Purpose
To analyze corneal confocal microscopy changes after combined photorefractive keratectomy (PRK) plus the cross linking (CXL) procedure.
Design
Prospective interventional case series.
Methods
At the Department of Basic Medical Sciences, Neuroscience, and Sense Organs of the University of Bari, Bari, Italy, 17 eyes of 17 patients with progressive keratoconus underwent confocal microscopy examination before and after 1, 3, 6, 12, 18, and 48 months following PRK plus the CXL procedure. The main outcome measures were mean superficial epithelial cell density; mean basal epithelial cell density; mean anterior, mid and posterior keratocyte density; qualitative analysis of stromal backscatter; sub-basal and stromal nerve density parameters; and mean endothelial cell density.
Results
During the 4-year follow-up, the mean superficial epithelial cell density, mean basal epithelial cell density and mean endothelial cell density remained unchanged ( P > 0.05). The anterior mid-stromal keratocyte density showed a significant decrease ( P < 0.05) as compared with preoperative values, and the posterior stromal keratocyte density showed a significant increase at 1 and 3 months of follow-up. Sub-basal and stromal nerve density parameters were significantly decreased until postoperative month 6 ( P < 0.05 at 1, 3, and 6 months) and then tended to increase up to preoperative values by the 18th postoperative month.
Conclusion
Corneal changes after the PRK plus CXL procedures seem to be pronounced and long lasting as far as keratocyte density of the anterior and mid stroma is concerned. Sub-basal nerve densities tend to reach preoperative values 6 months after surgery.
Corneal cross-linking (CXL) with riboflavin and ultraviolet-A (UVA) is currently the predominant treatment for mild and moderate progressive keratoconus. It is generally preferred to keratoplastic surgeries because it is noninvasive and requires a shorter visual rehabilitation time and, in particular, because there is no risk for graft failure. However, there are instances in which keratoplasty is preferred to CXL for the treatment of patients with severe keratoconus with corneal scarring.
Since CXL was introduced, the technique has been enhanced by combining certain elements of photorefractive keratectomy (PRK) to improve the refractive outcomes. Instead of mechanical removal of the epithelium, according to the technique described by Wollensak and associates, ablation of the epithelium and superficial stroma is used in the PRK plus CXL procedure to remodel the corneal surface and reduce irregular astigmatism. In our experience, as compared with standard CXL, the PRK plus CXL procedure offers better visual outcomes in terms of lower keratometric values, changes in the elevation of the anterior corneal surface and greater reductions in root mean square values. Some studies have reported changes in corneal structure investigated by in vivo confocal microscopy after CXL alone, but only a few studies have been made of the outcomes of the PRK plus CXL procedure, and a follow-up of 4 years has never been reached previously.
The aim of this study was to analyze morphologic changes in all corneal layers, as examined by confocal microscopy, over a 4-year follow-up period after PRK plus CXL for keratoconus.
Methods
Subjects
The study protocol was approved and monitored by the Local Ethics Committee of Azienda Ospedaliera Policlinico di Bari (protocol no. 839/C.E.) and conformed to the tenets of the Declaration of Helsinki. Informed consent was obtained from all patients taking part in the study. This is a registered clinical trial: clinical trial registration at http://www.controlled-trials.com/ISRCTN57262986 .
Between July 2008 and June 2009, 17 patients with keratoconus (34 eyes) (mean age, 31.17 ± 8.12 years; range, 21–46 years) were recruited at the Cornea Service of the Department of Basic Medical Sciences, Neuroscience, and Sense Organs of the University of Bari to take part in this prospective nonrandomized clinical study. The 34 eyes were graded as stage I-III according to the Aliò and Shabayek classification and were assigned to 1 of 2 groups: the worse eye underwent PRK followed by CXL (17 eyes), and the better eye (fellow eye) underwent CXL alone (17 eyes). The diagnosis of keratoconus was based on corneal topography (OrbscanIIz; Bausch & Lomb, Rochester, New York, USA) as an asymmetric bowtie pattern, with or without skewed axes, and a paracentral inferior-superior dioptric difference of more than 1.4 diopters (D).
Enrolment depended on documented progression of keratoconus in the previous 6 months, a corneal thickness of at least 450 μm at the thinnest point in the worse eye, hard contact lens and full spectacle correction intolerance, and age older than 18 years. Progression of keratoconus was defined based on serial differential topography, as an increase in the apex keratometry by more than 1.0–1.5 D and a corresponding change (>1.0–1.5 D) in the refractive cylinder in the previous 6 months. Exclusion criteria were a corneal thickness of less than 450 μm at the thinnest point in the worse eye, corneal scarring or Vogt striae, any ocular disorder other than keratoconus, any history of eye surgery, any systemic disease, pregnancy, and contact lens wear. Patients who failed to attend the follow-up visits were excluded.
