CORNEAL INFECTIONS, INFLAMMATIONS, AND SURFACE DISORDERS
BACTERIAL KERATITIS
Bacterial keratitis is a serious, potentially sight-threatening corneal infection which typically develops in patients with a compromised corneal surface.
Predisposing Factors
• Contact lens wear: especially extended-wear soft lenses
• Corneal trauma, foreign bodies
• Ocular surface disease (e.g., exposure/neurotrophic keratopathy, chronic bullous keratopathy, dry eye syndrome, trichiasis, distichiasis, entropion)
• Topical immunosuppressive therapy (e.g., corticosteroids)
• Immunocompromised patient
• Postoperative: corneal wound or suture-related (e.g., corneal graft)
Etiology
• Staphylococcus
• Streptococcus
• Pseudomonas
• Moraxella
• Atypical mycobacteria, others
Symptoms
• Pain, irritation, redness, photophobia, discharge, decreased vision, contact lens intolerance
Signs
• Vary according to the severity of the infection and, to a lesser extent, on the causative organism.
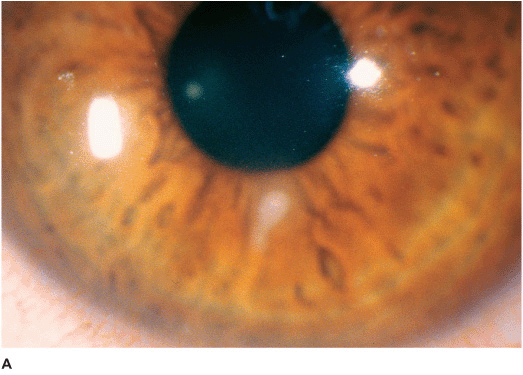
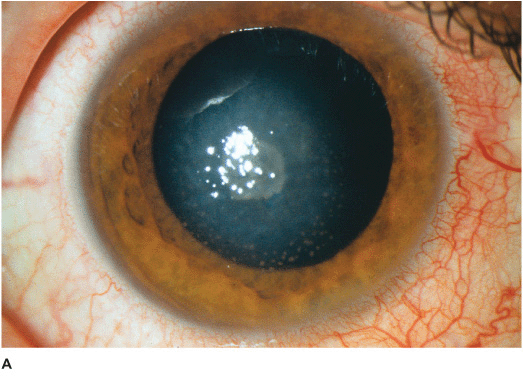
• White stromal infiltrate associated with conjunctival injection and typically with an overlying epithelial defect. There may be stromal loss (ulcer) (Figures 7-1A,B).
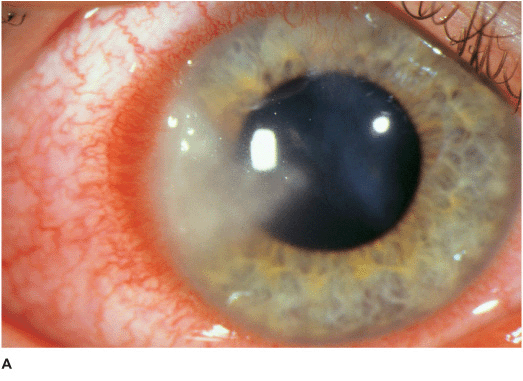
• There may be surrounding stromal edema, Descemet’s folds, secondary reactive iritis, and hypopyon (Figures 7-1C,D).
• Staphylococcal keratitis is characterized by a well-defined, white-gray or creamy stromal infiltrate that may enlarge to form a dense stromal abscess.
• Streptococcal keratitis can be suppurative or have a crystalline appearance. Severe anterior uveitis and hypopyon formation are common.
• Pseudomonal keratitis presents as a rapidly progressive, suppurative infiltrate associated with hypopyon and a mucopurulent discharge. Corneal perforation may occur (Figure 7-1E).
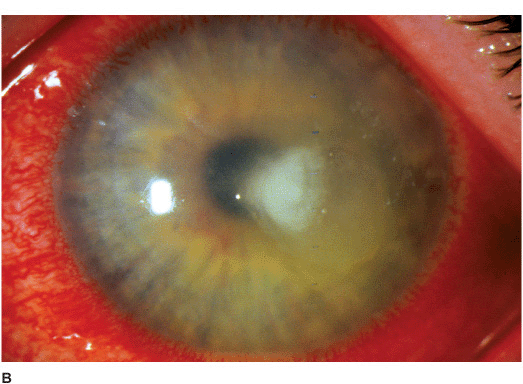
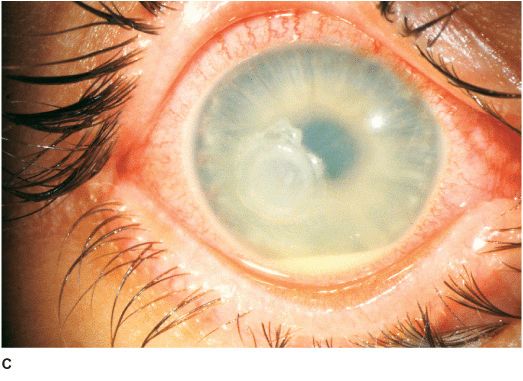
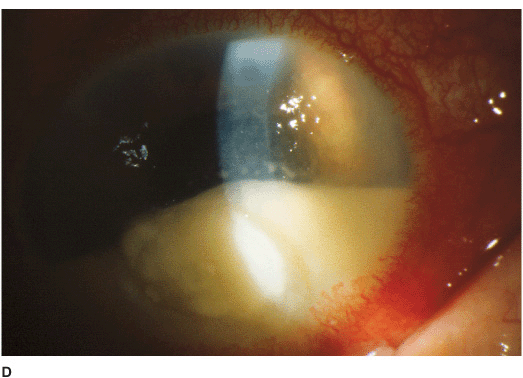
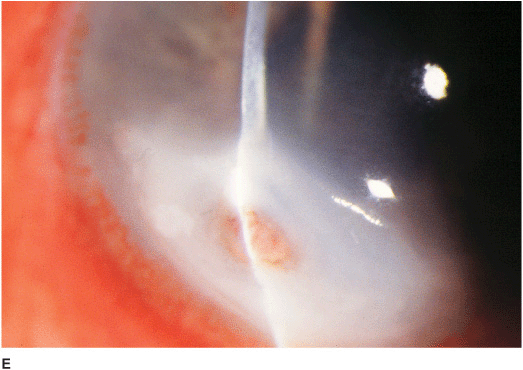
Figure 7-1 Bacterial keratitis A. This small inferior corneal infiltrate in an overnight soft contact lens wearer has some underlying corneal edema. Because it may be an early infectious keratitis, it should be treated with topical antibiotics and followed closely. B. This dense central corneal ulcer has a large overlying epithelial defect and moderate underlying corneal edema. There is a small hypopyon inferiorly. C. This corneal infection was due to Pseudomonas aeruginosa. There is a large circular corneal ulcer with overlying mucopurulent discharge, underlying corneal edema and a moderately large hypopyon. D. This large dense corneal ulcer is associated with a hypopyon that fills approximately 50% of the anterior chamber. E. This infected corneal ulcer caused a perforation. Iris is plugging the wound. The anterior chamber is shallow but formed.
Differential Diagnosis
• Sterile ulcers: vernal shield ulcer, neurotrophic or exposure keratitis, autoimmune keratitis, contact-lens induced sterile keratitis, medicamentosa keratitis. Usually less painful, minimal or no iritis or corneal edema and culture for bacteria is negative.
• Staphylococcal hypersensitivity keratitis: infiltrates may be bilateral; multiple; peripheral; located at 2, 4, 8, or 10 o’clock positions; associated with blepharitis; epithelial defect is absent or is smaller than the infiltrate; and there is minimal anterior chamber activity.
• Other microbial (nonbacterial) keratitis: bacterial cultures are negative. Fungal and special cultures and stains are necessary for diagnosis.
Diagnosis
• Corneal scraping for Gram’s stain, Giemsa stain, calcofluor white stain, cultures, and sensitivity testing. Routine media include blood, chocolate, Sabouraud’s agars, and thioglycolate broths.
• For deep lesions or when repeated cultures are negative in recalcitrant cases, a corneal biopsy may be necessary.
Treatment
• Empirical outpatient treatment with broad-spectrum, topical, nonfortified antibiotic drops may be sufficient for small (2 mm or less) peripheral ulcers with minimal symptoms and minimal anterior chamber activity. Topical fluoroquinolone (e.g., levofloxacin, ciprofloxacin, or ofloxacin) drops q 30 to 60 min around the clock initially after a loading dose of 1 drop q 5 min for 15 min.
• For larger ulcers or when the ulcers involve the visual axis, or are associated with significant discharge, anterior chamber activity, and hypopyon, treatment may require intensive fortified antibiotic drops. Patients are often hospitalized. Fortified cefazolin (50 mg/mL) or vancomycin (25 mg/mL) and fortified gentamicin or tobramycin (15 mg/mL). Frequency of instillation: 1 drop q 5 min for 30 min, then q 30 to 60 min for 24 hours of each drop. Wait 5 minutes between administration of each medication.
• Subconjunctival antibiotics are only necessary if fortified eye drops cannot be started soon.
• Oral antibiotics (e.g., ciprofloxacin 500 mg bid or levofloxacin 500 mg qd) are helpful when the ulcer involves the sclera or has extended into the eye. Systemic antibiotics are also required for Neisseria and Haemophilus infection (e.g., ceftriaxone 1 g IV or IM q 12 to 24 h).
• Cycloplegics are often used to reduce ciliary spasm and to prevent posterior synechiae (e.g., scopolamine 0.25% or atropine 1% tid).
• Modify regimen according to clinical response and culture and sensitivity results.
• Topical corticosteroids can be used for severe inflammation only after the organism is identified and the infection is under control.
• Corneal graft may be necessary in severe cases that are progressing despite aggressive treatment or for ulcers that have perforated.
Prognosis
• Close follow-up is required. Prognosis is very good for small ulcers, good for moderate ulcers, poor for severe ulcers. Better prognosis for ulcers outside the visual axis than ulcers in the visual axis.
FUNGAL KERATITIS
Fungal keratitis is a very serious, potentially sight-threatening corneal infection which most commonly develops in patients after trauma or those with a compromised corneal surface.
Etiology
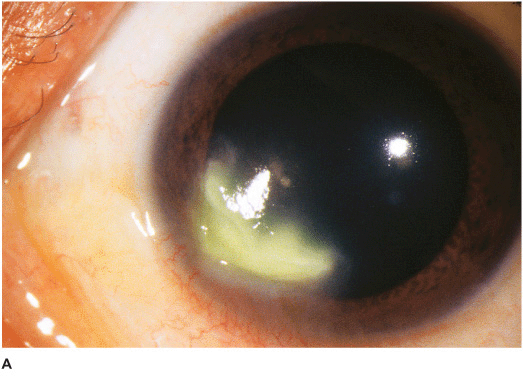
Nonfilamentous (e.g., Candida) Candida keratitis is a rare, unilateral, insidious fungal infection that usually occurs in eyes with pre-existing chronic corneal disease (e.g., dry eyes, herpes keratitis, exposure keratopathy, postkeratoplasty, chronic use of corticosteroid drops) or in severely debilitated patients. Features include a gray-white stromal infiltrate similar to a bacterial ulcer. May have an anterior chamber reaction and hypopyon (Figures 7-2A,B).
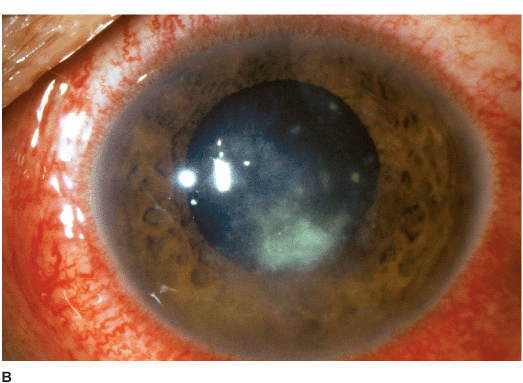
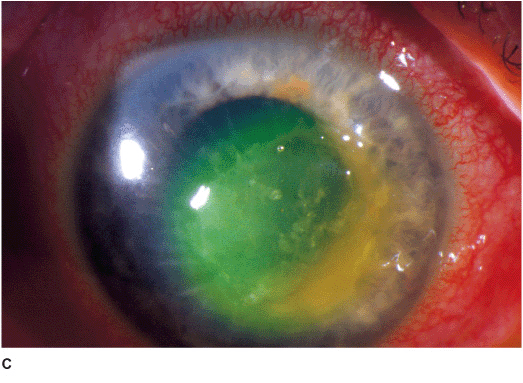
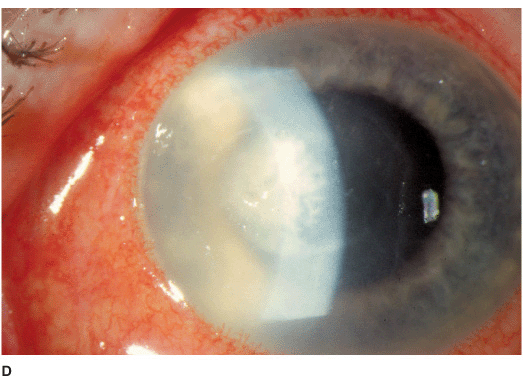
Figure 7-2 Fungal keratitis A. This multilobulated dense infiltrate was caused by a Candida infection. There is an overlying epithelial defect. Peripheral corneal neovascularization suggests it is a long-standing ulcer. B. This Candida corneal ulcer is slowly improving. The denser infiltrate at the inferior pupillary margin is surrounded by multiple satellite lesions. C. This multicentric corneal ulcer was caused by Fusarium. There is a large epithelial defect with significant underlying corneal edema. D. This dense white infiltrate with feathery borders was due to a Fusarium infection. A ring infiltrate is beginning inferiorly.
Filamentous (e.g., Aspergillus, Fusarium, etc.) Filamentous keratitis is a rare, unilateral, insidious fungal infection that frequently affects normal eyes following ocular trauma associated with vegetative matter and in soft contact lens wearers. Features include a grayish-white infiltrate with indistinct feathery borders, typically surrounded by finger-like satellite infiltrates in adjacent stroma. The infiltrates may extend beyond the epithelial defect. May have an associated ring infiltrate, anterior chamber reaction, and hypopyon (Figures 7-2C,D).
Symptoms
• Pain, photophobia, tearing, decreased vision; may have a history of trauma or corticosteroid eye drop usage
Differential Diagnosis
• Fungal keratitis should be considered in the differential diagnosis of bacterial or herpetic keratitis that does not respond to conventional treatment or has an unusual history or suspicious appearance.
Diagnosis
• History of trauma (which is often minor) involving vegetative matter is highly suggestive.
• Lack of response to conventional antibacterial therapy.
• Corneal scrapings for Giemsa, calcofluor white, or Gomori methenamine silver stain, and culture (may take up to a week for fungus to grow).
• Corneal biopsy may be required if smears and cultures are negative.
Treatment
• Topical natamycin 5% (especially for filamentous fungi) and/or amphotericin B 0.15% (especially for Candida) q 1 h around the clock and taper over 4 to 6 weeks. Patients are often hospitalized initially.
• Oral itraconazole or fluconazole 200 to 400 mg loading dose followed by 100 to 200 mg qd.
• Cycloplegics (e.g., scopolamine 0.25% or atropine 1% tid).
• Corticosteroids are contraindicated.
• Epithelial debridement may facilitate topical therapy by enhancing penetration of antifungals.
• Modify regimen according to clinical response and culture results.
• Therapeutic penetrating keratoplasty may be necessary for unresponsive cases or perforated ulcers. Lamellar keratoplasty is discouraged as there is a high risk of recurrence of infection.
Prognosis
• Fair for mild to moderate infections. Poor for severe infections.
ACANTHAMOEBA KERATITIS
Acanthamoeba keratitis is a rare parasitic infection of the cornea associated with the use of soft contact lenses and inadequate contact lens hygiene (e.g., using tap water or homemade saline solutions, swimming or hot tub use while wearing contact lenses). It should be considered in nonhealing, culture-negative keratitis.
Etiology
• Acanthamoeba species
Symptoms
• Severe pain out of proportion to severity of keratitis, redness, tearing, decreased vision, photophobia, minimal discharge. Symptoms typically develop over a period of weeks, but onset can be more rapid.
• History of soft contact lens use and occasionally trauma.
Signs
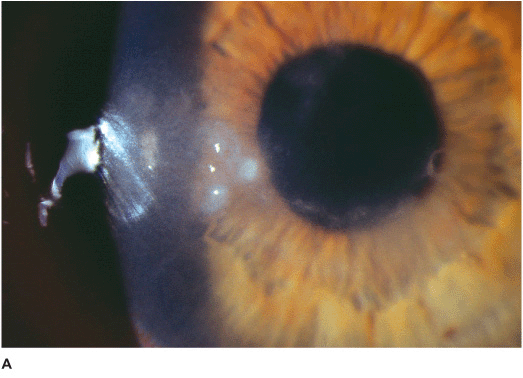
• Epithelial or subepithelial infiltrates appearing as pseudodendrites early on (Figure 7-3A).
• A nonsuppurative stromal ring infiltrate often with variable epithelial breakdown can develop over weeks. The degree of inflammation is disproportionate to the amount of pain (Figures 7-3B,C).
• Radial keratoneuritis.
• In advanced cases, corneal thinning or perforation, scleritis, or hypopyon may develop.
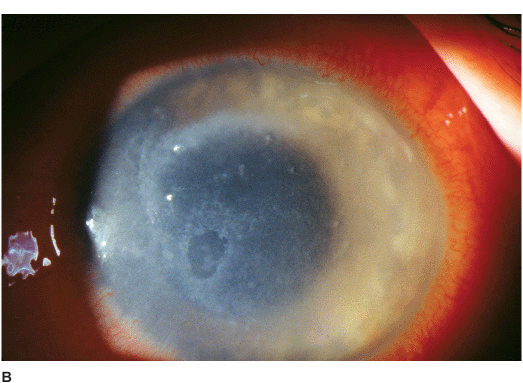
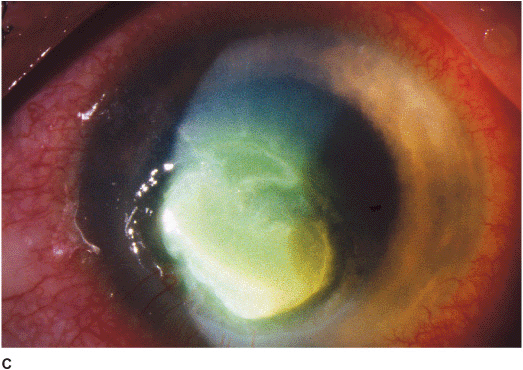
Figure 7-3 Acanthamoeba keratitis A. This early Acanthamoeba infection has several subepithelial infiltrates in a linear pattern reminiscent of a dendrite, hence the term pseudodendrite. There was no frank epithelial defect, but there was epithelial irregularity B. After several weeks, a ring infiltrate can develop, as can be seen especially superiorly. There is a small epithelial defect inferocentrally. C. After several months of antiacanthamoeba treatment, this dense infiltrate is finally scarring. The active infection eventually resolved but the eye was left with a significant corneal scar.
Differential Diagnosis
• Herpes simplex keratitis
• Fungal keratitis
• Bacterial keratitis
Diagnosis
• Pain disproportionate to severity of inflammation.
• Lack of response to antibacterial and antiviral therapy.
• Ring infiltrate and radial keratoneuritis are highly suggestive.
• Corneal scrapings for Gram’s, Giemsa, or calcofluor white stain for amoebic cysts.
• Culture on non-nutrient agar with E. coli overlay or special media (e.g., buffered charcoal yeast extract agar).
• Corneal biopsy may be necessary if smears and cultures are negative.
Treatment
• Propamidine isethionate 1% (e.g., Brolene®) drops q 1 h.
• Neomycin-polymyxin B-gramicidin (e.g. Neosporin®) drops q 1 h.
• Polyhexamethylene biguanide (PHMB) 0.02% drops q 1 h. Chlorhexidine 0.02% can be used as an alternative to PHMB.
• Oral itraconazole 100 to 200 mg qd or ketoconazole 200 mg qd.
• Other drops (e.g., clotrimazole) may be added depending on severity or treatment response of the infection.
• Cycloplegics (e.g., scopolamine 0.25% or atropine 1% tid).
• Low-dose topical corticosteroids may be helpful in reducing inflammation.
• Oral nonsteroidal anti-inflammatory agents or narcotics for pain relief.
• Modify regimen according to clinical response.
• Penetrating keratoplasty may be required if medical therapy fails, but there is risk of recurrence.
Prognosis
• Fair to good if diagnosed and treated appropriately within the first month of development of symptoms. Poor if significant corneal involvement is present.
HERPES SIMPLEX KERATITIS (HSV)
Herpes simplex infection is an extremely common condition affecting a major proportion of the population, although most infections are subclinical. The eyes can be affected in primary ocular herpes or in recurrent disease.
Etiology
Herpes Simplex Virus Type 1 Causes infection above the waist, especially the face, lips, and eyes. Transmitted by close contact. Much more common in the eye.
Herpes Simplex VirusType 2 Causes infection below the waist, particularly the genitalia. Transmitted sexually, but neonates can be infected during vaginal delivery. Uncommon in the eye.
PRIMARY OCULAR HERPES
• Unilateral or bilateral facial and/or eye infection
Etiology and Epidemiology
• Primary contact with HSV
• Usually occurs in children or adolescents
Symptoms
• Fever, flu-like symptoms
• Facial vesicular rash. Ocular redness, pain, decreased vision, and tearing
Signs
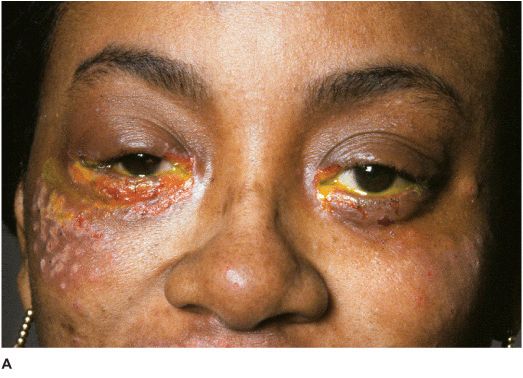
• There may be vesicular blepharoconjunctivitis or periorbital dermatitis. The vesicles usually progress to form crusts (Figures 7-4A,B). There may be associated acute follicular conjunctivitis with preauricular lymphadenopathy.
• The cornea may be involved in the form of coarse macropunctate epithelial keratitis or multiple small branching epithelial dendrites without stromal involvement.
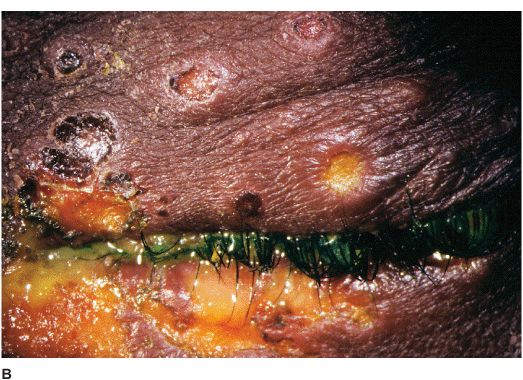
Figure 7-4 Herpes simplex dermatitis A. This patient had recurrent herpes simplex dermatitis. Note the numerous ulcerated skin lesions around the right eye and cheek. The right eye appears uninvolved, but should receive prophylactic treatment due to skin lesions on the eyelid margin. B. Multiple ulcerated skin lesions of herpes simplex can be seen in the upper eyelid. Confluent skin ulcerations are present in the lower eyelid with a mucoid discharge.
Treatment
Blepharoconjunctivitis Vidarabine (e.g., Vira-A®) ointment, trifluridine (e.g., Vir-optic®) drops, or acyclovir (e.g., Zovirax® ophthalmic) ointment 5 times a day.
Corneal Involvement Trifluridine drops (e.g., Viroptic) 9 times a day.
• Consider acyclovir 200 to 400 mg po 5 times a day for 7 to 14 days.
• Consider topical antibiotic ointment or acyclovir to help heal skin lesions away from the eyelid margin.
Prognosis
• Good. This is usually a benign and self-limited condition, but the virus subsequently establishes a latent infection in the trigeminal ganglion and may reactivate, especially during periods of physical or emotional stress, to cause recurrent disease.
RECURRENT OCULAR HERPES SIMPLEX
Recurrent ocular herpes may take the form of infectious epithelial keratitis, non-necrotizing stromal keratitis (disciform keratitis), necrotizing stromal keratitis, neurotrophic keratitis, and keratouveitis.
Etiology and Epidemiology
• Recurrent HSV is due to a reactivation of latent infection in the trigeminal ganglion, especially during periods of physical or emotional stress.
• It occurs in children and adults.
HSV: EPITHELIAL KERATITIS (DENDRITIC ULCER)
Epithelial keratitis is a very common, usually unilateral condition due to the presence of live virus within corneal epithelial cells.
Symptoms
• Unilateral redness, tearing, irritation, decreased vision, photophobia, history of previous episodes
Signs
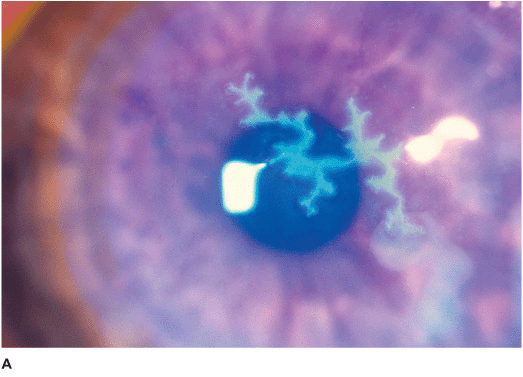
• Single or multiple branching, ulcerating epithelial lesions with raised edges and terminal bulb formation (Figures 7-5A,B).
• Enlargement of ulcers can lead to the formation of an amebic-shaped geographic ulcer.
• The ulcer bed stains with fluorescein. The built-up, swollen, opalescent margins of the lesion containing virus-laden cells stain with rose bengal.
• Anterior stromal haze called “ghost dendrites” may develop below the epithelial lesions (Figure 7-5C).
• Corneal sensation may be diminished.
