CN palsies, use of gradient echo images to detect hemorrhage is helpful. Ischemia of the oculomotor nerve causes most cases of nontraumatic oculomotor nerve palsy.38 MRI and lumbar puncture are helpful in diagnosing cases caused by inflammatory or neoplastic meningitis. A cerebral aneurysm, which can be fatal, can be diagnosed by cerebral angiography, but this test has a 1% to 2% morbidity and mortality rate. Magnetic resonance angiography is a variant of MRI that highlights blood vessels; however, it is only 95% accurate in detection of aneurysms. Trobe38 therefore concludes that because pupil involvement occurs in 96% with aneurysms, and if anisocoria exceeds 2.0 mm, that catheter angiography is justified. In a series39 of 148 patients with posterior communicating aneurysm, 74 (50%) had concurrent unilateral oculomotor palsy. All patients had a craniotomy after the diagnosis using whole brain digital subtraction angiography. Surgery involved simple pedicle clipping of the aneurysm for the 40 patients in group A, and the 34 patients in group B had pedicle clipping and decompression of the oculomotor nerve. Surgery was timed at within 14 days from diagnosis, from 14 to 30 days, or 30 to 90 days in both groups. In both groups, all 35 patients undergoing surgery within 2 weeks had complete recovery. Four patients in group A and three in group B had incomplete recovery; therefore in group A 90% had complete recovery and 10% incomplete recovery, and in group B 91% had complete recovery and 9% incomplete recovery. There was no correlation found between decompression of the oculomotor nerve, either puncture or excision of the aneurysm, and postop recovery time.
The causes can be classified as follows:
VII. M.
Brainstem lesion
Benedikt syndrome manifested by homolateral third CN paralysis and contralateral intention tremor
Weber syndrome manifested by homolateral third CN paralysis and contralateral hemiplegia
Inflammatory conditions
Meningitis
Encephalitis
Polyneuritis from toxins such as alcohol, lead, arsenic, and carbon monoxide, and from diabetes
Temporal arteritis40
Herpes zoster infection41
Echovirus infection42
Vascular lesions
Pituitary Infarct (apoplexy)49
Carotid cavernous fistulas50
Orbital dural arteriovenous malformation51
Churg-Strauss syndrome (midbrain infarction)52
Demyelinating diseases
Multiple sclerosis57
Trauma
Miscellaneous
Partial third CN palsy in the form of isolated inferior rectus paresis may be the presenting sign of myasthenia gravis, with sudden onset of diplopia.75 A rare inferior division third nerve paresis involving the pupil caused by an intraorbital dural arteriovenous malformation has been reported.40 The oculomotor nerve aberrant regeneration or misdirection syndrome is believed to result from extensive and haphazard growth that characterizes the regeneration of injured nerve fibers.18,34 The third CN misdirection syndrome includes the following.18
Retraction of the globe on attempted vertical gaze
Adduction of the globe on attempted vertical gaze
Upper lid retraction on attempted downgaze (pseudo-Graefe sign)
Narrowing of the fissure on abduction, and widening of the fissure on attempted adduction (horizontal gaze lid dyskinesis)
Pseudo–Argyll Robertson pupil
Relative monocular vertical optokinetic responses
Miller and Lee82 described adult-onset acquired oculomotor nerve paresis with cyclic spasms in relation to ocular neuromyotonia in two patients, years after irradiation for intracranial tumors. Findings included unilateral eyelid retraction, ipsilateral esotropia with limited abduction during the spastic phase and with ipsilateral ptosis, exotropia, and variable limited adduction during the paretic phase. The cause of cyclic oculomotor paresis is usually unknown, and most authors speculate some element of aberrant regeneration after nuclear or nerve damage.83 In a case of third CN palsy with trigeminal sensory loss caused by herpes zoster, Quisling et al41 reported results of the brain MRI, which demonstrated enhancement and thickening of the cisternal and cavernous portions of the right third cranial nerve, and FLAIR MRI revealed high signal intensity in the right posterolateral medulla, consistent with presumed involvement of the trigeminal nucleus and tract. Oculomotor tremor (OMT) is a high-frequency small tremor of the eyes first described as one of the fixational eye movements in 1934 by Adler and Fliegelman.84 Bolger et al85 found reduced or absent OMT in six adult cases of acquired third nerve palsy, suggesting that innervation of the extraocular muscles is required for normal OMT activity and therefore OMT has a neurogenic origin.
Treatment involves relief of the patient’s diplopia, which usually is not a problem in complete third CN paralysis because of the associated ptosis covering the pupil. However, in partial involvement, the lid may sufficiently clear the pupillary space so that diplopia is a problem. Occlusion therapy is the best solution for the patient’s diplopia. The patient usually wishes to have the involved eye occluded rather than the uninvolved eye. Surgery is indicated for associated strabismus and ptosis if the patient’s general condition permits it and if a significant residual paralysis is present 6 months after onset of third CN palsy. The surgery described for congenital third CN palsy is also applicable for acquired third CN palsy. Kushner86 described surgical treatment in five patients with the rare finding of paralysis of the inferior division of the third CN. Clinical findings included a large exotropia and hypertropia of the affected eye, intorsion, and internal ophthalmoplegia. As described by Knapp,87 Kushner performed a superior oblique tenotomy along with transposing the superior rectus toward the insertion of the superior border of the medial rectus, following the spiral of Tillaux and transposing the lateral rectus toward the lateral border of the inferior rectus, following the spiral of Tillaux. In follow-up lasting between 3 and 10 years, all patients maintained satisfactory eye alignment and were free from diplopia in the primary position. In the presence of oculomotor palsy with aberrant regeneration, surgery on the nonparetic eye may be indicated.86 The eye deviation is measured with the dominant eye fixating, and on the contralateral, nonparetic eye, a lateral rectus recession and medial rectus resection is performed, along with downward transposition of the horizontal rectus muscles to offset the vertical deviation.
FOURTH CRANIAL NERVE PALSY
The most common isolated cyclovertical muscle palsy encountered by the ophthalmologist is involvement of the trochlear nerve. The cause of congenital trochlear nerve palsy is a defect in the nucleus or motor portion of the nerve; a defect in the motor nerve occurs either inside or outside the medulla. The most common cause of acquired fourth CN palsy is closed head trauma.1,2,3,87,88,89,90,91,92
Of 3,000 cases of strabismus reviewed by Khawam et al,87 closed head trauma ranging from minor to severe caused 68% of the 40 cases of acquired superior oblique palsy. Uncertain etiology was noted in 20%, and the remainder of the cases included cerebrovascular accident, diabetes, brain tumor, ethmoiditis, and mastoiditis. The cause of acquired bilateral superior oblique palsy following severe head injury is hemorrhage in the roof of the midbrain at the caudal end of the quadrigeminal plate, the area of decussation of the fourth CN.73 Even mild head trauma causing a mass lesion, a hematoma, may cause traumatic fourth CN palsy.91
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree