Complications of Chronic Otitis Media
Jeffrey P. Harris
David W. Kim
David H. Darrow
A mastoid abscess was first opened. A sinus persisted; bony sequestra were then removed to expose the dura. After probing, an incision was made into the dura and a finger introduced; this resulting in a release of pus. The cavity was irrigated with barley water containing a little turpentine and balsam. A silver tube was made and this was inserted for drainage. The patient recovered.
—S.F. Morand, 1768 (1)
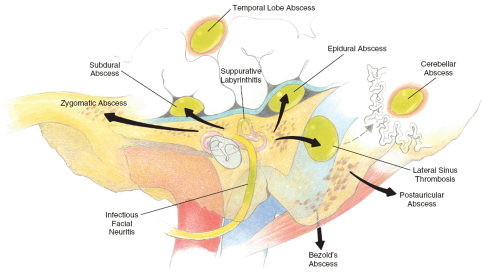
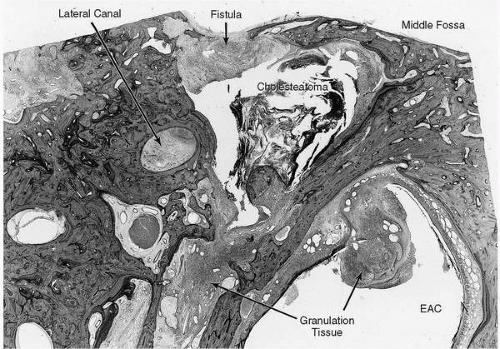
Complications of chronic otitis media (COM) occur when chronic infection and/or cholesteatoma within the middle ear and mastoid spaces extend to regions beyond their bony confines. They result from the erosion of bone by cholesteatoma or by the spread of infection through intact bone via small venules or preformed pathways. They may involve adjacent sites within the temporal bone, the posterior or middle cranial fossae, or the extratemporal soft tissues or more distal sites reached by hematogenous spread (Fig. 18.1).
Once considered commonplace in the course of otitis media and mastoiditis, otogenic complications have become rare sequelae in these disorders. The advent of modern antibiotic therapy has brought a dramatic decline in the incidence and morbidity of otogenic complications. In 1935 Kafka (2) studied a series of 2,100 patients with acute mastoiditis and 1,125 patients with chronic mastoiditis and reported an intracranial complication rate of 6.4%; mortality was 76.4% among those with complications. In contrast, a series of 100 patients with central nervous system (CNS) complications of active ear disease studied by Gower and McGuirt (3) in 1983 represented less than 0.5% of all admissions coded for intracranial complications or mastoid surgery, and mortality was 10% in this group. Similarly, an autopsy review by Courville (4) found a tenfold reduction in the death rate from acute and chronic ear disease attributable to the development of antibiotics. Prellner and Rydell estimate a reduction of the incidence of intracranial complications from 2% to 0.02% with the advent of antibiotics (5).
An unfortunate consequence of the decline of otogenic complications has been a decreased recognition of the impending complications of otitis media. Fever and inflammation, which once heralded the progression of disease, are now often suppressed by medical therapy. Thus the effectiveness of modern therapy and the low incidence of complications have led to complacency in the treatment of middle ear infections and a lack of familiarity with the manifestations of complications.
The otolaryngologist must bear in mind that complications of COM, especially in the immunocompromised host, may rapidly result in death. Therefore it is imperative that he or she be familiar with the clinical manifestations of these complications.
FACTORS IN THE SPREAD OF INFECTION
Most reports published during the antibiotic era suggest that otogenic complications are more likely to arise from cases of chronic middle ear disease rather than from acute processes, though complications due to acute otitis media may be more common in the pediatric age group (6, 7, 8). The pathophysiology and initial management of COM have been enumerated in previous chapters. However, several factors determining the spread of infection beyond the middle ear space bear mention here:
Species and virulence of the organism. Aerobic bacteria may be cultured from some 70% of middle ears with COM, and Proteus sp. and Pseudomonas aeruginosa are the most common offending organisms (9). Anaerobes are present in nearly equal frequency, most notably Bacteroides sp., Peptococcus/Peptostreptococcus sp., and Propionibacterium acnes. Mixed infections are present in more than 50% of these cases. Many organisms cultured from chronic suppurative effusions may represent colonization of the ear, and their preponderance may mask the presence of more virulent organisms.
Host conditions. The host tissue responds to the release of toxic substances from the infecting organisms by mounting an inflammatory reaction, and cells involved in the response assist in the removal of the offending bacteria. Several conditions associated with immune
compromise, such as infancy, old age, diabetes, malnutrition, malignancy, acquired immunodeficiency syndrome (AIDS), and immunodeficiency, are known to diminish the effectiveness of this response and therefore increase the likelihood of spread and the risk of complications. Under these conditions, opportunistic organisms may predominate among the tympanic flora and colonizing organisms may become more virulent. Recent work suggests that patients with complications of COM are more likely to be younger (less than 25 years old) and that younger patients undergoing mastoid surgery for COM were more likely to have complications (10). Disease may be more aggressive in the pediatric population because of increased exposure to respiratory pathogens, chronicity of disease, and immaturity of immune defenses.
Prior therapy. Although topical antibiotic combinations are commonly used to treat COM, they are often met by resistant organisms and are unreliably distributed to the affected area. Ciprofloxacin, an antipseudomonal antibiotic, has demonstrated efficacy in the control of chronic ear disease in adults (11,12). However, timely surgical intervention, aimed at debridement of the middle ear and mastoid spaces, establishment of adequate drainage, and reconstruction of defects in the tympanic membrane, remains the cornerstone of therapy in the prevention of otogenic complications. In tympanomastoid surgery, Merchant et al. reported poorer control of infection after surgery for COM with granulation tissue compared with COM with cholesteatoma. This may be due to an inherent problem of the mucosa. Thus even after disease and unfavorable anatomy are removed, the abnormal mucosa predisposes to the production of more granulations. This variant of COM would therefore be more likely to produce complications (13).
PATHWAYS IN THE SPREAD OF INFECTION
When the mucosal defense system is unable to deter the progression of disease, the integrity of the bone of the middle ear becomes critical. Infection must extend through this bone for complications of middle ear disease to occur. Spread of infection through the osseous barrier may occur in the following several ways:
Direct erosion through bone demineralized by cholesteatoma or chronic inflammation.
Thrombophlebitic spread through small veins and haversian canals to the dural sinuses.
Extension through normal anatomic pathways, including the oval and round windows, the cochlear and vestibular aqueducts, and bony dehiscences and fissures.
Extension through iatrogenically or traumatically created bony defects.
Direct extension is the route most commonly implicated in complications due to COM. Extradural abscesses, subperiosteal abscesses, and lateral sinus thrombosis usually occur by this mechanism. Patients will commonly present
with a prodrome of partial or intermittent impairment of structures affected by the advancing infection, most notably pain, abducens palsy, facial paralysis, and vertigo. Thrombophlebitic spread is more common in acute infections, in acute exacerbations of chronic infection, and in brain abscess formation. A prodromal period is often absent, and intact bony walls are often found at surgery.
with a prodrome of partial or intermittent impairment of structures affected by the advancing infection, most notably pain, abducens palsy, facial paralysis, and vertigo. Thrombophlebitic spread is more common in acute infections, in acute exacerbations of chronic infection, and in brain abscess formation. A prodromal period is often absent, and intact bony walls are often found at surgery.
CLASSIFICATION
Complications of COM are generally grouped into two main categories:
Complications involving the cranial cavity, including the following:
Extradural abscess
Lateral sinus thrombosis
Brain abscess
Meningitis
Subdural infection
Otitic hydrocephalus
Complications involving the temporal bone and extratemporal soft tissues, including the following:
Subperiosteal and Bezold’s abscesses
Facial paralysis
Petrous apicitis
Labyrinthitis
Sensorineural hearing loss
Among the intracranial complications, meningitis is probably the most commonly encountered (3,7,14), although some authors report more cases of brain abscess (15) and epidural abscess (16) in their series. Brain abscess is generally considered to have the highest rate of mortality (6,14). Similar data regarding the relative incidence of temporal bone complications have not been reported.
In approximately one third of cases, two or more otogenic complications are present concomitantly (17), usually reflecting the path of least resistance chosen by the spreading infection. During an exploration for lateral sinus thrombosis, for example, it is not uncommon to also find a perisinus abscess from which it may have originated or a brain abscess to which it may have progressed. It is therefore incumbent upon the otolaryngologist to thoroughly investigate all potential complications prior to the initiation of therapy.
With the rapid spread of human immunodeficiency virus (HIV) and AIDS in the last quarter century, a growing number of immunosuppressed patients have incurred certain complications of COM that were not commonly seen before. The spread of disease in this patient population tends to be more extensive and follow a more fulminant course. In addition, opportunistic pathogens not usually found in the ear and adjacent structures are more likely to flourish in the immunocompromised host. As more and more severely immunocompromised patients live longer, recognition of these rare complications of COM will become increasingly important.
SYMPTOMS AND SIGNS OF IMPENDING COMPLICATION
The presentation of complicated COM is well described by Neely (8) and Snow (18) and has been reviewed by Schwaber et al. (16). Early signs and symptoms include fetid otorrhea, deep pain, and headache. The development of malodorous, purulent discharge from the ear is highly suggestive of bone destruction. It is postulated that the longer the period of drainage, the greater the amount of bone destruction that has occurred (16). Pain is generally not associated with COM but may arise in exacerbations of the disease or with progressive destruction of bone or obstruction of drainage of purulent material from the mastoid space via the aditus. Uncomplicated exacerbations are associated with transient pain, whereas bone erosion or obstruction to drainage causes deep and constant pain. The latter may suggest a developing complication.
The appearance of the tympanic membrane may provide important information about the condition of the middle ear. Although the presence of chronic perforation with associated otorrhea is classically cited as the tympanic membrane feature in COM, other abnormalities may herald middle ear disease. Pathologic correlates between tympanic membrane and middle ear findings in temporal bones with COM reveal that myringosclerosis even without perforation is significantly associated with middle ear granulation tissue and ossicular pathology. Although both retraction pockets and perforations are associated with middle ear disease, the presence of both together have an additive effect on predicting cholesteatoma, ossicular pathology, and cholesterol granuloma. In addition, a significant fraction of middle ear problems may be hidden behind a normal-appearing tympanic membrane, particularly in infants (19). This underscores the need for tests such as tympanometry or imaging in patients with persistent aural symptoms and normal findings on otoscopic examination.
Fever, nausea, and altered mental status may be present at the time that a complication of COM is diagnosed, but generally these symptoms occur later in the course of the infection. Rarer findings at presentation include dysequilibrium, hearing loss, subperiosteal abscess, cranial neuropathies, nuchal rigidity, and seizures and imply that a complication is imminent or has already occurred.
INITIAL MANAGEMENT
A thorough history and physical examination should be performed on all patients suspected of having a complication of COM. Symptoms of headache, neck pain, and visual changes suggest intracranial involvement, and patients with signs of papilledema, impaired extraocular movement, nuchal rigidity, or hemiparesis warrant immediate neurosurgical evaluation. When intracranial extension is uncertain, lumbar puncture may be helpful in arriving at a diagnosis. All patients suspected of having intracranial disease should undergo a period of observation by qualified neurosurgical personnel.
Computed tomography (CT) scanning with intravenous contrast is the imaging modality of choice in screening for complications of COM. Magnetic resonance imaging (MRI) and MR angiography (MRA) are useful in the definitive diagnosis of certain of these complications. However, CT is almost uniformly positive in cases of intracranial extension and gives the most accurate assessment of bone involvement. CT scanning is therefore an indispensable part of the evaluation of all patients suspected of having otogenic complications.
Intravenous antibiotic therapy should be started as soon as possible. Polymicrobial infection is common in COM and its complications, and broad-spectrum coverage is indicated. Multiple drug therapy may be necessary to accomplish this goal. Traditionally, the penicillins, in combination with stronger anaerobic coverage such as metronidazole, were considered standard primary therapy. However, the high incidence of Pseudomonas in these infections, as well as the increasing resistance of gram-positive organisms to penicillins, have prompted the use of alternative agents. Chloramphenicol has often been used and provides exceptional broad-spectrum coverage but has numerous side effects. Its use may be most appropriate in complications of acute otitis media in which Haemophilus influenzae is the likely causative organism. Ticarcillin/clavulanic acid provides excellent coverage for the organisms most commonly implicated in these infections (including Pseudomonas) and achieves reasonable cerebrospinal fluid (CSF) levels in the presence of meningeal inflammation (20). Ceftazidime, a third-generation cephalosporin with enhanced efficacy against Pseudomonas, may be useful in combination with anaerobic coverage. Ciprofloxacin, a fluoroquinolone, is also excellent against Pseudomonas and has the added benefit of having similar circulating concentrations in the oral and parenteral routes.
In general, it is desirable to delay surgical treatment of the infected ear until the patient is neurologically and systemically stable. However, judicious surgical intervention at the first signs of clinical deterioration and progression of infection may be life-saving.
INTRACRANIAL COMPLICATIONS
Extradural Abscess
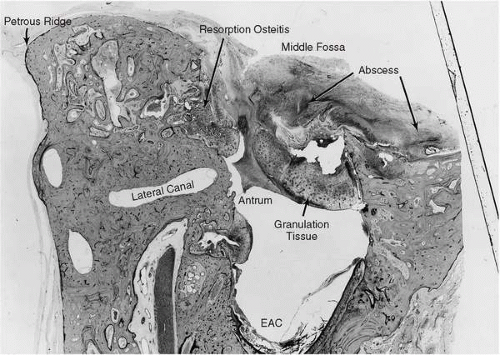
Cholesteatoma and granulation tissue often complicate chronic suppurative otitis media and have a propensity to erode adjacent bony structures. When these processes reach the periphery of the mastoid space, erosion often proceeds until dura is exposed but is then impeded by this relatively resistant structure. The result is an inflammation of the dura known as pachymeningitis, which is often followed by the production of granulations along the dura. The accumulation of purulent material in this area results in a collection of pus between the dura and the bony wall known as an extradural or epidural abscess (21) (Fig. 18.2).
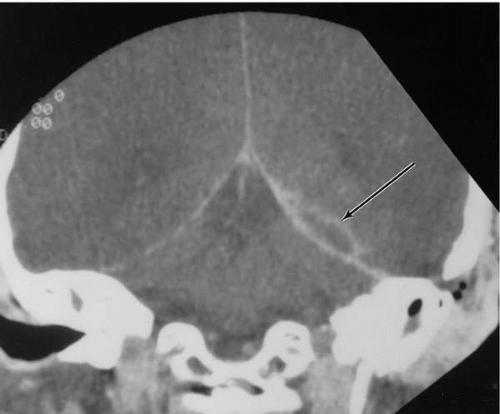
Commonly, resorption of bone will occur in the region of the tegmen, resulting in an extradural abscess of the middle cranial fossa (Fig. 18.3). The infectious process in such cases begins lateral to the attachment of the dura at the arcuate eminence, which serves to limit the extent of the abscess. An abscess developing medial to the arcuate eminence may abut the petrous apex, involving the fifth and sixth cranial nerves and resulting in facial pain and diplopia (Gradenigo’s syndrome). In the middle fossa, lateral to the arcuate eminence, the dura is loosely attached and large abscesses may develop, accompanied by neurologic signs of mass effect. Lateral erosion through the cranium produces the subperiosteal abscess known as Pott’s puffy tumor. Posterior fossa extradural abscesses usually result from the erosion of bone in Trautmann’s triangle or over the lateral sinus. The latter, known as a perisinus abscess, is often associated with the development of lateral sinus thrombosis. Untreated posterior fossa abscesses may also extend to the neck via the jugular foramen.
Symptoms of extradural abscess may include temporal headache, ear pain, or low-grade fever, but often both physician and patient become aware of its presence only after exposure during elective mastoidectomy. Extradural abscesses of long standing are an exception to this rule and are often accompanied by mild nuchal rigidity and meningismus. Characteristically, when symptoms are present, they are suddenly relieved during periods of spontaneous drainage into the mastoid. Exacerbation of the aural discharge on compression of the ipsilateral jugular vein is also highly suggestive. Lumbar puncture may reveal an increased white cell count, but usually with lymphocyte predominance. Glucose levels are normal, and no organisms are seen. When extradural abscess is suspected prior to surgery, CT scanning is preferred for confirmation of the diagnosis.
Treatment
Surgical intervention is indicated whenever physical examination or CT scan is suggestive of extradural abscess; antimicrobial therapy will not effectively penetrate the walls of the abscess. Mastoidectomy is performed, and the tegmen and sinus plates are removed. Loose granulation tissue is removed, and a border of normal dura is exposed peripherally around the affected site. Cautious manipulation of adherent granulations is necessary to avoid violation of the dura. Cholesteatoma matrix covering a dural exposure may be left behind if well exteriorized. Removal of cholesteatoma is otherwise completed, and a Penrose drain is placed into the mastoid cavity at the end of the procedure. Culture-specific systemic and topical postoperative antibiotics help to prevent the spread of residual organisms throughout the tissues and the incised auricular cartilage and help to prevent postoperative wound breakdown and perichondritis. A 6-week course of antibiotic therapy is generally recommended in cases where a diagnosis or suspicion of osteomyelitis is made.
Lateral Sinus Thrombosis
Extension of mastoid cavity disease to adjacent venous sinuses may occur through direct extension through the dura or by spread along mastoid or middle emissary veins. The transverse sinus, which begins at the internal occipital protuberance, exits the tentorium as the sigmoid sinus. The sigmoid is joined by the superior and inferior petrosal sinuses before leaving the cranium as the internal jugular vein. The lateral sinus is the segment of the transverse-sigmoid complex immediately beneath the mastoid air cells and is the region most susceptible to thrombosis or thrombophlebitis due to mastoid disease (Fig. 18.4). However, otogenic involvement of other dural sinuses has been reported (22).
Septic lateral sinus thrombosis was first described in the mid-nineteenth century and carried a mortality rate of 100% until the first successful surgical intervention by Lane in 1888 (cited by Tveteras et al. [23]). With improvement in surgical technique, this rate fell to less than 30% to 50% by the early 1900s (14,24). Mortality in the antibiotic era remains in the range of 10% to 36% (14,22,25,26). Thus lateral sinus thrombosis continues to be a significant and lethal complication of COM.
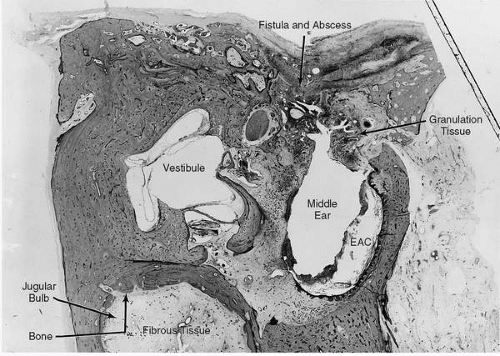 FIG. 18.4 Same case as Fig. 18.2. Left ear, vertical section. Granulation tissue in the middle ear extends superiorly through a large defect in the tegmen into the middle cranial fossa. There is fibrocystic obliteration of the oval and round window niches. Fibro-osseous obliteration of the jugular bulb is evidence of chronic healed phlebitis of the jugular vein. 6.6X. |
Thrombosis of the dural sinuses usually begins as a result of damage to the tunica intima by an adjacent inflammatory process, most commonly an extradural perisinus abscess. Damage to the vessel wall initiates fibrin formation and platelet aggregation. This process is enhanced by the thrombogenic capacity of the neighboring bacteria (27). Classically, the thrombus becomes infected by spread of the abscess through the dura and vessel wall. However, it has also been suggested that seeding occurs primarily due to thrombophlebitic spread from the pneumatized spaces and that dural inflammation and fibrosis due to perisinus abscess actually serve to improve resistance to direct spread of infection (18). Thrombus may then propagate along vessel walls, causing local invasion, or it may produce bacteremia, septicemia, or septic embolization. Involvement of the cerebrum, meninges, and cerebellum has been reported in 45% of patients with lateral sinus thrombosis (28).
Recent publications indicate an evolving microbiology in this disorder. Once dominated by beta-hemolytic Streptococcus and Pneumococcus, cultures now reflect the flora of COM, revealing multiple pathogens, including Proteus sp., Staphylococcus aureus, Bacteroides sp., and Escherichia coli, as well as aerobic and anaerobic streptococci (17,22,23). These results suggest a higher incidence of dural sinus thrombosis in chronic ear disease (17,22,23,25), although in one recent series acute ear disease was the more common etiology (3).
The use of aggressive antibiotic therapy has altered the presentation of this disease to one of a less acute nature. The classical high, spiking “picket fence” fever and rigor are now rarely seen. Cervical or orbital extension of the clot, resulting in paralysis of cranial nerves or proptosis of the eye, is now also an unusual finding.
Typically, patients present after approximately 2 weeks of symptoms with headache that is usually lateral (17,22). The headache is often preceded by ear pain or discharge on the ipsilateral side and may be accompanied by nausea and vomiting. Visual changes, nuchal rigidity, and dysequilibrium are rare complaints.
Physical examination will almost always reveal purulent drainage through a perforated tympanic membrane. Pitting edema and tenderness over the postauricular area due to involvement of the mastoid emissary vein (Griesinger’s sign) may be present. Fever remains a prominent feature in most patients, and papilledema due to increased intracranial pressure is present in more than 50% of cases (17,22). Less commonly the clinician may note palsy of the abducens nerve, believed to result from compression of the nerve by a distended inferior petrosal sinus within the closed space of Dorello’s canal (Gradenigo’s syndrome) (22). Nuchal rigidity and nystagmus are rarer findings.
Routine blood work usually reveals an elevated white cell count with a left shift and an elevated erythrocyte sedimentation rate. Blood cultures are usually negative. Lumbar
puncture should be performed unless uncal herniation is likely to result from the procedure. Commonly, an elevated CSF pressure is noted, probably due to impaired CSF reabsorption through the arachnoid granulations. Cerebrospinal fluid chemistry and cell counts are usually unremarkable. A positive Queckenstedt’s or Tobey-Ayer test, once considered pathognomonic of the disorder, has been found to be unreliable and to elevate intracranial pressure to dangerous levels (17). The test is considered positive when compression of the internal jugular vein on the side of the thrombosis fails to raise CSF pressure, but compression of the contralateral vein results in a rapid and marked increase in pressure.
puncture should be performed unless uncal herniation is likely to result from the procedure. Commonly, an elevated CSF pressure is noted, probably due to impaired CSF reabsorption through the arachnoid granulations. Cerebrospinal fluid chemistry and cell counts are usually unremarkable. A positive Queckenstedt’s or Tobey-Ayer test, once considered pathognomonic of the disorder, has been found to be unreliable and to elevate intracranial pressure to dangerous levels (17). The test is considered positive when compression of the internal jugular vein on the side of the thrombosis fails to raise CSF pressure, but compression of the contralateral vein results in a rapid and marked increase in pressure.
Definitive diagnosis of dural venous thrombosis is now based primarily on radiographic studies. Cerebral angiography, the gold standard study, is particularly effective in delineating the cerebral venous anatomy, as well as in demonstrating the presence of vascular occlusion. Traditionally a venous phase arteriogram is performed, and the study may be enhanced by subtraction techniques. More recently, subtraction venography has been employed to avoid the risks associated with arteriography (17).
CT provides a less invasive examination of the dural sinuses and is useful in screening for concomitant otogenic complications. Increased density of the sinus on noncontrast scans suggests the presence of fresh clot, whereas contrast enhancement reveals filling defects and highlights inflamed sinus walls and dura. Soft tissue changes such as cerebral edema, decreased ventricular size, and hemorrhage or abscess may be detected. CT is unquestionably the method of choice for assessment of bony changes.
MRI and MRA have emerged as highly accurate modalities in the diagnosis of venous thrombosis (18,29, 30, 31, 32). The defect is determined by the absence of flow-induced signal loss (29). These modalities have the additional advantage of reconstruction from several angles of view.
Treatment
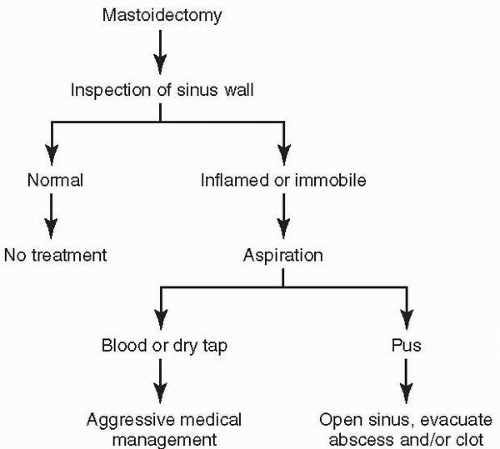
Modern treatment of lateral sinus thrombosis is based on controlling infection with minimal surgical debridement and intensive antibiotic therapy. When thrombosis of the sinus is suspected, intravenous antibiotics should be started to minimize hematogenous spread. Until specific organisms and sensitivities are isolated, empiric therapy should target Staphylococcus, aerobic and anaerobic Streptococcus, and upper respiratory anaerobes. Historically, a combination of penicillin and chloramphenicol was often used. Given new understanding of relative drug penetration of the blood-brain barrier, it has been recommended to start with a combination of intravenous penicillin or nafcillin, intravenous ceftriaxone, and oral or intravenous metronidazole (25). Surgical intervention is directed by radiographic studies. Mastoidectomy is performed with wide exposure of the sigmoid sinus. Bone is removed until normal dura is exposed peripherally around the region of suspected thrombosis. Dural granulations are carefully removed and the sinus wall is inspected.
A normal-appearing, compressible sinus wall requires no further surgical treatment. If the sinus wall is inflamed or immobile on palpation, however, evaluation of the sinus lumen is indicated. A small-gauge needle may be used to aspirate the sinus, and, if bleeding is encountered, no further surgical treatment is necessary. Failure to obtain blood on aspiration suggests thrombosis, and aspiration of pus indicates infection of the thrombus. The discovery of pus upon aspiration requires drainage of the collection. Under these circumstances, evacuation of the infected clot should be performed. Control is gained distally and proximally within the mastoid cavity by extraluminal compression using absorbable sponges between the sinus wall and the overlying bone. An incision is then made in the lateral sinus wall in the direction of the vessel. If blood is obtained, the incision may be simply covered with absorbable sponge or temporalis muscle. If infected thrombus is noted, it is removed until only healthy clot or bleeding is encountered.
The patient is maintained on intravenous antibiotics. Ligation of the internal jugular vein, once popular in the management of lateral sinus thrombosis, is now reserved only for cases that are unresponsive to initial surgical and antibiotic therapy or those in which septic emboli are developing (14,26). Anticoagulation is still used by many clinicians to prevent extension of the thrombus and to reduce the risk of thrombosis of the cavernous sinus (30). Use of anticoagulant therapy, however, incurs the risks of septic embolization and hemorrhage due to clot breakdown (14). Also controversial is the dry tap of the sinus, which may be managed conservatively by aggressive antibiotic therapy or by opening the sinus and evacuating the clot (Fig. 18.5).
Brain Abscess
Abscess formation within the parenchyma of the brain, once considered the cause of suppurative otorrhea, has been
recognized as a sequela of otitis media since the time of Morgagni (1682-1771, cited by Glasscock and Shambaugh [7]). Its successful treatment was first described by Morand in 1768 (1) and advanced by numerous nineteenth century surgeons, most notably MacEwen (31), who reported favorable outcomes in 18 of 19 operated cases of brain abscess. Treatment by aspiration through a bur hole was described by Dandy in 1926, and complete excision of an abscess was reported by Vincent in 1936 (cited by Haines et al. [32]).
recognized as a sequela of otitis media since the time of Morgagni (1682-1771, cited by Glasscock and Shambaugh [7]). Its successful treatment was first described by Morand in 1768 (1) and advanced by numerous nineteenth century surgeons, most notably MacEwen (31), who reported favorable outcomes in 18 of 19 operated cases of brain abscess. Treatment by aspiration through a bur hole was described by Dandy in 1926, and complete excision of an abscess was reported by Vincent in 1936 (cited by Haines et al. [32]).
For many years brain abscess was most commonly associated with otitic infection, and its management lay primarily with the otologist. In 1931 Evans (33) attributed 56% of brain abscesses at autopsy to otitis media, and in 1955 Courville (4) reported an otitic etiology in 46% of these cases. The development of antibiotics during the early part of the twentieth century, combined with improved neurosurgical management of early intracranial complications, has led to a dramatic decline in the incidence of otogenic brain abscess. In 1969 Beekhuis and Taylor (34) reported an association with otitis media in just 12.8% of all cases of brain abscess treated during the years 1956 to 1965. Bradley et al. (35) more recently reviewed cases of otogenic brain abscess at three hospitals between 1950 and 1980 and found an incidence of 25%.
Despite the decreased incidence of otogenic brain abscess, the mortality of the disorder remains high. Death rates in the literature of the last 30 years range from 14% to 47% (14,34,35), making brain abscess the deadliest complication of otitis media. It is therefore essential that the otologist consider the possibility of brain abscess when treating any otogenic complication.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree