Purpose
To compare the clinical features, risk factors, and outcome of polymicrobial keratitis with monomicrobial keratitis due to fungus.
Design
Retrospective, comparative interventional case series.
Methods
Consecutive cases of microbial keratitis with significant growth of more than 1 organism in culture and culture-proven fungal keratitis treated with natamycin alone were retrieved from the microbiology department. Complete success was defined as resolution of the infiltrate with scar formation on medical treatment, partial success as resolution following tissue adhesive application, and failure as inadequate response to medical therapy with increasing infiltrate size, corneal melting, and/or perforation necessitating therapeutic penetrating keratoplasty (PKP) or evisceration.
Results
There were 34 eyes of 34 patients with polymicrobial keratitis and 60 cases of fungal keratitis. Compared to patients with fungal keratitis, patients with polymicrobial keratitis were significantly older (50.03 ± 9.81 years vs 42.79 ± 12.15 years, P = .0038), with larger infiltrates at presentation (61.8% vs 24.1%, P = .0007), a higher association with endophthalmitis (11.8% vs 0%, P = .03), previous history of corneal graft (20.6% vs 0%, P = .0012), and prior topical corticosteroid use (23.5% vs 5%, P = .019). In the polymicrobial group, a combination of bacteria and fungus was more frequently isolated (23, 67.6%), among which filamentous fungi (25, 39.1%) and coagulase-negative staphylococci (14, 21.9%) comprised a majority. Complete success was significantly lower in the polymicrobial group compared to the fungal keratitis group (39.3% vs 73.7%, P = .0045). In multivariate logistic regression analysis comparing factors affecting the outcome between the 2 groups, older age ( P = .027) and ulcers larger than 6 mm ( P = .001) at presentation adversely affected outcome.
Conclusions
Polymicrobial keratitis with fungus and bacteria was more common and more challenging to treat, with a poorer outcome than fungal keratitis. Medical treatment may be effective; however, therapeutic PKP provided globe salvage at best. Early PKP may be advocated for larger ulcers at presentation.
Microbial keratitis (MK) is a significant cause of unilateral corneal blindness. In tropical regions, fungal keratitis comprises almost 40% of all microbial keratitis, while in temperate climates bacterial infections are more prevalent. For nonsevere ulcers, empirical treatment may be started. The standard of care involves a microbiological evaluation to determine the possible organism causing the infection, followed by commencement of appropriate therapy, which may be modified based on culture and sensitivity reports. Unless there are associated systemic risk factors like uncontrolled diabetes or immune compromise, or ocular risk factors like ocular surface disease, previous use of corticosteroids, late presentation, inadequate treatment prior to presentation, noncompliance, or antibiotic resistance, the ulcer responds to treatment with formation of a corneal scar. Mixed infections with more than 1 type of bacteria or a combination of bacteria and fungus or acanthamoeba are not uncommon, ranging from 1.9% to 15.8% of cases. However, there are few reports in the literature describing the outcome of these infections and possible risk factors in their etiology. Owing to multiple organisms infecting the cornea, the classical clinical picture of bacterial or fungal keratitis may be distorted, diagnosis may be delayed, progress may be rapid, and response to treatment may be slow. The aim of this study was to describe the clinical features, risk factors, and outcome of polymicrobial keratitis, and to compare these with monomicrobial keratitis due to fungus.
Methods
The study was a retrospective chart analysis carried out at L. V. Prasad Eye Institute, Visakhapatnam, India between June 1, 2009 and June 30, 2013. The tenets of Declaration of Helsinki were adhered to and institutional review board approval for performing a retrospective chart review was obtained from the Ethics Committee of L. V. Prasad Eye Institute, Visakhapatnam.
We defined polymicrobial keratitis as corneal infection caused by significant growth of more than 1 organism in culture of corneal scraping, or fungus identified in smears of corneal scraping and significant growth of another organism in culture, or isolation of more than 1 organism from culture of the half corneal button obtained from therapeutic penetrating keratoplasty (PKP).
We identified cases with polymicrobial keratitis from the microbiology department, based on the above criteria, and reviewed the medical records. Records of culture-proven cases of fungal keratitis treated with natamycin alone were identified and reviewed as well. Known cases of herpes simplex keratitis and peripheral ulcerative keratitis were excluded.
Clinical Assessment
At presentation, all patients with MK underwent a detailed history evaluation for possible risk factors and comprehensive ocular examination consisting of slit-lamp biomicroscopy (BM900; Haag Streit, Koeniz, Switzerland), fundus evaluation by indirect ophthalmoscopy (Heine Omega 500; Heine Optotechnik, Kientalstr, Herrsching, Germany), and, if necessary, ultrasound biomicroscopy (OTI Ultrasound; Optos, Haileah, Florida, USA). On slit-lamp examination, the following ulcer characteristics were noted: epithelial defect and infiltrate size along the longest dimension and, perpendicular to this, the widest dimension; depth (anterior, mid, posterior stromal); location (central, peripheral, total); associated thinning (percentage); presence of perforation; limbal or scleral involvement; presence of hypopyon.
Microbiological Processing
All patients underwent a microbiological evaluation at presentation according to the Institute protocol, which has been described earlier. Briefly, under topical anesthesia with 0.5% proparacaine hydrochloride eyedrops (Sunways Pvt Ltd, Mumbai, India), corneal scrapings were obtained using a #15 blade on a Bard Parker handle. Smears were taken on glass slides for 10% potassium hydroxide (KOH) with 0.1% calcofluor-white wet mount and observed with a fluorescence microscope. Gram stain was also done. The corneal scrapings were inoculated on 5% sheep blood agar, chocolate agar, Sabouraud dextrose agar (SDA), potato dextrose agar (PDA), brain heart infusion broth, thioglycollate broth, and non-nutrient agar with Escherichia coli overlay. All media were incubated at 37 C except SDA and PDA, which were incubated at 27 C. Chocolate agar was incubated in 5% CO 2 at 37 C.
Significant growth of bacteria and fungus in culture was defined as (1) growth in ≥2 media; (2) confluent growth, in any solid media, on the site of inoculation or from the half corneal button obtained at the time of therapeutic penetrating keratoplasty; or (3) bacterial or fungal elements seen in smear coupled with growth in a single medium. The cultures were observed daily for any growth until 2 weeks. All bacterial and fungal isolates were identified using standard microbiological methods. When the laboratory service began in 2009, some of the bacterial isolates could not be identified beyond morphology owing to nonavailability of biochemical tests. Antibiotic susceptibility testing was done using the Kirby Bauer disc diffusion method.
Treatment Protocol
The Institute protocol for initial management of MK is as follows:
- •
Gram-positive bacteria or no organisms in smear: fortified cefazolin (50 mg/mL) eye drops and ciprofloxacin hydrochloride (0.3%) eye drops every hour for the first 48 hours. Based on the culture report, antibiotic sensitivity, and/or response to treatment, the antibiotic was then modified.
- •
Gram-negative bacteria in smear: ciprofloxacin hydrochloride (0.3%) eye drops every hour for the first 48 hours. This was then modified based on the culture report, antibiotic sensitivity, and/or response to treatment.
- •
Fungal filaments in smear: natamycin (5%) eye drops every half hour and oral ketoconazole (200 mg twice daily) or voriconazole (200 mg twice daily).
Adjunctive medications included topical cycloplegics and oral acetazolamide. Baseline liver function tests were obtained prior to commencement of oral antifungal agents.
All patients were observed for response to treatment. For progressive thinning and melting, impending perforation, or perforation (<2 mm), cyanoacrylate tissue adhesive was applied, followed by insertion of a bandage contact lens. Therapeutic PKP was done for perforation >2 mm, located either centrally or paracentrally; infiltrate at or close to limbus with risk of scleral involvement; progressive increase in size despite appropriate therapy; and recurrent graft infiltrate.
Outcome Measures
Complete success was defined as resolution of the infiltrate with scar formation on medical treatment. Partial success was defined as resolution following tissue adhesive application. Failure was defined as inadequate response to medical therapy with progressive increase in infiltrate size, corneal melting, and/or perforation necessitating 1 or more therapeutic penetrating keratoplasties or evisceration. Primary graft failure was defined as persistence of corneal edema from the day PKP was performed. Graft failure was defined as an opaque graft due to edema secondary to primary graft failure or allograft rejection, or scarring due to graft infiltrate.
Data Collection and Analysis
The following details- were noted from the medical record: age, sex, eye affected, occupation (indoor/outdoor), predisposing risk factors, duration of symptoms, ulcer characteristics, microbiology culture results, antibiotic susceptibility data, treatment received, surgical interventions performed, outcome, duration of follow-up, and presenting and final visual acuity.
Statistical analysis included t tests and χ 2 tests to compare means and proportions, respectively, between the 2 groups. Factors affecting the outcome were determined using Fisher exact test. A P value of <.05 was considered as statistically significant. To determine factors predicting success or failure between the 2 groups, univariate and multivariate logistic regression analysis was done.
Results
Out of a total of 975 culture-positive cases of bacterial and fungal keratitis seen between June 2009 and June 2013, 34 eyes of 34 patients (3.5%) with polymicrobial keratitis were identified, of which 27 (79.4%) were male. More than two-thirds of the patients were outdoor workers. The right eye was affected in 21 of 34 cases (61.8%). The mean duration of symptoms was 14.3 ± 8.6 days (range 2–30 days, median 10 days). More than three-fourths of the cases (26/34, 76.5%) presented beyond a week of onset of symptoms. We identified 60 cases of fungal keratitis between May 1, 2012 and December 31, 2013. Patients with polymicrobial keratitis were a decade older (50.03 ± 9.81 years vs 42.79 ± 12.15 years, P = .0038) than those with fungal keratitis. The demographic data are shown in Table 1 .
| Polymicrobial Keratitis (n = 34) | Fungal Keratitis (n = 60) | P Value | 95% CI | |
|---|---|---|---|---|
| Demographic Data | ||||
| Age (y) | 50.03 ± 9.81 | 42.79 ± 12.15 | .0038 a | −12.09 to −2.39 |
| Sex | ||||
| Male | 27 (79.4) | 38 (63.3) | .16 | −5.0 to 34.04 |
| Female | 7 (20.6) | 22 (36.7) | ||
| Occupation | ||||
| Outdoors | 21 (61.8) | 31 (51.7) | .46 | −12.33 to 30.94 |
| Indoors | 13 (38.2) | 29 (48.3) | ||
| Eye affected | ||||
| OD | 21 (61.8) | 39 (65.0) | .93 | −17.72 to 24.93 |
| OS | 13 (38.2) | 21 (35.0) | ||
| Duration of Symptoms (d) | 14.3 ± 8.6 | 15.4 ± 9.5 | .57 | −2.82 to 5.01 |
| Ulcer Characteristics | ||||
| Location | ||||
| Central | 26 (76.5) | 37 (61.7) | .21 | −6.68 to 33.35 |
| Peripheral | 8 (23.5) | 13 (21.6) | .96 | −15.98 to 21.97 |
| Paracentral | – | 3 (5) | ||
| Total/near total | – | 7 (11.7) | ||
| Scleral involvement | 2 (5.9) | 1 (1.67) | .6 | −4.7 to 18.12 |
| Endophthalmitis | 4 (11.8) | 0 (0) | .03 a | 1.43–27.5 |
| Size >6 mm (1 dimension) | 21 (61.8) | 13 (24.1) | .0007 a | 15.46–56.67 |
| Thinning >70% | 9 (26.5) | – | ||
| Perforation at presentation | 6 (17.6) | 3 (5) | .10 | −1.46 to 29.93 |
| Hypopyon | 13 (38.2) | 26 (43.34) | .78 | −17.08 to 26.07 |
Ulcer Characteristics
A majority of polymicrobial ulcers were located centrally (26, 76.5%) and 21 (61.8%) measured >6 mm along 1 dimension. The mean area of the infiltrate was 35.9 ± 17.8 mm 2 (range: 6–64 mm 2 , median: 35.8 mm 2 ). Six eyes (17.6%) had a perforation at presentation ( Table 1 ). Patients with polymicrobial keratitis had larger infiltrates at presentation (61.8% vs 24.1%, P = .0007) and a higher association of endophthalmitis (11.8% vs 0%, P = .03) compared to cases with fungal keratitis.
Microbiological Data
Sixty-seven organisms were isolated in culture from corneal scrapings and/or half corneal buttons of the 34 cases of polymicrobial keratitis. Of the 34 cases, in 27 cases (79.4%), the organisms were identified in the smears and/or culture and in 17 (50%), 1 organism was identified in smears and both organisms grew in culture. In 5 cases (14.7%) no organisms were identified in the smears but in 1 case both organisms grew in culture and in 4 (11.8%), these grew from cultures of the corneal buttons. Figure 1 depicts the details of the methods by which organisms were identified. With culture as the gold standard, for identifying at least 1 bacterium, Gram stain had a sensitivity of 70.27% (95% confidence interval [CI] 53.02%–84.11%). Specificity could not be determined, since all cases had bacterial growth in culture and there were no true negatives or false positives on Gram stain. For fungus, Gram stain had a sensitivity of 95.45% (95% CI 77.08%–99.24%) and specificity of 100% (95% CI 62.91%–100%) and 10% KOH with 0.1% calcofluor-white had a sensitivity of 77.27% (95% CI 54.62%–92.09%) and specificity of 100% (95% CI 62.91%–100%). The combined sensitivity and specificity for fungus with Gram and KOH with calcofluor-white was 95.45% and 100%, respectively.
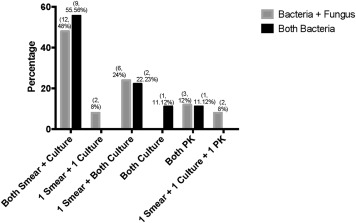
A combination of bacteria and fungus was noted in 25 cases (73.5%), while 2 different bacteria were identified in each of the remaining 9 (26.5%). The most common isolate was fungus (25, 39.1%), followed by coagulase-negative Staphylococcus (CONS) (14, 21.9%), Streptococcus species (7, 10.9%), and Pseudomonas aeruginosa (6, 9.4%). The details of all isolates are given in Table 2 .
| Classification | Organism | N (%) |
|---|---|---|
| Gram-positive cocci | Coagulase-negative Staphylococcus | 14 (17) |
| Alpha-hemolytic Streptococcus | 3 (7.1) | |
| Streptococcus pneumoniae | 3 (7.1) | |
| Gram-positive cocci | 2 (4.8) | |
| Streptococcus sp | 1 (2.4) | |
| Micrococcus sp | 1 (2.4) | |
| Gram-negative cocci | Neisseria sp | 3 (7.1) |
| Gram-negative cocci | 2 (4.8) | |
| Gram-positive rods | Gram-positive bacilli | 3 (7.1) |
| Corynebacterium sp | 1 (2.4) | |
| Gram-negative rods | Pseudomonas aeruginosa | 6 (14.3) |
| Enterobacter sp | 1 (2.4) | |
| Escherichia coli | 1 (2.4) | |
| Gram-negative bacilli | 1 (2.4) | |
| Total bacterial isolates | 42 (100) | |
| Filamentous fungi | Aspergillus flavus | 9 (36) |
| Fusarium solani | 4 (16) | |
| Fusarium oxysporum | 1 (4) | |
| Fusarium sp | 3 (12) | |
| Unidentified hyaline fungus | 7 (28) | |
| Unidentified dematiaceous fungus | 1 (4) | |
| Total fungal isolates | 25 (100) | |
| Total Isolates | 67 |
The growth of the first organism in culture took 1.71 ± 0.88 days (range 1–4 days, median 1.5 days). The second organism grew in 2.24 ± 1.38 days (range 1–6 days, median 2 days). Table 3 shows the antibiotic sensitivity data of the 5 most common bacterial isolates: coagulase-negative Staphylococcus , Streptococcus pneumoniae , alpha-hemolytic Streptococcus , Pseudomonas aeruginosa , and Neisseria sp.
| Coagulase-Negative Staphylococcus (n = 12) | Streptococcus Pneumoniae (n = 3) | Alpha-hemolytic Streptococus (n = 3) | Pseudomonas Aeruginosa (n = 6) | Neisseria Sp (n = 3) | |
|---|---|---|---|---|---|
| Cefazolin | 8 (72.73) | 3 (100) | 2 (66.67) | 1 (100) | |
| Chloramphenicol | 9 (81.82) | 2 (66.67) | 3 (100) | 3 (60) | 3 (100) |
| Ciprofloxacin | 6 (54.55) | 3 (100) | 1 (33.34) | 4 (80) | 1 (33.34) |
| Vancomycin | 6 (54.55) | 3 (100) | 1 (50) | ||
| Gatifloxacin | 7 (63.64) | 3 (100) | 2 (66.67) | 4 (80) | 0 |
| Moxifloxacin | 5 (45.46) | 3 (100) | 2 (66.67) | 2 (50) | 1 (33.34) |
| Ofloxacin | 6 (60) | 3 (100) | 2 (66.67) | 4 (80) | 2 (66.67) |
| Gentamycin | 9 (81.82) | 2 (66.67) | 1 (33.34) | 4 (80) | 1 (33.34) |
| Imipenem | 7 (87.5) | 2 (66.67) | 3 (100) | 4 (80) | 1 (50) |
| Piperacillin Tazobactum | 6 (75) | 2 (66.67) | 3 (100) | 3 (60) | 1 (50) |
| Ceftazidime | 2 (40) | ||||
| Tobramycin | 2 (50) | 1 (33.34) |
For cases with fungal keratitis, the organism was identified in Gram stain in 47 cases (79.67%) (sensitivity 79.66%, 95% CI 67.17%–89.02%) and in 10% KOH with calcofluor-white in 54 cases (91.53%) (sensitivity 91.53%, 95% CI 81.32%–97.19%). The most common isolates were filamentous fungi belonging to Aspergillus sp (38.35%) and Fusarium sp (35.01%).
Risk Factors
Ocular and systemic risk factors predisposing patients to polymicrobial and fungal keratitis are outlined in Table 4 . Patients with polymicrobial keratitis were more likely to have had a previous corneal graft (20.6% vs 0%, P = .0012) with prior topical corticosteroid use (23.5% vs 5%, P = .019).
| Ocular Risk Factors | Polymicrobial Keratitis (n = 34) | Fungal Keratitis (n =60) | P Value | 95% Confidence Intervals |
|---|---|---|---|---|
| Trauma | 11 (32.4) | 28 (46.7) | .26 | −8.05 to 34.35 |
| Previous corneal graft | 7 (20.6) | 0 | .0012 | 7.3–37.9 |
| Contact lens wear | 0 | 1 (1.67) | .77 | −8.74 to 8.95 |
| Therapeutic contact lens wear | 3 (8.8) | 0 | .08 | −0.36 to 23.65 |
| Ocular surface disorder | 4 (11.7) | 1 (1.67) | .1 | −1.11 to 25.78 |
| Exposure keratopathy | 2 (5.9) | 1 (1.67) | .61 | −4.69 to 18.12 |
| Topical corticosteroids | 8 (23.5) | 3 (5) | .019 | 2.9–36.57 |
| Nasolacrimal duct obstruction | 1 (2.9) | 0 | .78 | −3.7 to 15.26 |
| Systemic risk factors | ||||
| DM | 3 (8.8) | 3 (5) | .77 | −7.5 to 19.1 |
| HIV | 1 (2.9) | 1 (1.67) | .72 | −6.57 to 13.7 |
Outcome
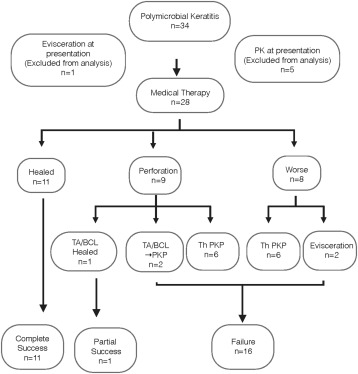
The mean duration of follow-up was 9.9 ± 11.2 months (range 0.75–49.5 months, median 5.75 months). Of the 34 cases with polymicrobial keratitis, 5 (14.7%) underwent therapeutic PKP at presentation and 1 (2.9%) had to be eviscerated; hence, these were excluded from the outcome analysis. Thus, of the remaining 28 cases, complete success was noted in 11 (39.3%), partial success in 1 (3.6%), and failure in 16 (57.1%). The mean duration of medical therapy in patients whose infiltrate resolved was 70.35 ± 35.84 days (range 21–150 days, median 62 days). During the course of therapy, 9 eyes (32.1%) developed a perforation. Three of these underwent cyanoacrylate tissue adhesive application and 1 healed with medical therapy following this intervention; however, the other 2 eyes subsequently underwent therapeutic PKP. The remaining 6 received a therapeutic PKP, as the perforation was too large to be sealed with cyanoacrylate tissue adhesive. Eight eyes (28.6%) worsened on medical therapy, with an increase in the size of the infiltrate and limbal involvement. Two of these had to be eviscerated, while the remaining 6 underwent a therapeutic PKP. The flow chart ( Figure 2 ) shows the outcome of all cases of polymicrobial keratitis. All the therapeutic grafts failed owing to primary graft failure or following secondary infection and subsequent corneal opacification. Table 5 shows the organisms isolated in culture for each patient along with the risk factors, surgical intervention, and outcome.
| Patient | Age/Sex | Organism 1 | Organism 2 | Ocular Risk Factor | Surgical Interventions | Outcome |
|---|---|---|---|---|---|---|
| 1 | 41/M | Alpha-hemolytic Streptococcus | Fusarium solani | PKP, steroid | Therapeutic PKP | Failed graft |
| 2 | 47/F | CONS | GNC | PKP, BCL, steroid | Therapeutic PKP | Failed graft |
| 3 | 32/M | GPB | Fusarium solani | Trauma | None | Corneal scar |
| 4 | 42/M | CONS | Unidentified hyaline fungus | None | None | Corneal scar |
| 5 | 35/M | Unidentified hyaline fungus | GPC | Lagophthalmos | None | Corneal scar |
| 6 | 60/F | Corynebacterium sp | Fusarium solani | None | Evisceration | – |
| 7 | 32/M | Alpha-hemolytic Streptococcus | Unidentified hyaline fungus | Trauma | None | Corneal scar |
| 8 | 65/M | CONS | Aspergillus flavus | Trauma | Therapeutic PKP | Failed graft |
| 9 | 27/M | Unidentified hyaline fungus | CONS | Trauma | None | Corneal scar |
| 10 | 24/M | Pseudomonas aeruginosa | Aspergillus flavus | None | None | Corneal scar |
| 11 | 45/M | CONS | Fusarium sp | Trauma | None | Corneal scar |
| 12 | 55/F | Alpha-hemolytic Streptococcus | Fusarium solani | NLDO | None | Corneal scar |
| 13 | 54/M | CONS | Pseudomonas aeruginosa | PED, BCL | Therapeutic PKP (at presentation) | Failed graft |
| 14 | 50/M | Aspergillus flavus | CONS | Trauma | Evisceration | – |
| 15 | 65/M | Neisseria sp | Aspergillus flavus | None | Therapeutic PKP | Failed graft |
| 16 | 66/M | CONS | Escherichia coli | PKP, steroid | Therapeutic PKP | Failed graft |
| 17 | 62/M | GPB | CONS | PKP, steroid | Therapeutic PKP | Failed graft |
| 18 | 34/M | Pseudomonas aeruginosa | Fusarium oxysporum | PKP, steroid | TA BCL; Therapeutic PKP | Failed graft |
| 19 | 65/M | Pseudomonas aeruginosa | Streptococcus pneumoniae | PED, BCL | Therapeutic PKP | Failed graft |
| 20 | 50/F | Neisseria sp | CONS | PKP, steroid | None | Corneal scar |
| 21 | 48/M | Unidentified dematiaceous fungus | GPC | None | Therapeutic PKP | Failed graft |
| 22 | 65/M | Neisseria sp | Streptococcus sp | PBK, steroid | None | Corneal scar |
| 23 | 56/M | GPB | Fusarium sp | Trauma | None | Corneal scar |
| 24 | 30/M | Fusarium sp | Micrococcus sp | Trauma | Therapeutic PKP (at presentation) | Failed graft |
| 25 | 55/M | CONS | Aspergillus flavus | TED | TA BCL | Adherent leucoma |
| 26 | 50/M | Streptococcus pneumoniae | Unidentified hyaline fungus | None | Therapeutic PKP | Failed graft |
| 27 | 60/F | Pseudomonas aeruginosa | Aspergillus flavus | None | Therapeutic PKP | Failed graft |
| 28 | 50/M | Enterobacter sp | Streptococcus pneumoniae | PKP, Steroid | TA BCL; therapeutic PKP | Failed graft |
| 29 | 65/M | Beta-hemolytic Streptococcus | Aspergillus flavus | None | Therapeutic PKP (at presentation) | Failed graft |
| 30 | 45/M | Unidentified hyaline fungus | Pseudomonas aeruginosa | Trauma | Therapeutic PKP | Failed graft |
| 31 | 65/M | GNB | Aspergillus flavus | Trauma | Evisceration | – |
| 32 | 43/M | Unidentified hyaline fungus | CONS | Trauma | Therapeutic PKP | Failed graft |
| 33 | 54/F | CONS | GNC | PKP, PED | Therapeutic PKP; evisceration | – |
| 34 | 64/F | CONS | Aspergillus flavus | None | Therapeutic PKP; evisceration | – |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


