Fig. 16.1
A 52-year-old African woman was diagnosed with HIV, with a CD4 count of 56 × 106/mm3 and viral load >100,000 copies/mL. At presentation, a fleshy conjunctival growth extending onto the cornea was initially diagnosed as a pterygium (a). The ocular symptoms worsened with enlargement of the mass, which on CT scan revealed extension into the medial rectus muscle insertion. Conjunctival biopsy confirmed diffusely infiltrating invasive, moderately differentiated squamous cell carcinoma. The patient declined lid-sparing exenteration. In the interim she was started on antiretroviral therapy (zidovudine, lamivudine, and efavirenz). Over the next several months her symptoms improved. At 12 months, CD4 count was 221 × 106/mm3 and the viral load undetectable. The conjunctival mass had completely resolved (b). In the setting of HIV, even Kaposi’s sarcoma and non-Hodgkin’s lymphoma are recognized as responding to highly active antiretroviral therapy via immune restoration (Reproduced with permission from Holkar et al. [46])
16.3.4 Stem Cell Theory
Because of the tendency for OSSN to arise in the limbal area, where the stem cells for the corneal and conjunctival epithelium are located, Lee and Hirst [1] proposed the limbal transition zone/stem cell theory for the development of OSSN. Based on Tseng’s concept of the long-living and high proliferation rate of stem cells in the limbal area, they postulated that alterations in this anatomical site influenced by other factors cause abnormal maturation of the conjunctival and corneal epithelium resulting in the formation of OSSN.
16.4 Clinical Features
16.4.1 Symptoms
In addition to the presence of the lesion on the ocular surface, other symptoms include ocular redness and irritation. Visual acuity is usually not reduced, unless the center of the cornea is affected [14]. OSSN may grow within weeks to years; in most cases, the history is of several months.
16.4.2 Signs
Clinically, it may be difficult to distinguish among conjunctival epithelial dysplasia, carcinoma in situ, and invasive squamous cell carcinoma, although suspicion towards one of these three lesions may exist. These lesions arise commonly within the interpalpebral fissure, mostly at the limbus, although they may be found in any part of the conjunctiva and cornea (Figs. 16.2, 16.3, and 16.4). OSSN may appear gelatinous with superficial vessels; papilliform, when it has a papillary appearance; or leukoplakic, with a white keratin plaque covering the lesion [3]. It may also appear as a nodular lesion, especially when it is invasive SCC or as a diffuse lesion masquerading as chronic conjunctivitis. Usually OSSN appears as a nonpigmented lesion, although pigmented conjunctival SCC has been reported (Fig. 16.5). According to one study, clinical characteristics that may predict high-grade lesions are temporal and superior location, papillomatous and nodular-appearing lesions, and size of lesional area [5].
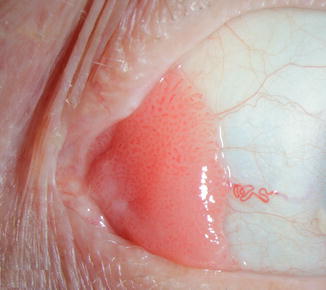
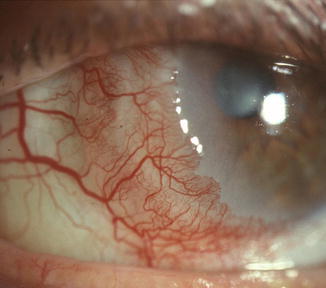
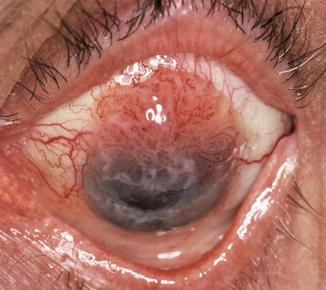
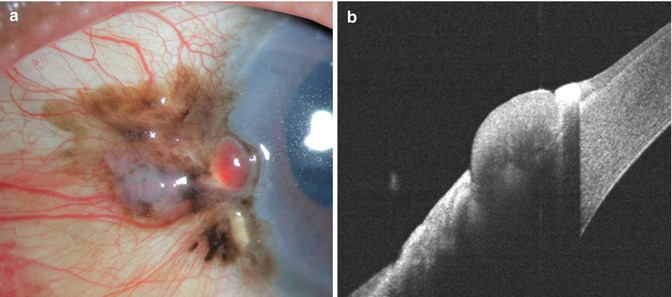