Visual acuity, refractive, topographic, and corneal higher order aberrations outcomes after PRK plus CXL and after CXL alone up to 24 months of follow-up have already been reported. The current study investigated the confocal microscopy findings in 17 corneas of 17 patients after the PRK plus CXL procedure over a 4-year follow-up period. In this trial the eyes treated only by CXL were not taken into consideration because there are already many publications concerning confocal microscopy analyses in these eyes, reporting similar results in all cases.
Topographic-Guided PRK Plus CXL Procedure
The steps of topographic-guided PRK plus CXL are reported briefly below. After acquisition of the corneal shape by means of the Scheimpflug-based Precisio tomographer (Ligi Tecnologie Medicali, Taranto, Italy), elevation data together with the patient’s subjective refraction were imported into Corneal Interactive Programmed Topographic Ablation (CIPTA) software to plan the ablation procedure. To ensure the maximum reduction of irregularities with minimum tissue consumption, the Restored Morphological Axis strategy of the CIPTA software was used. The center of the planned ablation was the corneal apex in all treated eyes. The stromal ablation depth was between 18 and 49 μm (mean, 31.1 ± 9.5 μm). A supplementary depth of 50 μm was selected by the surgeon for epithelium ablation because of transepithelial PRK. The corneal epithelium was removed by laser within a 9 mm diameter. Transepithelial topography-guided PRK was performed with the high-resolution 1-KHz flying spot laser iRES (Ligi Tecnologie Medicali). Immediately after PRK, riboflavin 0.1% solution (10 mg riboflavin-5-phosphate in 20% dextran T500 solution; Ricrolin; SOOFT Italia, Montegiorgio, Italy) was administered topically every 2 minutes for 30 minutes, under topical anesthesia. Riboflavin absorption throughout the corneal stroma and anterior chamber was confirmed on slit-lamp examination. Then the cornea was exposed to UVA 365 nm light at an irradiance of 3.0 mW/cm 2 for 30 minutes. During UVA exposure, riboflavin drops were continued every 2 minutes. At the end of the procedure, a soft bandage contact lens was applied and ofloxacin and 0.1% indomethacin drops were administered until re-epithelialization was complete. After epithelial healing and removal of the contact lens, 0.1% fluorometholone was instilled 4 times daily for the first month. The dosage was tapered by 1 drop monthly over the next 3 months.
Confocal Microscopy and Outcomes
The Confoscan 4 (NIDEK Technologies, Padova, Italy) confocal microscope equipped with a 40× objective lens was used in all eyes. After the instillation of local anesthetic (benoxinate hydrochloride 0.4 g eye drops, Alfa Intes Industria Terapeutica Splendore, Casoria, Italy), the instrument lens was advanced until the high-viscosity gel (hydroxypropyl methylcellulose 2.5%) contacted the cornea. A fixed device setting was used for all examinations: full-thickness mode, 72% light intensity; 7 μm scan step; autoalignment function. The Z-ring was used in all cases. Each image represented a coronal section of 460 × 345 μm with magnification of ×500. A total of 4 scans were obtained at the optical center of each cornea for all patients at each follow-up visit. The confocal microscopy examination was performed by the same researcher in all study participants. The best-focused 3 frames per layer were selected for analysis of each cornea. Evaluations were repeated at 1, 3, 6, 12, 18, and 48 months postoperatively. Image parameters were:
- (1)
Superficial epithelial cell density (cells/mm 2 ); a minimum of 35 cells were counted manually by Confoscan 4 NAVIS analysis software in a randomly selected rectangular area. The software highlights the results as unreliable if fewer than 35 cells are present in the selected rectangle.
- (2)
Basal epithelial cell density (cells/mm 2 ). At least 75 cells were counted manually by Confoscan 4 NAVIS analysis software in a randomly selected polygonal area.
- (3)
Keratocyte density (cells/mm 2 ) in the anterior, mid, and posterior thirds of stroma. The stroma was divided into 3 equal-thickness anteroposterior regions, and 3 frames were selected from each of these 3 regions, expressed as percent stromal depth (1%–33%, 34%–66%, and 67%–100%). Images of the anterior boundary of the stroma immediately posterior to the Bowman layer (the layer with the highest concentration of keratocyte nuclei ) were included in the analysis of each eye at each follow-up time. At least 35 keratocyte nuclei were counted manually, marking each cell or nucleus inside a randomly selected rectangular area. Cells that touched the top or left edge of the rectangle were counted, but cells that touched the bottom or right edge were not.
- (4)
Sub-basal nerve density, defined as the mean total length (μm) within the region of interest (by default, 81,972.17 μm 2 ), mean total length of the nerves per image (μm/mm 2 ), and mean total number of nerves within the region of interest (including main nerve trunks and branches). The semiautomated Nerves Tracking Tool of NAVIS software was used in all cases.