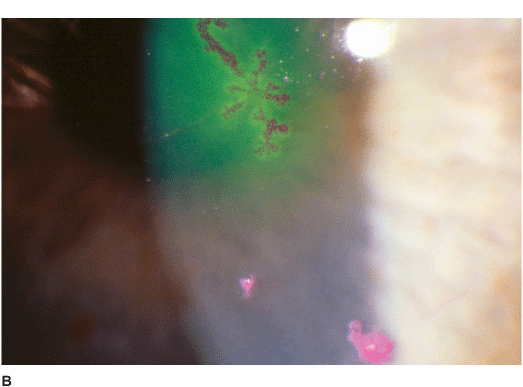
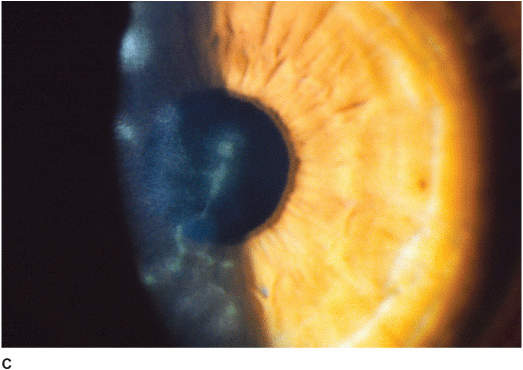
Figure 7-5 Herpes simplex keratitis A. Fluorescein staining with a cobalt blue light of an active herpes simplex epithelial dendrite. Note the “tree branching” pattern of the dendrite. The central bed stains well with fluorescein while the elevated edges do not. The ends of the dendrite have classic terminal “end bulbs.” B. Rose bengal dye on this active dendrite stains the built-up edges which contain virus-laden cells. C. This resolving epithelial dendrite barely stains with fluorescein. There is residual underlying corneal haze in the pattern of the previous dendrite, often termed a “ghost dendrite.”
Differential Diagnosis
• Herpes zoster keratitis: associated with herpes zoster ophthalmicus with typical skin vesicles found along dermatomal distribution of the face. May have elevated epithelial lesions with tapered ends, which lack terminal bulbs. The entire lesion stains with rose bengal and mildly with fluorescein. Prior to development of the typical zoster rash, early zoster dendrites can look very similar to HSV dendrites.
• Acanthamoeba pseudodendrites.
• Healing epithelial defects.
• Toxic epitheliopathy.
Treatment
• Trifluridine (e.g., Viroptic) drops q 2 h during the day or vidarabine (e.g., Vira-A) or acyclovir (e.g., Zovirax ophthalmic) ointment 5 times a day.
• If the patient is already on corticosteroids, they should be tapered rapidly.
• Epithelial debridement can help reduce viral load.
• If there is no response to treatment after 1 week, then poor compliance, resistance to antiviral therapy, antiviral toxicity, or neurotrophic disease should be considered.
• A short course of systemic acyclovir is unnecessary as it does not prevent subsequent development of stromal keratitis or uveitis, but can be used when frequent topical antivirals cannot be given.
• Consider long-term oral antiviral prophylaxis (e.g., acyclovir 400 mg bid) if a patient has had multiple episodes of herpetic eye disease.
Prognosis
• Good. Recurrences are common.
HSV: NON-NECROTIZING STROMAL KERATITIS (DISCIFORM KERATITIS)
Disciform keratitis is a primarily inflammatory condition caused by a hypersensitivity reaction to the herpes simplex viral antigen in the cornea.
Symptoms
• Unilateral redness, tearing, irritation, blurred vision, photophobia, history of previous episodes
Signs
• Central disc of stromal and epithelial edema (Figure 7-6A).
• Small keratic precipitates localized to the underlying endothelium.
• Folds in Descemet’s membrane.
• Surrounding stromal immune ring (Wessley ring) may be present.
• The limbal tissue may be thickened and inflamed (limbitis) (Figure 7-6B).
• Anterior uveitis (Figures 7-6C,D).
• Intraocular pressure may be elevated.
• Corneal sensation is typically reduced.