Fig. 16.2
Papillomatous ocular surface squamous neoplasia

Fig. 16.3
Carcinoma in situ with early subepithelial invasion growing from the temporal conjunctiva into the cornea

Fig. 16.4
Papillary conjunctival SCC invading into the upper half of the cornea

Fig. 16.5
Invasive conjunctival squamous carcinoma in an Indian man. The lesion is pigmented, resembling conjunctival melanoma (a). Anterior segment OCT indicates superficial epithelial involvement with shadowing due to pigmentation (b)
16.5 Differential Diagnosis
The main lesions in the differential diagnosis of OSSN are pinguecula, pterygium, and squamous papilloma [3].
16.6 Diagnostic Evaluation
It may be difficult to clinically distinguish between intraepithelial and invasive squamous neoplasia and between them and other lesions such as pinguecula and pterygium, especially with leukoplakia and squamous papilloma.
16.6.1 Fluorescein Staining
In our experience, the use of fluorescein staining can help in the diagnosis, emphasizing the papillary or granular surface of part of the OSSN lesion and delineating its borders (Fig. 16.6) [14, 15]. Others have used rose bengal staining [16] or 1 % toluidine blue eyedrops [17].
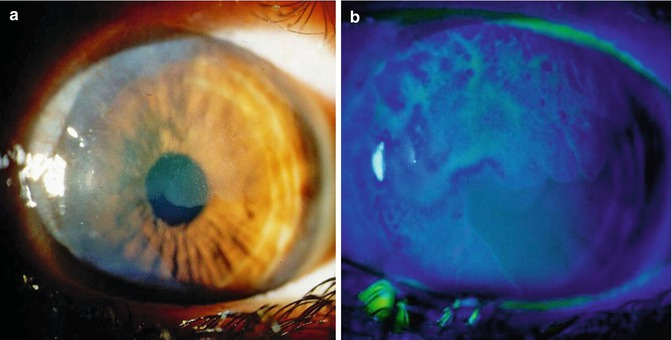

Fig. 16.6
Diffuse corneal involvement by CIN showing hazy and irregular corneal surface (a). Staining with fluorescein shows the granular surface of the involved cornea and clearly delineates the border between the affected and non-affected areas (b)
16.6.2 Imaging Techniques
Recent publications have described the use of high-frequency ultrasound in the diagnosis of OSSN and particularly in estimating the depth of invasion [18]. Others have used confocal microscopy [19] or ultrahigh resolution optical coherence tomography (Chap. 13) [20]. However, the definitive diagnosis must be a histological one.
16.6.3 Diagnostic Cytology
Preoperative cytologic diagnosis may be of value in planning surgery in order to prevent unnecessary removal of large pieces of normal conjunctiva in the case of a benign lesion and to prevent partial excision in cases of malignant lesions.
16.6.3.1 Exfoliative Cytology
Cells from the conjunctival surface are obtained by platinum spatula, brush, or cotton-wool tip. Papanicolaou and Giemsa stains are used to examine the specimen [21, 22]. The advantages of this technique are the ability to obtain prospective cytologic information on the nature of the lesion, mainly in differentiating between benign and malignant lesions, and in the ability to sample multiple sites and in easy follow-up evaluation after treatment. The major disadvantage is the superficial nature of the sample of the tissues obtained by this technique. Sometimes only keratinized cells are obtained. It does not provide information about the degree of the tumor invasion, which may be crucial in the overall management.
16.6.3.2 Impression Cytology
Another method of obtaining cells from the surface of the conjunctival lesion is by impression cytology [23]. In this technique several types of filter paper, such as cellulose acetate filter paper, millipore filter paper, or biopore membrane device, are gently placed in contact with the ocular surface, sampling the most superficial cells. These are fixed and stained with Papanicolaou stain. The advantages and disadvantages of exfoliation cytology also apply to impression cytology.
16.7 Histopathologic Features
Only histological evaluation of excised lesions, either from incisional or excisional biopsy, can differentiate between the three lesions within the spectrum of OSSN [1, 2].
16.7.1 Dysplasia
Dysplastic lesions exhibit mild, moderate, or severe degrees of cellular atypia that may involve various thicknesses of the epithelium, starting from the basal layer outwards (Fig. 16.7a). They show modification of epithelial cell organization with various degrees of loss of the normal cellular polarity. Usually the most superficial layers are uninvolved. In cases with severe dysplastic changes, it may be difficult to distinguish the lesion from carcinoma in situ.
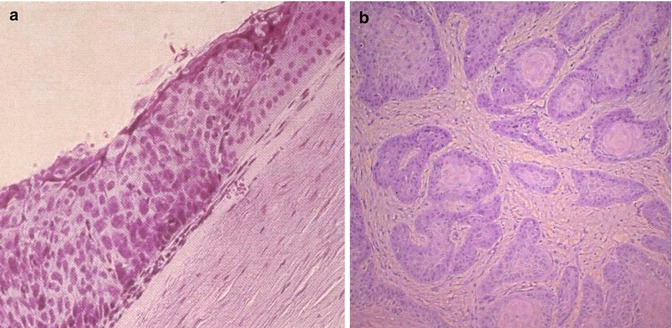

Fig. 16.7
Histological picture of acanthotic conjunctival epithelium with dysplastic changes involving most of the epithelial thickness. The epithelium lost its normal cellular polarity. Normal conjunctival epithelium is seen on the right side (a, hematoxylin and eosin, original magnification × ~ 100). Note deep invasion of tumor cell islands of well-differentiated conjunctival SCC (b, hematoxylin and eosin, original magnification × ~ 100)
16.7.2 Carcinoma In Situ
Carcinoma in situ may exhibit all the histological features of SCC. However, it usually remains confined to the epithelium, respecting the basement membrane. Carcinoma in situ usually shows a total loss of normal cellular maturation, affecting the full thickness of the epithelium. The cells are large and usually elongated. Keratinized cells may be identified and mitotic figures can be present in all layers.
16.7.3 Invasive Squamous Cell Carcinoma
Invasive squamous cell carcinoma shows features similar to carcinoma in situ, but the basement membrane of the epithelium is breached and the subepithelial tissue of the conjunctiva is invaded (Fig. 16.7b). Most of the conjunctival SCC are well differentiated and they often show surface keratinization. The tumor may show various degrees of cellular pleomorphism. In examining such lesions, hyperplastic and hyperchromatic cells, individually keratinized cells (dyskeratosis), concentric collections of keratinized cells (horn pearls), loss of cellular cohesiveness, and atypical mitotic figures may be observed. The subepithelial tissue in invasive SCC is usually inflamed and contains islands of atypical epithelial cells. In pigmented individuals, OSSN can be pigmented due to abnormal proliferation of melanocytes in the lesions.
16.7.4 Histopathologic and Clinical Variants
Several types of invasive conjunctival SCC with rather aggressive behavior have been reported [2]. Because of the aggressiveness of these variants that often invade the eyeball and the orbital tissue and even metastasize to lymphatics and distant sites, they should be histopathologically differentiated from less aggressive conventional SCC.
16.7.4.1 Spindle Cell Squamous Carcinoma
Spindle cell variant of SCC exhibits spindle-shaped cells that may be difficult to distinguish from fibroblasts.
16.7.4.2 Mucoepidermoid Carcinoma
Mucoepidermoid carcinoma is a variant of conjunctival SCC that shows, besides the squamous cells, mucus-secreting cells that are positively stained for mucopolysaccharides.
16.7.4.3 Adenoid Squamous Cell Carcinoma
Another variant of conjunctival SCC with aggressive behavior is the adenoid squamous carcinoma, which histologically shows extracellular hyaluronic acid but no intracellular mucin.
16.7.4.4 Squamous Cell Carcinoma of Cornea
OSSN arising in the corneal epithelium is rarely observed [7, 24]. There is controversy about its origin. Some authors support the possible potential of the corneal epithelium to undergo dysplastic and cancerous changes, while others believe that the origin of corneal OSSN is at the limbus (Fig. 16.8). Histologically, the corneal CIN is similar to that in the limbus and conjunctiva. Usually the Bowman’s layer is intact. Corneal CIN has a tendency to recur because of inadequate scraping, but with current methods of treatment, this rarely occurs [14, 15].
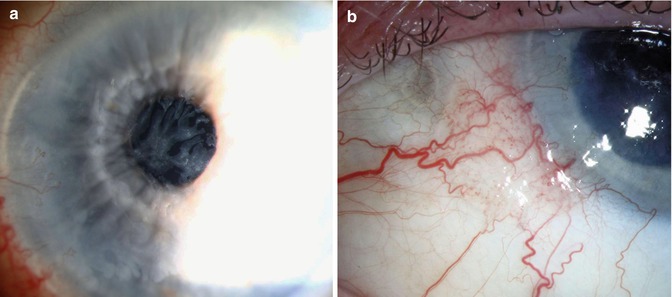

