Purpose
To report the clinical features and management of mitogen-activated protein kinase kinase inhibitor–associated ocular side effects in 4 patients with advanced melanoma and a review of literature.
Design
Interventional case series.
Methods
Four patients with advanced cutaneous melanoma were treated with a mitogen-activated protein kinase kinase (MEK) inhibitor as single therapy or together with a v-raf murine sarcoma viral oncogene homolog B1 (BRAF) inhibitor. All patients underwent ophthalmologic examinations at regular intervals or as needed, including visual acuity, intraocular pressure, external eye examination, and funduscopy. When pathologic findings were found, patients underwent visual field examination, optical coherence tomography (OCT), and/or fluorescein angiography. Ocular toxicity was assessed and handled according to the Common Terminology Criteria for Adverse Events.
Results
Ocular adverse events appeared early in the treatment. In 3 patients OCT revealed subfoveal neuroretinal elevation, often asymptomatic, also after discontinuation and re-starting of MEK inhibitor. Vascular injury appeared in 2 patients, in 1 case associated with a visual field defect reduced after discontinuation of the drug and use of systemic therapy. In 1 case an inflammatory reaction was observed in the anterior chamber. Visual symptoms were usually mild and short-lived.
Conclusions
MEK inhibitor as a single agent or in combination with BRAF inhibitor induces transient retinopathy with time-dependent recurrence and usually mild visual symptoms. Vascular injuries can be observed and their management is essential in clinical practice. It is important to investigate all previous ocular disorders, systemic conditions, and pharmacologic interactions of MEK inhibitor that could facilitate the onset of associated ocular effects.
Malignant melanoma is the seventh most common cancer worldwide and the main cause of skin cancer deaths. Its incidence has been rising faster than any other cancer type for at least 30 years, but fortunately, in very recent years, we have also seen the dawn of a new era in the treatment options available for patients with metastatic disease.
Collectively these new treatments have tripled the median overall survival, from 6–8 months with the old standard of care to about 20 months using new drugs, which include anti–cytotoxic T-lymphocyte antigen 4 (CTLA4)/anti–programmed cell death protein 1 (PD1) immunotherapy and targeted therapies. The development of these novel agents was driven by the rapidly evolving advancements in the understanding of environmental and genetic influences on melanoma. In particular, a considerable subset of about two-thirds of metastatic melanoma patients is defined by the presence of gene target mutations that involve v-raf murine sarcoma viral oncogene homolog B1 (BRAF) or neuroblastoma RAS viral oncogene homolog (NRAS), both along the Ras/Raf/mitogen-activated protein kinase (MAPK)/extracellular signal regulated kinase (ERK) pathway. For metastatic melanoma patients who carry BRAF V600 mutations, the combination with BRAF and mitogen-activated protein kinase kinase (MEK) inhibitors is becoming the new standard of cure, while patients with NRAS Q61 mutations are encouraged to participate in clinical trials with MEK inhibitors ( NCT01693068 , NCT01763164 ). Moreover, MEK inhibitors are being tested in metastatic uveal melanoma with guanine nucleotide binding protein (Gnaq) and subunib alpha of Gnaq (Gna11) mutations, as well as in a plethora of cancer types characterized by aberrant activation of the Ras/Raf/MEK/ERK pathway, which is observed in one-third of all tumors.
MEK inhibitors have a peculiar toxicity profile with a unique ocular toxicity. As these agents move from the trial context to a “real-world” clinical practice, education and awareness of these adverse events is essential in order to optimize patient outcomes. However, there is still a lack of published data regarding ocular side effects.
In this report we describe 4 metastatic melanoma patients who experienced a spectrum of MEK inhibitor–associated ocular adverse events. Details of their diagnostic and therapeutic management are reported, as well as data on their pathogenesis and clinical course.
Methods
Four patients with advanced cutaneous melanoma were treated with a selective MEK inhibitor as single therapy or together with BRAF inhibitor in 2 different clinical trials ( NCT01689519 , NCT01693068 ). Three patients (Cases 2, 3, 4) on combination therapy (vemurafenib plus GDC-0973) and 1 (Case 1) on MEK inhibitor single therapy (pimasertib) underwent an ophthalmologic examination at baseline, before each cycle, and as needed.
The ophthalmologic assessment included visual acuity (Snellen scale), intraocular pressure, external eye examination, and funduscopy. When some pathologic findings at baseline or at follow-up were found, patients also underwent visual field examination (Humphrey HFA; Carl Zeiss Meditec, Dublin, California, USA), optical coherence tomography (OCT) scans (Cirrus HD-OCT; Carl Zeiss Meditec, or OCT RTVue; OPTOVUE Inc, Fremont, California, USA). and/or fluorescein angiography (Spectralis HRA-OCT; Heidelberg Engineering, Heidelberg, Germany).
Toxicity was assessed according to the Common Terminology Criteria for Adverse Events (CTCAE version 4.0) grading for eye disorders ( Table ). The management of ocular side effects was carried out in partnership by oncologists and ophthalmologists.
| Grade 1 | Asymptomatic or mild symptoms; clinical or diagnostic observations only; intervention not indicated |
| Grade 2 | Moderate or minimal, local or noninvasive intervention indicated; limiting age, appropriate instrumental Activity Daily Living |
| Grade 3 | Severe or medically significant, but not immediately sight threatening; hospitalization or prolongation of existing hospitalization indicated; disabling; limiting self-care Activity Daily Living |
| Grade 4 | Sight-threatening consequences; urgent intervention indicated; blindness (20/200 or worse in the affected eye) |
The research followed the tenets of the Declaration of Helsinki and all patients were informed about possible and variable ocular toxic effects. The studies were conducted in accordance with the international standards of good clinical practice. The protocols were approved by the institutional review board of the National Cancer Research Centre “Giovanni Paolo II,” Bari, Italy, and complied with country-specific regulatory requirements. Written informed consent was obtained from the patients for publication of their cases and any accompanying images.
Case Reports
Case 1
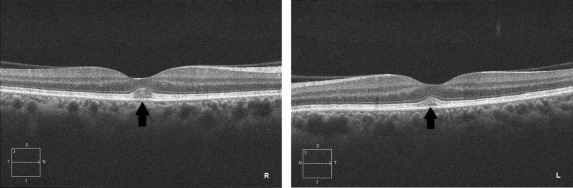
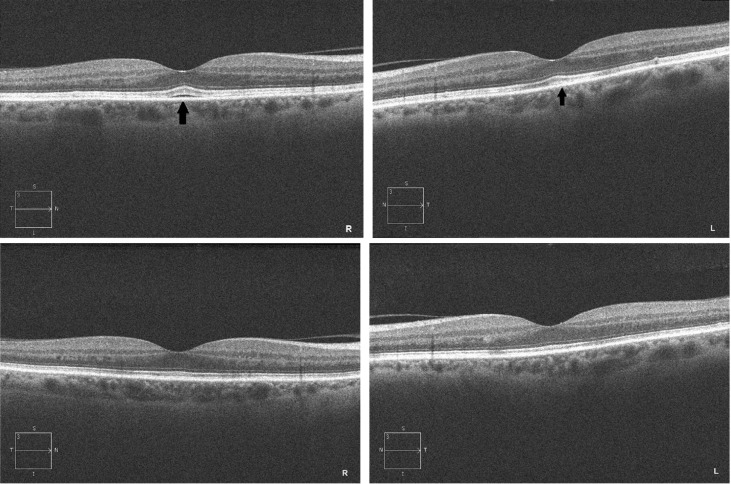
A 62-year-old woman underwent radical surgery for cutaneous malignant melanoma of her left shoulder. Unfortunately, after 1 year a computed tomography scan highlighted metastatic lesions of soft tissue, lymph nodes, and bones. A genetic analysis of her metastatic melanoma lesion by pyrosequencing showed the presence of an NRAS Q61L mutation and a concomitant BRAF G466E mutation on exon 11. As a comorbidity, the patient had hypertension, treated with ramipril, an ACE inhibitor. She was enrolled in a clinical trial with the MEK inhibitor pimasertib, receiving 60 mg of the drug twice a day. After 1 day of therapy she reported a visual impairment lasting about 12 hours and then regressed spontaneously. She underwent an ophthalmologic evaluation, which showed visual acuity of 20/20 in both eyes, but the Amsler test revealed metamorphopsia. The slit-lamp examination and funduscopy did not reveal any abnormalities, but OCT showed a bilateral mild foveal serous neuroretinal detachment of a height around 30 μm. In accordance with the adverse events management protocol, which provided no dose modification for grade 1 ocular toxicity, the patient continued treatment with a full dose of pimasertib and a new ophthalmologic evaluation was scheduled 2 weeks later. In the meantime, the patient discontinued the therapy because of a grade 3 increase of creatine phosphokinase. The second ophthalmologic assessment performed during this therapeutic interruption showed the complete disappearance of the ocular findings ( Figure 1 ). After resolution of the increase in creatine phosphokinase, the patient re-started MEK inhibitor with a lower dose of 45 mg twice a day. The dosage was further reduced to 30 mg owing to the reappearance of a grade 3 creatine phosphokinase increase. A week after restarting the treatment she underwent a new ophthalmologic assessment as per protocol. Although she was asymptomatic, a new subfoveal neurosensory retinal detachment higher than the previous (right eye 56 μm, left eye 53 μm) was highlighted by an OCT scan ( Figure 2 ). Given the grade 1 ocular toxicity and as in the previous episode, no action was taken for visual adverse events, but a new ophthalmologic assessment was scheduled. However, soon after the patient permanently withdrew from the MEK inhibitor owing to a third grade 3 creatine phosphokinase increase.
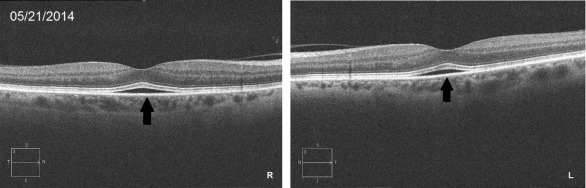
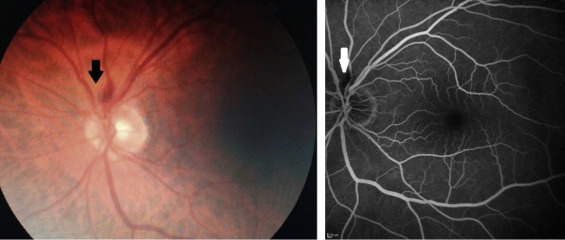
The ophthalmologic assessment, performed 2 weeks after therapy discontinuation, highlighted the resolution of the previous subfoveal retinal detachment, but also the appearance of an isolated asymptomatic parapapillary hemorrhage in the left eye. Fearing retinal vascular abnormalities, we performed fluorescein angiography, which revealed an unchanging hypofluorescence due to a single hemorrhage, but no other vascular injuries ( Figure 3 ). After 2 weeks the hemorrhage disappeared and no further pathologic findings were revealed.
During the treatment with MEK inhibitor the patient gained a partial response, according to Response Evaluation Criteria in Solid Tumors (RECIST), lasting for 3 months after discontinuation. When the disease progressed, the patient was treated with ipilimumab and achieved a complete remission of disease, which still remains. No other ocular adverse events were documented during the subsequent treatment.
Case 2
A 46-year-old woman affected by a vulvar melanoma showed spreading of the disease to soft tissue, lymph nodes, liver, and lung. She had no other comorbidity.
Owing to the presence of BRAFV600E following genetic testing of her melanoma specimen, the patient was enrolled in a clinical trial that planned to treat metastatic melanoma until progression with vemurafenib 960 mg twice a day continuously and cobimetinib 60 mg daily on days 1–21 of each 28-day treatment cycle.
The pretreatment ophthalmologic evaluation did not reveal morphologic or functional abnormalities. After 1 week of treatment she felt a mild visual impairment and distortion. A new ophthalmologic assessment was performed, this time showing a bilateral subfoveal neurosensory retinal detachment measuring about 40 μm at OCT ( Figure 4 ). As per guidelines for a grade 2 visual adverse event, treatment was temporarily interrupted. After 1 week, as the visual symptoms had subsided and OCT showed resolution of the subfoveal retinal detachment, she resumed use of vemurafenib and cobimetinib at the previous dose. Further ophthalmologic evaluation showed recurrence of serous retinal detachment similar to the previous examination in terms of thickness.