- (5)
Stromal nerve density, defined as the total number of nerves.
- (6)
Endothelial cell density (cells/mm 2 ). In a randomly selected polygonal area, at least 75 cells were counted by Confoscan 4 NAVIS software.
Qualitative analysis of corneal haze was also performed. Evaluation of stromal backscatter served to identify the corneal layer associated with pathologic scatter and for follow-up of corneal haze.
All counts were performed independently, by 2 experienced examiners blinded to the study identifiers. Comparisons between preoperative and postoperative values at each time point were performed for all parameters by paired-samples t test. A P value of less than 0.05 was considered statistically significant.
Results
The clinical features of the subjects are shown in Table 1 .
| Baseline Data | 4-year Postoperative Data | |
|---|---|---|
| CCT, μm | 516 ± 26.05 | 486.8 ± 28.3 |
| CTT, μm | 489.2 ± 23.6 | 451.8 ± 29.7 |
| SimK1, D | 44.83 ± 11.67 | 42.98 ± 3.01 |
| SimK2, D | 47.41 ± 12.44 | 44.86 ± 4.23 |
| SimKavg, D | 46.12 ± 12.04 | 43.98 ± 3.34 |
| Kapex, D | 54.46 ± 14.3 | 49.87 ± 4.67 |
The mean values and P values for comparisons of the preoperative and the postoperative results are shown in Table 2 .
| Preoperatively | Postoperatively | ||||||
|---|---|---|---|---|---|---|---|
| 1 mo. | 3 mo | 6 mo | 12 mo | 18 mo | 48 mos. | ||
| Superficial epithelial cell density (cells/mm 2 ) | 1481 ± 597.5 (636–2561) | 1414.4 ± 448.6 (831–2127) | 1414 ± 484.4 (841–2110) | 1294.3 ± 465.9 (820–2009) | 1385.6 ± 423 (879–2078) | 1234.8 ± 371.4 (724–2069) | 1308.7 ± 414.3 (897 to 2523) |
| P value | 0.74 | 0.74 | 0.21 | 0.48 | 0.15 | 0.28 | |
| Basal epithelial cell density (cells/mm 2 ) | 5759.5 ± 1785.3 (2891–8743) | 5459.3 ± 1300.3 (3256–7958) | 5412.7 ± 1324.4 (3674–7700) | 5653.4 ± 2057.5 (1775–8835) | 6150.1 ± 1513.2 (3984–8041) | 5735.7 ± 1467.8 (3827–8020) | 6020.7 ± 1094.5 (4034 to 7839) |
| P value | 0.60 | 0.51 | 0.87 | 0.58 | 0.96 | 0.65 | |
| Anterior stromal keratocyte density (cells/mm 2 ) | 880.9 ± 199.7 (643–1345) | NA | NA | NA | 138.3 ± 247.4 (0 to 518) | 210.3 ± 262.3 (0–768) | 253 ± 295.3 (0 to 798) |
| P value | 1.98 10 −9 | 1.98 10 −9 | 1.98 10 −9 | 3.88 10 −7 | 2.81 10 −6 | 8.08 10 −5 | |
| Mid stromal keratocyte density (cells/mm 2 ) | 618.3 ± 90.7 (534–710) | NA | NA | NA | 68.54 ± 127.5 (0–305) | 99.2 ± 139.2 (0–315) | 102.2 ± 143.5 (80 to 308) |
| P value | 4.78 10 −15 | 4.78 10 −15 | 4.78 10 −15 | 8.53 10 −11 | 3.18 10 −9 | 3.54 10 −9 | |
| Posterior stromal keratocyte density (cells/mm 2 ) | 736.2 ± 97.6 (516–909) | 831.3 ± 86.3 (635–969) | 830.2 ± 88.7 (642–956) | 782.8 ± 139.7 (590–1006) | 695.9 ± 73.8 (603–832) | 740.6 ± 157.5 (547–1080) | 647.2 ± 167.2 (339 to 920) |
| P value | 0.04 | 0.02 | 0.35 | 0.20 | 0.93 | 0.06 | |
| Endothelial cell density (cells/mm 2 ) | 2945 ± 115 (2731–3012) | 2932.2 ± 269.7 (2171–3365) | 2965.8 ± 216.9 (2180–3152) | 2947.7 ± 105 (2734–3105) | 2978.2 ± 65.3 (2856–3098) | 2984 ± 67.3 (2828–3084) | 2981.3 ± 60.1 (2876 to 3087) |
| P value | 0.84 | 0.63 | 0.93 | 0.23 | 0.12 | 0.11 | |
| Total length of sub-basal nerves (μm) | 588 ± 97 (419.6–724.8) | NA | NA | 300.6 ± 161 (280.7–529.8) | 397.9 ± 145.7 (332.9–600.6) | 487.7 ± 119.8 (187.9–671.8) | 554.6 ± 137.5 (237.6 to 712.7) |
| P value | 3.03 10 −14 | 3.03 10 −14 | 1.22 10 −5 | 7.01 10 −3 | 0.01 | 0.35 | |
| Length density of sub-basal nerves (μm/mm 2 ) | 9610.6 ± 1325.9 (7865–11765) | NA | NA | 5245.2 ± 931.8 (3829–6463) | 5983.4 ± 1327.9 (3127–7489) | 8085.4 ± 2875.9 (2293–11232) | 9921.9 ± 1173.6 (7865 to 11977) |
| P value | 1.82 10 −15 | 1.82 10 −15 | 4.46 10 −8 | 6.66 10 −7 | 0.05 | 0.51 | |
| Total number of sub-basal nerves (no.) | 6 ± 2.5 (2–10) | NA | NA | 4.5 ± 1.8 (2–9) | 4.8 ± 1.3 (3–7) | 6 ± 1.8 (3–8) | 6.2 ± 1.9 (3 to 10) |
| P value | 4.75 10 −8 | 4.75 10 −8 | 0.1 | 0.16 | 1 | 0.68 | |
| Total number of stromal nerves (no.) | 2.1 ± 1 (1–4) | NA | NA | 1.9 ± 1.1 (0–4) | 2.2 ± 1.1 (1–4) | 2.4 ± 1 (1–4) | 2.3 ± 1 (1 to 4) |
| P value | 1.47 10 −7 | 1.47 10 −7 | 0.58 | 0.75 | 0.48 | 0.66 | |
Compared with baseline values, mean superficial epithelial density, mean basal epithelial cell density, and mean endothelial cell density were not significantly different at each postoperative time point. Anterior stromal keratocyte nuclei were present in no eyes (n = 0) at 1, 3 and 6 postoperative months; in 23.5% of eyes (n = 4) at 12 months; in 35.2% of eyes (n = 6) at 18 months; and in 41.1% of eyes (n = 7) at 48 months. Mid stromal keratocyte nuclei were present in no eyes (n = 0) at 1, 3 and 6 postoperative months; in 23.5% of eyes (n = 4) at 12 months; and in 35.2% of eyes (n = 6) at 18 and 48 months. The mean anterior and mid stromal keratocyte densities were significantly lower at each follow-up as compared with the preoperative values and in no case reached the preoperative value. Compared with baseline values, mean posterior keratocyte density at 1 and 3 months was significantly increased, while at 6, 12, 18, and 48 months it was not statistically different. Postoperatively, sub-basal nerves were present in no (n = 0) eyes at 1 and 3 months; in 82.3% of eyes (n = 14) at 6 months; in 94.1% of eyes (n = 16) at 12 months; and in 100% of eyes (n = 17) at 18 and 48 months. Postoperatively, stromal nerves were present in no eyes (n = 0) at 1 and 3 months; in 88.2% of eyes (n = 15) at 6 months; and in 100% of eyes (n = 17) at 12, 18 and 48 months. The mean total length of nerves, mean total length of nerves per image, mean total number of sub-basal nerves, and mean total number of stromal nerves were drastically lower and undetectable at 1 and 3 months postoperatively and then tended to increase up to 18 months postoperatively. By 4-year follow-up, the preoperative values for mean total length of nerves, mean total length of nerves per image, mean total number of sub-basal nerves, and mean total number of stromal nerves were restored.
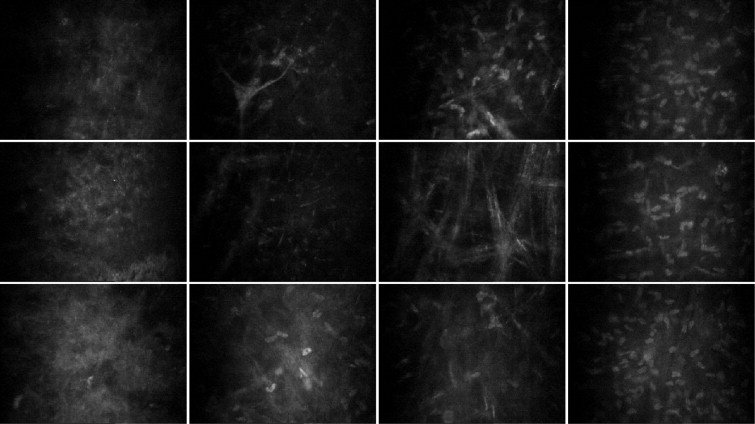
Confocal images of corneal stroma at different time points in a 32-year-old patient are illustrated in Figure 1 and Figure 2 . Confocal microscopy findings in keratoconic eyes before treatment are not reported because they have already been described and are very similar to ours.