Fig. 16.8
Squamous cell carcinoma of cornea resembling filamentary keratitis (a). Examination of the limbus reveals small area of conjunctival thickening with fine intrinsic vascularity (b)
16.7.4.5 Squamous Cell Carcinoma in Anophthalmic Socket
Review of nine published cases indicates a long interval between enucleation and the diagnosis (mean, 45 years), predominant diffuse involvement of the upper eyelid, advanced stage at diagnosis, and tendency for metastases (Fig. 16.9). It is speculated that presence of prosthesis-induced chronic inflammation may be a factor in initiation and masking of signs and symptoms of OSSN [25].
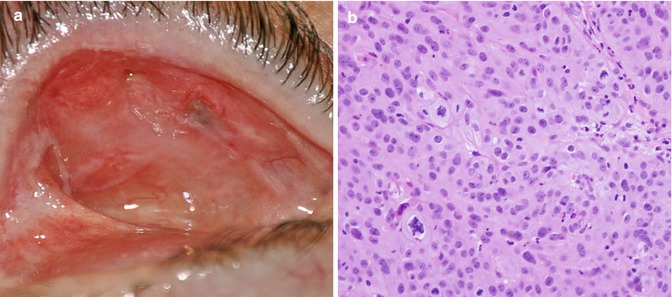

Fig. 16.9
Recurrent squamous cell carcinoma in the superior nasal aspect of tarsal conjunctiva of the upper lid in anophthalmic socket (a). Poorly differentiated invasive carcinoma without keratinization was associated with an overlying squamous carcinoma in situ. Although some areas were suggestive of sebaceous differentiation, oil red O and immunostaining for androgen receptor were both negative (b, 40× magnification, hematoxylin and eosin) (Reproduced with permission from Espana et al. [25])
16.8 Treatment
16.8.1 Surgery
Surgical excision of the OSSN lesion is the traditional method of treatment (Chap. 21). Its success depends on the involvement of the peripheral and deep surgical margins. In order to avoid recurrence, it is recommended to excise the tumor tissue with wide surgical margins of 2–3 mm [3, 26]. When the deep cornea or sclera are involved, deep lamellar keratectomy or sclerectomy is performed. Recurrence rates following excision of OSSN alone range from 15 to 52 %, with an average of 30 % [1]. Erie et al. [7] found 5 % recurrence when the surgical margins were free and 53 % recurrence when they were involved. Similarly, Pizzarello and Jakobiec found 69 % recurrence when dysplastic tissues were left at the surgical margins [3].
Therefore, techniques to ensure clear surgical margins have been applied. Frozen sections were used by Char et al. to assess the surgical margins [27]. However, there was a disparity between the apparently free surgical margins and recurrence of the OSSN. Buus et al. have used a modified Mohs’ micrographic technique that was developed for cutaneous tumors, in order to ensure clear surgical margins [28]. No recurrences were documented in their series of 19 patients.
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


