Purpose
To correlate postmortem histology with previously recorded multimodal imaging from a patient with type 1 neovascularization (NV) associated with an acquired vitelliform lesion in the setting of age-related macular degeneration (AMD).
Design
Case study.
Methods
Multimodal imaging that was obtained antemortem was matched with ex vivo and high-resolution histologic images of the preserved donor macula. Anatomic correlates for multimodal imaging findings were then defined.
Results
Spectral-domain optical coherence tomography (OCT) revealed a split in the retinal pigment epithelium–Bruch membrane band. Type 1 NV in this case was composed of 6 layered components: (1) retinal pigment epithelium, (2) basal laminar deposits, (3) fibrovascular membrane, (4) fibrocellular scar, (5) hemorrhage, and (6) Bruch membrane. The anatomic correlates for the hyporeflective band on spectral-domain OCT included a thick basal laminar deposit. Not all structures could be readily separated on the basis of their reflectivity patterns.
Conclusions
This is an important clinicopathologic correlation of NV secondary to AMD in the spectral-domain OCT era. Our findings of 6 layers include and extend the anatomic framework encapsulated by the double-layer and triple-layer signs. The resolution of current devices does not always permit distinction of the different layers of NV tissue. Thick basal laminar deposits may appear hyporeflective on spectral-domain OCT and may be confused with fluid from a neovascular process. It will be important to perform a larger clinicopathologic series to aid our anatomic interpretation of spectral-domain OCT images.
Choroidal neovascularization (NV) is a sight-threatening complication of age-related macular degeneration (AMD). The management of neovascular AMD has been greatly aided by spectral-domain optical coherence tomography (OCT), an imaging modality that utilizes the properties of light backscatter to provide fine detail about retinal and choroidal structures. Spectral-domain OCT permits anatomic localization of NV and also allows inference about the biological effects of vascular endothelial growth factor (VEGF) inhibitors on NV tissues localized to different compartments. In clinical practice, changes in spectral-domain OCT reflectivity patterns in the sub–retinal pigment epithelium (RPE), subretinal, and intraretinal compartments are used to judge response to anti-VEGF therapy and guide decisions concerning frequency of repeat therapy and interval review.
Hemorrhage, exudates, and fibrovascular tissue can alter the reflectivity patterns of near-infrared light and thus confuse the interpretation of spectral-domain OCT images. Previous authors have described pathognomonic reflectivity patterns, such as the double-layer and triple-layer signs, to signify active angiogenesis in polypoidal choroidal vasculopathy (PCV) and AMD. However, these designations have yet to be validated histologically. Resolving the anatomic correlation for the reflectivity patterns seen on spectral-domain OCT is important, as it may aid our ability to assess neovascular disease activity in vivo. It may also provide new information that can be used to identify indolent NV structures that may have previously been unrecognized.
Vitelliform lesions may be seen in eyes with AMD and may also be encountered in Best macular dystrophy, cuticular drusen, vitreoretinal interface disorders, peripherin (retinal degeneration, slow) mutation-related macular dystrophies, and other degenerative maculopathies including adult-onset foveomacular vitelliform dystrophy. In this report we present a clinicopathologic correlation of type 1 (sub-RPE) NV in the setting of an acquired vitelliform lesion in an eye that demonstrated the clinical and pathologic characteristics of AMD. We correlate multimodal imaging results including fundus photography, indocyanine green angiography (ICGA), and spectral-domain OCT with histopathologic findings. Novel postmortem eye-tracking techniques are employed to ensure accurate alignment of in vivo and ex vivo B-scans and facilitate reliable comparisons between spectral-domain OCT and histology. The findings in this study are expected to aid in the clinical management of AMD.
Methods
This case study complied with the Health Insurance Portability and Accountability Act of 1996 and followed the tenets of the Declaration of Helsinki. The institutional review board of the Manhattan Eye, Ear and Throat Hospital/North Shore Long Island Jewish Hospital approved the retrospective review of clinical notes and imaging data. This board plus the Institutional Review Board at the University of Alabama at Birmingham approved the histopathology study.
Clinical Care and Multimodal Imaging
Postmortem histopathologic examination was performed on the right eye of a long-term patient with AMD who had undergone serial examination and multimodal imaging as part of her clinical management. She had received care at Vitreous Retina Macula Consultants of New York for 11 years.
Ocular morbidities other than AMD included pseudoexfoliation glaucoma with stable intraocular pressure control. The right eye also developed cystoid macular edema following cataract extraction and anterior chamber intraocular lens insertion 6 years prior to her death. Edema resolved following treatment with topical steroid and nonsteroidal anti-inflammatory therapy.
Serial multimodal imaging of her clinical course included fundus autofluorescence, spectral-domain OCT, high-resolution color fundus photography, red-free imaging, near-infrared reflectance imaging, and ICGA. Spectral-domain OCT images were obtained using the Spectralis HRA + OCT (Heidelberg Engineering, Heidelberg, Germany). Eye-tracking and image registration software was enabled during acquisition of spectral-domain OCT images. Color fundus photographs and red-free, fundus autofluorescence, and ICGA images were obtained with the Topcon TRC-50XF (Topcon America, Paramus, NJ). The time period between the most recent image by an imaging modality and the patient’s death was 13 months for color photography, 13 months for red-free photography, 13 months for fundus autofluorescence imaging, 8 months for near-infrared reflectance imaging, 8 months for spectral-domain OCT, and 28 months for ICGA.
Histopathology
Personnel of the Eye Bank for Sight Restoration (New York, NY) recovered eyes 8 hours and 55 minutes after donor death. Globes were opened with encircling cuts at the limbus and immersed in 10% neutral buffered formalin. Following overnight shipping to Birmingham, eyes were transferred to 2% glutaraldehyde and 1% paraformaldehyde in 0.1 M phosphate buffer and subject to multimodal ex vivo imaging as described. Eye-tracking software (Spectralis; Heidelberg Engineering, Heidelberg, Germany) was used to align in vivo and ex vivo B-scans from preserved globes. Then, 8-mm-diameter full-tissue-thickness punches centered on the fovea were postfixed in osmium tannic acid paraphenylenediamine, embedded in epoxy resin, sectioned at 0.8 μm thickness, and stained with toluidine blue. This preparation makes lipid-containing material in basal linear deposits and soft drusen gray or brown and basal laminar deposits predominantly blue. Tissue was oriented during sectioning so that a line between the centers of the fovea and optic nerve head was always parallel to the edge of a diamond knife, aided by a fine line drawn across the resin block over these landmarks under a dissecting microscope. The distance between sections indicated by the controls of the ultramicrotome guided the correspondence to spectral-domain OCT scans. Histologic sections matched to the spectral-domain OCT scans were digitized in their entirety using image-stitching software (CellSens; Olympus, Center Valley, Pennsylvania, USA) and a 20× plan-apochromat objective. Correspondences between histology and spectral-domain OCT were verified by comparing images of entire sections to scans, with specific reference to the contour of the inner retinal surface, contour of the inner nuclear layer, and the horizontal extent of basal laminar deposits and split RPE–Bruch membrane complex. Matching histologic sections and OCT scans at each level are presented in the Supplementary Figure (available at AJO.com ). Selected areas were imaged using 60× oil immersion objectives (numerical aperture 1.40 and 1.42). Images were montaged, composited, and adjusted for exposure, contrast, and sharpness (Photoshop CS6; Adobe, San Jose, California, USA). Measurements of horizontal extents and thicknesses of histologic layers were made using the digital readout on the motorized microscope stage. Findings were compared to those from 52 similarly prepared advanced AMD eyes available at http://projectmacula , and to the descriptions of basal laminar deposits and RPE cells generated from these eyes. We previously reported clinicopathologic correlations of outer retinal tubulation and RPE degeneration in this patient.
Results
Clinical Course and Multimodal Imaging Findings
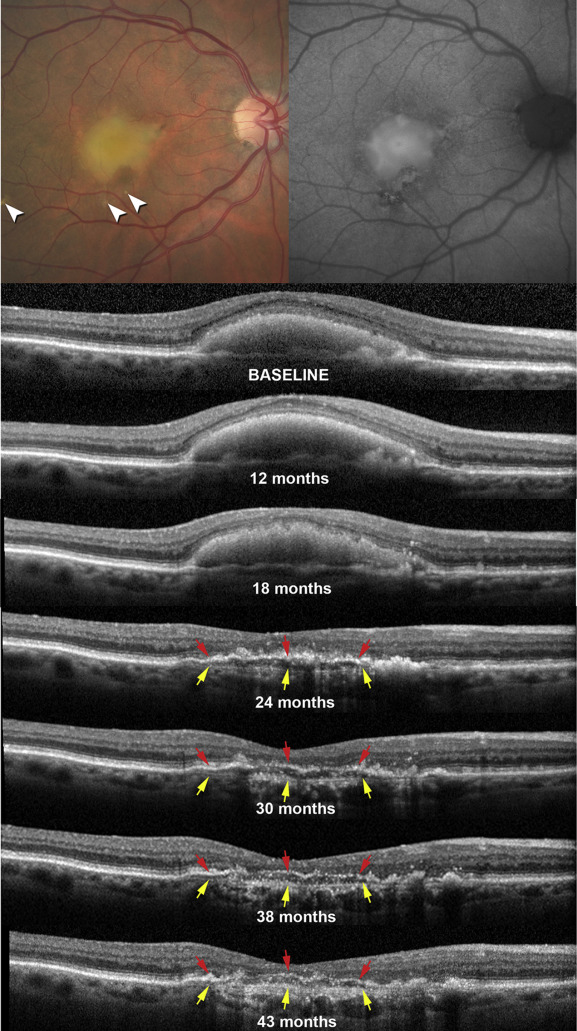
Presenting visual acuity in the right eye was 20/50. Baseline retinal examination revealed an acquired vitelliform lesion in the central macula, soft drusen, and focal areas of pigmentary changes ( Figure 1 , Top left), as well as reticular pseudodrusen in the superior macula ( Supplementary Figure , Left). There was no exudation, subretinal fluid, or hemorrhage at baseline examination.
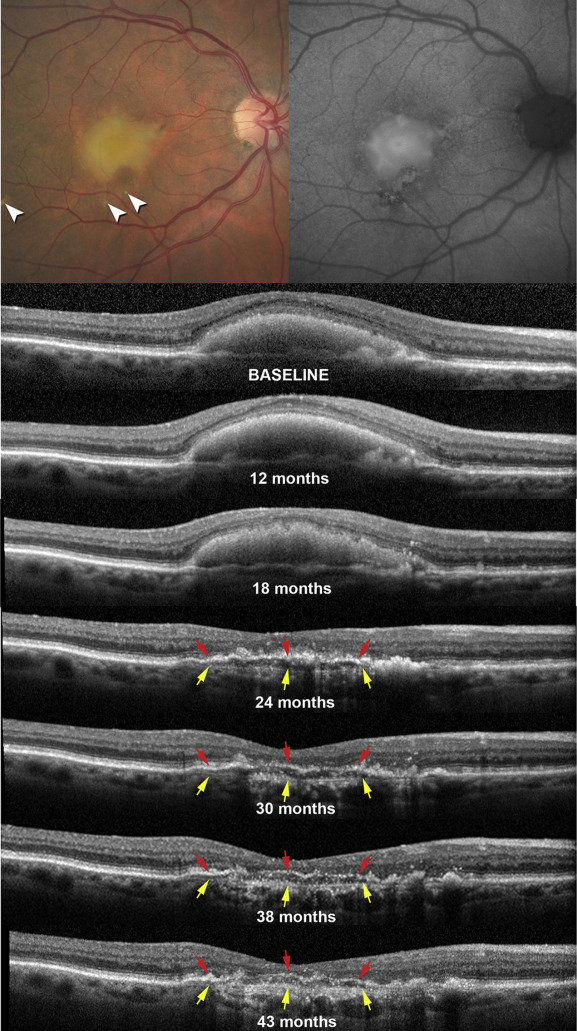
Fundus autofluorescence imaging demonstrated hyperautofluorescence that correlated with the acquired vitelliform lesion surrounded by variable autofluorescence that correlated with the pigmentary changes ( Figure 1 , Top right). The acquired vitelliform lesion was characterized by subretinal hyperreflective material on spectral-domain OCT, and reabsorption of this material occurred over the course of 2 years ( Figure 1 ). Following regression of the acquired vitelliform lesion, a hyporeflective band with punctate hyperreflective spots was observed to split the normally hyperreflective RPE–Bruch membrane complex. The RPE–Bruch membrane complex in this case had an irregular and sometimes interrupted hyperreflective band that correlated to the RPE ( Figure 1 , red arrows). Underlying this band was another band that correlated to a tissue complex (yellow arrows, Figure 1 ) notable for thin, closely spaced, and horizontally oriented hyperreflective lamellae. This distinctive signature was readily apparent on all spectral-domain OCT images from the point of acquired vitelliform lesion reabsorption to the final visit. We observed little change in the morphology, configuration, and reflectivity patterns of this finding between visits ( Figure 1 ), with the exception of the RPE band becoming thinner and discontinuous. The underlying tissue complex became more visible, presumably owing to greater light penetration subsequent to RPE loss. In contrast, the complex was just barely visible prior to the acquired vitelliform lesion collapse at 24 months.
Visual acuity remained at 20/400 following reabsorption of the acquired vitelliform lesion. Clinically, a legacy of central chorioretinal atrophy and pigmentary changes were evident at the site of the prior acquired vitelliform lesion. ICGA performed 12 months before the final visit showed an indeterminate area of central staining ( Figure 2 ), possibly consistent with type 1 NV. Intravitreal anti-VEGF injections were not administered during the course of this patient’s management. The death of the patient from gastric cancer occurred 8 months after her last clinic visit, where she was examined and also underwent spectral-domain OCT imaging.
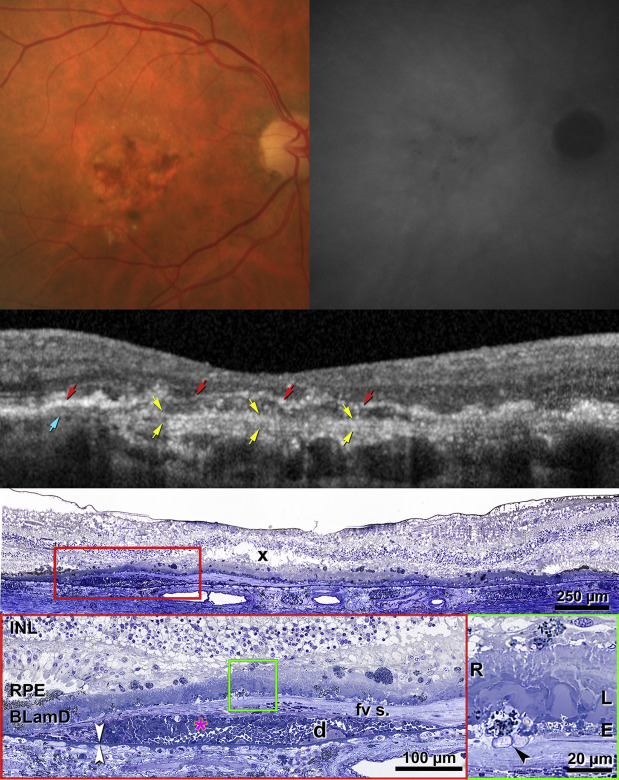
Histopathology and Spectral-Domain Optical Coherence Tomography Correlation
Contours of the inner retinal surface, inner nuclear layer, and RPE–Bruch membrane morphology were well aligned in histologic sections and in spectral-domain OCT scans ( Supplementary Figure ). Histopathologic findings are detailed in Figures 2–4 . Spectral-domain OCT scans and histologic sections covered the central 4.32 mm (superior to inferior diameter) of the macula and the histomorphometry is summarized in the Appendix .
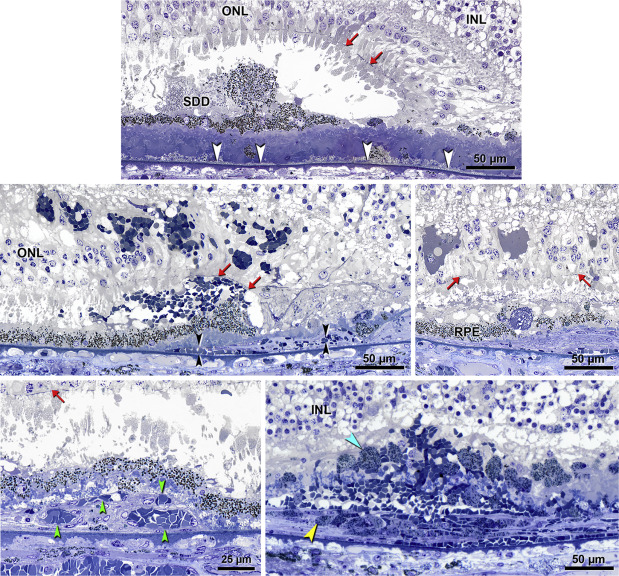
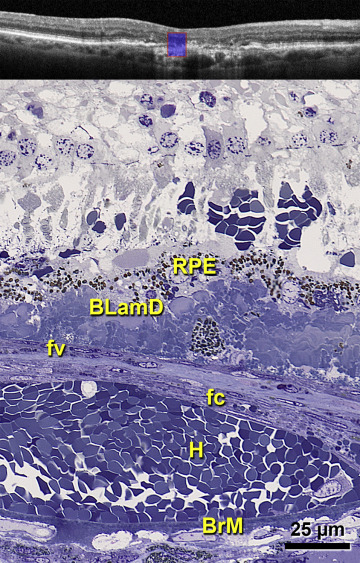
Figure 2 (Top row) demonstrates central atrophy and a faint hyperfluorescent plaque by ICGA. A magnified spectral-domain OCT scan ( Figure 2 , Second row) shows hyperreflective RPE, the hyporeflective band, and the tissue complex including the Bruch membrane. Corresponding histology in Figure 2 (Third row and Fourth row) reveals multiple layers in the tissue complex consistent with type 1 NV. From inner to outer ( Figure 2 , Fourth row, left), dissociated RPE cells sit atop thick basal laminar deposits, which are internal to a fibrovascular scar. The scar ranged from 3 to 73 μm thick across the atrophic area ( Appendix , available at AJO.com ). Further external to the scar is hemorrhage that bows the Bruch membrane outward. Extravascular erythrocytes with minimal associated plasma are also visible within the scar ( Figure 2 , Fourth row, left). The Bruch membrane was calcified in patches, and choriocapillaris was atrophied, especially under the fibrovascular scar.
Corresponding to the hyporeflective band on spectral-domain OCT ( Figure 2 , Second row) was an elaborate basal laminar deposit ( Figure 2 , Third row and Fourth row). In the atrophic area basal laminar deposits sharply elevated the RPE (median thickness, 50 μm; Appendix ), reaching a remarkable thickness of 90–120 μm in the fovea. This eye demonstrated early, late, and rivulet forms of basal laminar deposits ( Figure 2 , Fourth row, right). On its outer aspect, basal laminar deposits contained aggregations of granules shed from RPE and basal mounds (focal areas of soft druse material) ( Figure 2 , Fourth row, right).
Figure 3 demonstrates details of extracellular lesions, type 1 NV with associated exudation, and neurodegeneration that collectively establish this case as AMD. Figure 3 (Top panel) shows basal linear deposit and subretinal drusenoid deposit. In contrast to the extensive basal laminar deposits and fibrovascular scar ( Appendix ), active NV was confined to the superior-nasal quadrant. Figure 3 (Third row, left) shows sub-RPE NV with endothelium, pericytes, intravascular blood cells with plasma, minimal extracellular matrix, and overlying intraretinal exudation. Hemorrhage was found in subscar, intrascar, subretinal, and intraretinal locations throughout the atrophic area ( Figure 3 , Second row, left). In the center of the atrophic area and within the scar were ovoid RPE mixed with erythrocytes ( Figure 3 , Third row, right). Of note was a marked depopulation of the outer nuclear layer and corresponding swelling of Müller cell processes ( Figure 3 , Second row, left). Within the outer nuclear layer were pockets of fluid that contained lipid ( Figure 3 , Second row, right).
Results
Clinical Course and Multimodal Imaging Findings
Presenting visual acuity in the right eye was 20/50. Baseline retinal examination revealed an acquired vitelliform lesion in the central macula, soft drusen, and focal areas of pigmentary changes ( Figure 1 , Top left), as well as reticular pseudodrusen in the superior macula ( Supplementary Figure , Left). There was no exudation, subretinal fluid, or hemorrhage at baseline examination.