Purpose
To perform a comparative cost-effectiveness analysis of Descemet stripping automated endothelial keratoplasty (DSAEK) and penetrating keratoplasty (PK) for corneal endothelial disease.
Design
Retrospective cost-effectiveness analysis.
Methods
This cost-effectiveness analysis was performed from a third-party payer perspective with a 5-year time horizon. Probabilities of outcomes and complications of each of the procedures were calculated based on review of the published literature. A model was constructed to compare the costs and utilities associated with DSAEK and PK. Costs of donor tissue preparation, surgery, follow-up, postoperative complications, and procedures were considered. Utility values were based on quality-adjusted life years associated with visual acuity outcomes. Both costs and utilities were discounted at 3% per year. Sensitivity analyses were performed on key model inputs.
Results
Base case analysis found DSAEK to be less costly compared with PK ($9362 vs $10 239), with greater utility (3.15 vs 2.47 quality-adjusted life years). Sensitivity analyses revealed that even at graft failure rates for DSAEK approaching the rates for PK, DSAEK would still reduce costs. Varying the dislocation rate in our model showed that even at dislocation rates approaching 50%, DSAEK remained less costly. Further, with DSAEK rejection rates as high as 28%, DSAEK would remain a dominant procedure over PK.
Conclusions
Comparative cost-effectiveness analysis of DSAEK versus PK indicates favorable cost and utility outcomes associated with DSAEK for treatment of corneal endothelial disease. Longer follow-up of DSAEK outcomes will provide more accurate information regarding long-term cost-effectiveness of the procedure.
Penetrating keratoplasty (PK) long has been the definitive treatment for patients with corneal endothelial dysfunction, namely Fuchs corneal dystrophy, pseudophakic bullous keratopathy, and aphakic bullous keratopathy associated with visually significant corneal edema. Although PK offers patients the chance for markedly improved vision, it is associated with complications including prolonged visual recovery, induced regular and irregular astigmatism, suture-related problems, and late wound dehiscence.
Posterior lamellar keratoplasty techniques have gained increasing acceptance over the past decade. Initially described by Melles and associates in 1998, there have been multiple technique modifications, culminating in Descemet stripping automated endothelial keratoplasty (DSAEK). DSAEK involves removal of the diseased endothelium and Descemet membrane and subsequent replacement with a posterior corneal lamella prepared by automated microkeratome dissection. These technique refinements allowed endothelial keratoplasty to gain widespread acceptance for the treatment of endothelial diseases. In 2011, 21 555 endothelial procedures were performed, accounting for 47% of all corneal transplant surgeries in the United States. The selective transplantation of the endothelium through a small incision allows for speedier visual rehabilitation with lower rates of astigmatism and other suture-related complications. However, there are also complications unique to DSAEK, including difficulties in preparation of the donor tissue, graft adherence, and higher rates of iatrogenic graft failure.
Despite the rapid growth in DSAEK surgery, little is known about the cost-effectiveness of DSAEK compared with PK. Beauchemin and associates evaluated the cost-effectiveness of posterior lamellar surgeries and PK in Canada using a Canadian healthcare system perspective in a hypothetical cohort of 100 patients. The study found that posterior lamellar keratoplasty is less costly (−$688/patient) and provides more utility (+0.13 quality-adjusted life years [QALYs]/patient) than PK. In this study, a cost-effectiveness analysis was performed comparing DSAEK with PK for treatment of corneal endothelial disease in adults in the United States.
Methods
Visual Acuity Outcomes and Complication Rates Source Data
The peer-reviewed English literature was searched in PubMed in March 2011, without date restriction. Keywords in the search were Descemet’s stripping endothelial keratoplasty, Descemet stripping endothelial keratoplasty, Descemet’s stripping automated endothelial keratoplasty, Descemet stripping automated endothelial keratoplasty , and penetrating keratoplasty .
The abstracts of the 5554 resulting citations were reviewed, and the 114 citations that involved studies of possible clinical relevance were reviewed in full text. Citations that pertained to endothelial keratoplasty procedures other than DSAEK or Descemet stripping endothelial keratoplasty or to PK performed for reasons other than endothelial disease were excluded. Abstracts of meeting presentations also were excluded.
Studies with fewer than 30 eyes undergoing DSAEK were excluded to focus on studies with statistical analysis and not just case reports. Given the extensive literature on PK, studies with fewer than 200 eyes were excluded. For multiple studies that used the same database of patients for analysis, the study with the largest number of patients was included to capture the most inclusive group, while preventing duplicate counting of outcomes in the same patient cohorts.
Publications included were large case series (> 30 eyes for DSAEK and > 200 eyes for PK), observational studies, and randomized controlled trials on DSAEK or PK for corneal endothelial disease with results on outcomes, adverse events, or complications. For the purposes of this analysis, outcomes data regarding Descemet stripping endothelial keratoplasty and DSAEK were combined. Studies that analyzed DSAEK or PK with concurrent cataract extraction and intraocular lens placement also were included, because similar outcomes have been shown for these procedures. A total of 32 studies were determined to be relevant to the assessment objective.
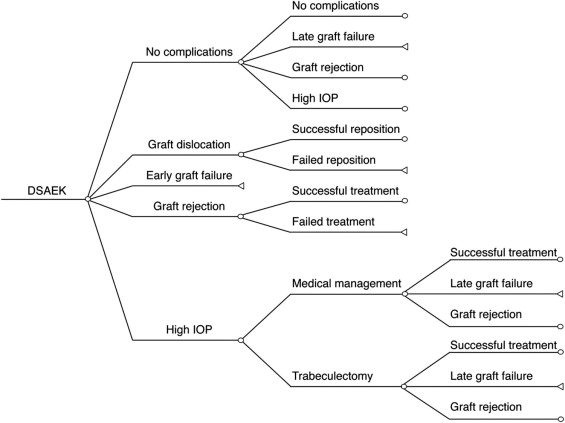
With the above-detailed inclusion and exclusion criteria, no studies were identified that presented data on rates of treatment for elevated intraocular pressure in patients undergoing PK for endothelial disease. Targeted search of the literature revealed 2 articles that offered information regarding treatment of elevated intraocular pressure after PK for other corneal conditions. These studies were incorporated in this analysis to provide insight regarding treatment of elevated intraocular pressure occurring after PK. Complication rates ( Table 1 ) used in our model were based on the average, weighted by number of patients, of those found in the 32 studies included in this analysis.
| Complication | DSAEK Probability (%) | PK Probability (%) | References |
|---|---|---|---|
| Graft failure in year 1 (not associated with dislocation or rejection) | 3.3 | 4.5 | |
| Graft dislocation | 16.1 | — | |
| Successful repositioning | 81.9 | — | |
| Graft failure | 18.1 | — | |
| Annual graft rejection | 5.9 | 10.6 | |
| Successful treatment | 91.0 | 61.3 | |
| Graft failure after treatment | 9.0 | 38.7 | |
| High IOP | 15.0 | 19.0 | |
| Requiring medical treatment | 99.3 | 85.2 | |
| Requiring surgical treatment | 0.7 | 14.8 | |
| Astigmatism >5 D | — | 29.0 | |
| Average annual late graft failure (years 2 through 5) | 1.4 | 3.8 |
Conversion of Visual Acuity to Utilities
Utility values were based on visual acuity, as has been described in previously published studies of patients with ophthalmic disease, and were derived from patient preferences in a time trade-off model. Average preoperative and postoperative best-corrected visual acuities (BCVAs) for DSAEK and PK were obtained through our systematic review of the literature and were converted to utility values using a previously published scale that relates Snellen visual acuity to utility values ( Table 2 ).
| Surgical State | BCVA | Associated Utility Value | References |
|---|---|---|---|
| DSAEK | |||
| Preoperative | 20/90 | 0.7 | |
| Postoperative | 20/40 | 0.8 | |
| PK | |||
| Preoperative | 20/120 | 0.67 | |
| Postoperative | 20/40 | 0.8 |
Identification of Costs
Services billable to third-party payers associated with surgery, patient follow-up, postoperative complications, and medications were identified through a review of patient bills for each type of surgery.
For the base case model, assumptions based on clinical practice were made regarding the number of follow-up visits. For DSAEK, the model we used included 2 follow-up visits in the first year outside the 90-day postoperative global period; for PK, 5 follow-up visits were included. We assumed more follow-up visits for PK in the first year after surgery given the need for sequential suture removal in many patients. The complexity of these visits was assumed to be at the level of an Evaluation and Management (E/M) office visit code 99213. We also assumed that in the first year after PK, topography would be obtained an average of 3 times outside the global period to aid in suture removal for astigmatism. Glaucoma specialist consultations were assumed to be at an E/M office visit level 99203. The assumed cost of rigid gas permeable lens for high astigmatism included an office visit for fitting, the estimated cost of a lens, and annual cost of cleaning solutions. In the base model, rebubble procedures were assumed to be performed in the operating room using Current Procedural Terminology-4 code 66020. Costs were applied to each service using 2010 Medicare Fee Schedules for physician and ambulatory facility services, the 2010 Drug Topics Red Book for medications, and personal communication with the Midwest Eye Banks (Ann Arbor, Michigan, USA) for an estimate of eye bank fees ( Table 3 ).
| Service | CPT or E/M Code | Costs ($) |
|---|---|---|
| Initial DSAEK | ||
| Eye bank fee | 3600 | |
| Surgeon’s fee | 65756 | 1168 |
| Ambulatory surgical center fee | 0244 | 2530 |
| Anesthesia fees | 144 | 237 |
| Medications | ||
| Prednisolone acetate 1% | 203 | |
| Moxifloxacin 0.5% | 82 | |
| Follow-up visits (×2) | 99213 | 104 |
| Total, 7924 | ||
| Initial PK | ||
| Eye bank fee | 2900 | |
| Surgeon’s fee | 65730 | 1231 |
| Ambulatory surgical center fee | 0244 | 2533 |
| Anesthesia fees | 144 | 237 |
| Medications | ||
| Prednisolone acetate 1% | 203 | |
| Moxifloxacin 0.5% | 82 | |
| Follow-up visits (×5) | 99213 | 260 |
| Topography (×3) | 92025 | 106 |
| Total, 7550 | ||
| Complications | ||
| Repeat DSAEK | 7924 | |
| Repeat PK | 7396 | |
| Graft dislocation (rebubbling) | 1459 | |
| Surgeon’s fee | 66020 | 127 |
| Ambulatory surgical center fee | 0233 | 1095 |
| Anesthesia fees | 144 | 237 |
| Graft rejection treatment | 307 | |
| Prednisolone acetate 1% | 203 | |
| Additional follow-up (×2) | 99213 | 104 |
| High IOP: medical management | 266 | |
| Timolol (0.5%) | 162 | |
| Additional follow-up visits (×2) | 99213 | 104 |
| High IOP: surgical management (trabeculectomy) | 4901 | |
| Glaucoma specialist initial visit | 99203 | 78 |
| Trabeculectomy surgeon’s fee | 66172 | 1497 |
| Ambulatory surgical center fee | 673 | 2823 |
| Anesthesia fees | 144 | 237 |
| Medications | 162 | |
| Follow-up visits (×2) | 99213 | 104 |
| Astigmatism > 5 D | 344 | |
| Lens fitting | 99203 | 78 |
| Rigid gas permeable lens | 150 | |
| Lens cleaning | 116 |
Model Design
This analysis was conducted from a third-party payer perspective, specifically Medicare in 2010. A decision tree with a 5-year time horizon was constructed to assess the costs per QALY associated with DSAEK compared with PK. Portions of the first 2 years of the decision tree are presented in Figures 1 and 2 . The probabilities, utilities, and costs associated with each event (or branch) were incorporated into the decision tree to derive the QALYs and costs for each procedure. Microscoft Excel (Microsoft, Redmond, Washington, USA) was used to conduct the analysis. Both costs and QALYs were discounted at 3% per year.