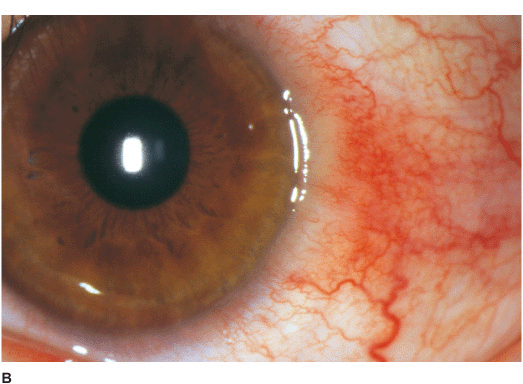
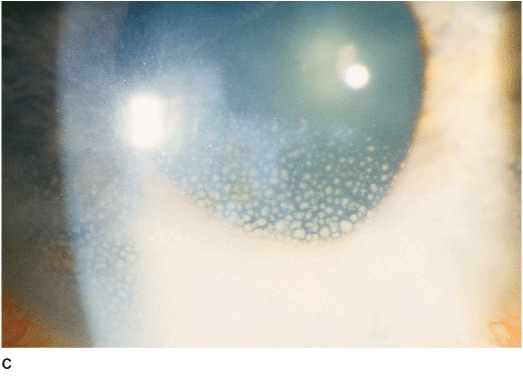
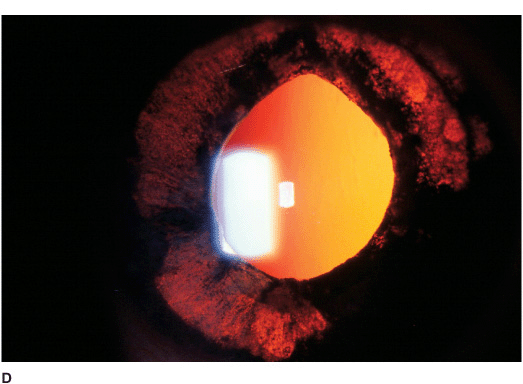
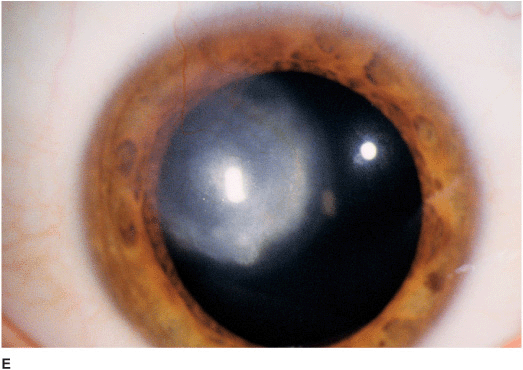
Figure 7-6 Herpes simplex disciform keratitis A. This eye has severe central corneal edema with underlying keratic precipitates. This disciform keratitis represents an inflammatory reaction to previous herpes simplex infection. It may resolve spontaneously, but often responds extremely well to topical corticosteroids with antiviral coverage. Herpes simplex limbitis B. This eye, with a previous history of herpes simplex keratitis, has severe limbal inflammation. Note the thickened, elevated limbal conjunctiva. This limbitis responded to topical corticosteroids and antiviral coverage. Herpes simplex iritis C. Hundreds of granulomatous keratic precipitates are present in this eye with a history of previous herpes simplex keratitis. Note the faint central corneal scarring of old herpes keratitis. Often the intraocular pressure is elevated in eyes with herpetic iritis. Herpes simplex iritis responds to topical corticosteroids with antiviral coverage. It likely benefits from treatment with oral antiviral agents in addition. Herpes simplex keratitis D. Retroillumination off the retina reveals significant iris stromal atrophy and iris transillumination defects after multiple episodes of herpes simplex keratitis and iritis. E. A large dense corneal scar with neovascularization remains after repeated episodes of herpes simplex keratitis.
Differential Diagnosis
• Acute corneal hydrops of keratoconus
• Fuchs’ endothelial dystrophy
• Herpes zoster disciform keratitis
• Contact lens overwear
Treatment
• If inflammation is mild and vision is good, the condition can be observed.
• In more severe cases, topical corticosteroids (e.g., prednisolone 1%, dexamethasone 0.1%, or loteprednol 0.5% drops qid) can be started, maintained for several days to weeks, then gradually tapered over weeks or months. Often, a very low dose (once or twice a week) may be required to prevent recurrent inflammation.
• While on corticosteroids more than once a day, concomitant antiviral drops (qid) or ointment (bid) is often required as prophylaxis.
• If an epithelial lesion is present, it should be treated before starting corticosteroids.
• Consider long-term oral antiviral prophylaxis (e.g., acyclovir 400 mg bid) if a patient has had multiple episodes of stromal keratitis.
Prognosis
• Good. Stromal scarring may occur and reduce vision (Figure 7-6E). Often recurs.
HSV: NECROTIZING STROMAL KERATITIS
Necrotizing stromal keratitis is unusual. It is most likely caused by viral infiltration and inflammation of the corneal stroma.
Symptoms
• Unilateral redness, tearing, irritation, blurred vision, photophobia, pain, history of previous episodes.
Signs
• Necrotic, cheesy, stromal infiltration which may be associated with an epithelial defect (Figure 7-7A).
• The appearance of the infiltrate can be confused with secondary bacterial or fungal keratitis.
• Corneal thinning, stromal neovascularization, scarring or perforation may develop (Figure 7-7B).
• There may be associated keratic precipitates, anterior uveitis, or hypopyon.
• Intraocular pressure can be elevated even in the presence of minimal anterior chamber reaction.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree