Purpose
To identify challenges and pose solutions for communications about ocular gene therapy between patients and clinicians as clinical research progresses.
Design
Literature review with recommendations.
Methods
Literature review of science communication best practices to inform recommendations for patient-clinician discussions about ocular gene therapy.
Results
Clinicians need to employ communications about ocular gene therapy that are both attentive to patient priorities and concerns and responsive to other sources of information, including overly positive news media and the Internet. Coverage often conflates research with therapy—clinical trials are experimental and are not risk free. If proven safe and efficacious, gene therapy may present a treatment but not a cure for patients who have already experienced vision loss. Clinicians can assist patients by providing realistic estimates for lengthy clinical development timelines and positioning current research within models of clinical translation. This enables patients to weigh future therapeutic options when making current disease management decisions.
Conclusions
Ocular gene therapy clinical trials are raising hopes for treating a myriad of hereditary retinopathies, but most such therapies are many years in the future. Clinicians should be prepared to counter overly positive messaging, found in news media and on the Internet, with optimism tempered by evidence to support the ethical translation of gene therapy and other novel biotherapeutics.
Recent successes in ocular gene therapy clinical trials have revitalized hopes for a field that was previously marked by high-profile disappointments. Pioneering clinical trials to address mutations of the RPE65 gene that cause Leber congenital amaurosis (LCA) established safety and demonstrated improvements in visual function. These studies were celebrated by the research community and the media, igniting hopes for gene therapies for related retinopathies. Our goal here is to identify challenges and pose solutions for communications about ocular gene therapy between patients and clinicians as clinical research progresses. Although we focus on gene therapy, similar considerations apply to other experimental biotherapeutics.
The discussion is timely. One research group at the University of Pennsylvania reports that positive safety and efficacy results from the LCA trials have been sustained over time in adults and children ; readministration in the second eye of 3 adult patients was both safe and efficacious after previous exposure to the vector. A fully enrolled phase III pediatric clinical trial is underway and expected to report in late 2015 ( NCT00999609 ). The pediatric trial is crucial because LCA studies suggest a therapeutic window for visual gain, with earlier application leading to more dramatic responses.
Although these results appear promising, studies in dogs suggest that degenerative processes may continue after the gene therapy intervention, if such degeneration has already commenced. In humans, a second research group reported that gene therapy improves vision for at least 3 years, but photoreceptor degeneration also continues at the same rate as in the natural course of the disease. Long-term follow-up (4.5-6 years) from 3 treated patients indicated progressive diminution of the areas of improved vision. Similar results were reported by a team from the United Kingdom: retinal sensitivity improved after gene therapy but diminished after 12 months. Gene therapy may therefore not offer a permanent treatment, and most benefit is likely if the intervention occurs prior to the onset of retinal degeneration. Some patients may require a second round of gene therapy, and gene therapy may best be used in combination with other medications, if and when these are developed. Research continues to improve gene therapy vectors and surgical delivery to ensure that patients receive an optimal dose for long-lasting effects.
The LCA trials have paved the way for other retinopathies ( Table ). Public and private investments now support companies created to advance clinical development of ocular gene therapy. Spark Therapeutics has licensed technologies from the Children’s Hospital of Philadelphia, including those designed for LCA and choroideremia ( http://www.sparktx.com/ ). NightstaRx was similarly launched in the United Kingdom to develop a gene therapy product for choroideremia ( http://www.nightstarx.com/ ). Spin-off companies such as these enable scale-up from clinical trials to gene therapies available to patients in the clinic. However, potential financial conflicts of interest for researchers conducting clinical trials and involved in sponsoring companies must be managed carefully. Clinical trial participants must be informed of any conflicts of interest during consent processes to ensure the integrity of the research. Lessons may be taken from the early days of gene therapy clinical trials. Financial conflicts of interest, along with improper informed consent and protocol violations, damaged the reputation and progress of the field following the death of Jesse Gelsinger in a gene therapy clinical trial at the University of Pennsylvania in 1999.
| Disease | Intervention | Phase | Enrollment | Age Group | Start Year – End Year | Status | Sponsor | Clinical Trial Identifier |
|---|---|---|---|---|---|---|---|---|
| Choroideremia | AAV2-hCHM | I/II | 10 | A,S | 2015–2021 | R | Spark Therapeutics a | NCT02341807 |
| rAAV2 REP1 b | II | 30 | A,S | 2015–2018 | NYR | University of Oxford | NCT02407678 | |
| rAAV2.REP1 b | I/II | 12 | A,S | 2011–2015 | R | University of Oxford | NCT01461213 c | |
| rAAV2.REP1 b | I | 6 | A,S | 2015–2018 | R | University of Alberta | NCT02077361 | |
| Leber congenital amaurosis | AAV2-hRPE65v2 | III | 24 | C,A,S | 2012–2029 | ONR | Spark Therapeutics a | NCT00999609 |
| AAV2-hRPE65v2 | I/II | 12 | C,A,S | 2010–2026 | ONR | Spark Therapeutics a | NCT01208389 | |
| AAV2-hRPE65v2 | I | 12 | C,A,S | 2007–2024 | ONR | Spark Therapeutics a | NCT00516477 d | |
| rAAV2-CB-hRPE65 | I/II | 12 | C,A,S | 2008–2027 | ONR | Applied Genetic Technologies a | NCT00749957 | |
| rAAV 2/2.hRPE65p.hRPE65 | I/II | 12 | C,A | 2007–N/A | ONR | University College, London | NCT00643747 e | |
| rAAV2-CBSB-hRPE65 | I | 15 | C,A,S | 2007–2026 | ONR | University of Pennsylvania | NCT00481546 f | |
| rAAV2/4.hRPE65 | I/II | 9 | C,A | 2011–2014 | Com | Nantes University Hospital | NCT01496040 | |
| rAAV2-hRPE65 | I | 10 | C,A,S | 2010–2017 | R | Hadassah Medical Organization | NCT00821340 | |
| Leber hereditary optic neuropathy | scAAV2-P1ND4v2 | I | 27 | A | 2014–2019 | R | John Guy, University of Miami | NCT02161380 |
| rAAV2-ND4 | N/A | 6 | C,A | 2011–2013 | R | Bin Li, Huazhong University of Science and Technology | NCT01267422 | |
| Neovascular age-related macular degeneration | rAAV.sFlt-1 | I/II | 40 | A,S | 2011–2015 | ONR | Lions Eye Institute, Australia | NCT01494805 |
| AAV2-sFLT01 | I | 34 | A,S | 2010–2018 | ONR | Genzyme, a Sanofi Co a | NCT01024998 | |
| RetinoStat | I | 21 | A,S | 2011–2015 | ONR | Oxford BioMedica a | NCT01301443 | |
| RetinoStat | I | 21 | A,S | 2012–2027 | R | Oxford BioMedica a | NCT01678872 | |
| AdGVPEDF.11D | I | N/A | A,S | N/A | Com | GenVec a | NCT00109499 | |
| Retinitis pigmentosa | rAAV2-VMD2-hMERTK | I | 6 | C,A,S | 2011–2023 | R | Fowzan Alkuraya, King Khaled Eye Specialist Hospital | NCT01482195 |
| Retinoschisis | rAAV2tYF-CB-hRS1 | I/II | 27 | C,A,S | 2015–2020 | NYR | Applied Genetic Technologies a | NCT02416622 |
| AAV-RS1 | I/II | 100 | A,S | 2014–2017 | R | National Eye Institute | NCT02317887 | |
| Stargardt macular degeneration | StarGen | I/II | 46 | C,A,S | 2011–2017 | R | Sanofi a | NCT01367444 |
| StarGen | I/II | 28 | A,S | 2012–2022 | R | Sanofi a | NCT01736592 | |
| Usher syndrome | UshStat | I/II | 18 | A,S | 2012–2017 | R | Sanofi a | NCT01505062 g |
| UshStat | I/II | 18 | A,S | 2013–2022 | R | Sanofi a | NCT02065011 |
b NightstaRx AAV2-REP1 product.
c Results published by Seitz et al (PubMed ID 25744334 ) and MacLaren et al (PubMed ID: 24439297 ).
d Results published by Melillo et al (PubMed ID: 22812667 ), Maguire et al (PubMed ID: 19854499 ), and Maguire et al (PubMed ID: 18441370 ).
e Results published by Bainbridge et al (PubMed ID: 25938638 ) and Bainbridge et al (PubMed ID: 18441371 ).
f Results published by Jacobson et al (PubMed ID: 25936984 ), Cideciyan et al (PubMed ID: 25537204 ), and Jacobson et al (PubMed ID: 21911650 ).
g Results published by Zallocchi et al (PubMed ID: 24705452 ).
Against this backdrop, patients and their families are increasingly likely to request information from clinicians about emerging therapies. Patients may ask about potential treatment options or about articles on experimental interventions that they have retrieved from external sources; patients may inquire about clinical trials and about the possibility of enrolling in such trials; and patients may ask about early or compassionate access programs that are available in some countries. These programs allow health care providers to apply for the use of investigational products such as unapproved drugs or devices that are still in clinical trial or market approval stages. To be eligible for such access programs, patients must have serious or life-threatening conditions, have no alternative therapy options, and have no other product access option (including clinical trial participation), and the potential benefits of the product must outweigh the risks. This risk-vs-benefit ratio is difficult to characterize for products that are in early phases of clinical trials.
A Communications Landscape of Promise
Communications about cutting-edge research will not occur in an informational vacuum. When discussing research with patients, clinicians need to be attentive to patient priorities and concerns but also responsive to other sources of information. These sources include news media, patient advocacy organizations, industry, social media, health and research organization websites, and other Internet sources, as well as family and friends. Forty percent of the American public follow health news stories closely, especially those that are personally relevant ; Internet access to health information is widespread and growing.
Media coverage of genetic research, particularly clinical research, is highly positive, framed as a celebration of progress in research. News articles focus on benefits, while omitting or downplaying risks, and provide limited background information for readers to assess the validity or credibility of the research (including funding sources and potential conflicts of interest). They accelerate timelines by presenting preclinical or early-stage research as an imminent therapy or cure. News articles often contextualize research with a human-interest story focused on a hopeful patient or a heroic researcher. Media coverage of ocular gene therapy clearly follows this pattern. It describes a range of visual outcomes from slowing vision loss to a cure, even though the latter is clinically infeasible. This positive coverage of ocular gene therapy generates high expectations in patient communities. For example, after the highly publicized successes in early-phase LCA trials, clinical reports documented a trend of LCA patients and families wishing to access clinical trials. Less publicized was the caveat that the initial LCA trials addressed only 1 of 17 known genetic mutations underlying the disease, representing only 5% of LCA cases.
Patient organizations, such as the Foundation Fighting Blindness ( www.ffb.ca ) and the Choroideremia Research Foundation ( cureCHM.org ), also contribute to the communications landscape. These organizations both fund innovative research and communicate with patients and the public about associated risks, benefits, and timelines. Featured prominently on these sites are current and planned gene therapy clinical trials. The Foundation Fighting Blindness provides up-to-date information on clinical trials and genetic testing resources and maintains a patient registry as a tool to connect individuals living with inherited eye diseases to scientists researching those conditions. The Choroideremia Research Foundation operates similarly, and has recently adopted the Foundation Fighting Blindness MyRetinaTracker ( https://www.myretinatracker.org ), a registry that allows patients to upload their genotypic, phenotypic, lifestyle, and other clinical data to enable de-identified research use. Such registries are facilitated by advancements in genetic testing, including next-generation sequencing, that enable molecular diagnostics. While access to genetic testing may be differentially covered by health care systems and insurance policies, if available, molecular diagnostics may improve health-related quality of life by providing valuable information for reproductive counseling in cases of de novo mutations, information necessary to enroll patients in clinical trials, and empowering patients with knowledge and the opportunity to actively drive the research process forward.
In summary, patients increasingly access health information from print and broadcast media sources and on the Internet and raise this information during clinical visits. However, it is challenging for patients to assess the accuracy and credibility of such information, and reports on research into ocular gene therapy and related biotherapeutics paint a highly optimistic view of timelines and expected outcomes. Studies consistently emphasize the relational nature of care (between patients and their families and clinicians) and demonstrate better outcomes if the concerns of patients are met. Here we discuss how clinicians might aid patients and families in navigating the complex web of information and supporting hope while keeping expectations grounded in current clinical and research realities.
Clinical Communication Topics and Recommendations
Research, Not Treatment
Coverage of novel biotherapeutics conflates research with therapy. The very use of the term “therapy” in a clinical research context implies demonstrated therapeutic effect, but cumulative evidence suggests that fewer than 1% of participants derive a direct benefit from phase I gene therapy clinical trials. Media articles use research and treatment terminology interchangeably, confusing research goals and the state of clinical development. This confusion is problematic in the context of clinical trial enrollment, as hope for therapeutic benefit from early-stage clinical trials exemplifies a “therapeutic misconception.” Such a misconception may undermine consent processes in clinical trial enrollment, which require participants to understand the research goals.
In reality, research extends along a developmental continuum from laboratory research using cell and animal models to preclinical or enabling research—the goals of which are to demonstrate safety and the proposed mode of action of the therapeutic for regulatory clinical trial applications—through the phases of clinical trials. Rates of attrition between phases of clinical trials are high. Of interventions that enter phase 1, only 15% receive market authorization from a regulatory agency following phase 3 clinical trials; the greatest attrition occurs in phase 2 and is likely worse for more complex biotherapeutics.
Phase I clinical trials are designed to test safety and the maximum tolerable dose. Though some safety outcome measures blur with those for efficacy, small phase 1 studies are inadequately powered to test the latter. For example, the first clinical study on choroideremia gene transfer published in The Lancet reported on 6 patients. Similarly, published accounts of the LCA trials reported on 3 participants in the New England Journal of Medicine (2008), a dose-escalation study in 12 participants in The Lancet , and a follow-up study 3 years after treatment in 5 participants. While preliminary results appear promising, these are not without the caveats discussed in the introduction. More definitive evidence of efficacy for LCA gene therapy will emerge from the phase III trial later this year; peer-reviewed, published accounts demonstrating efficacy of ocular gene therapy remain, at present, limited.
Recommendation
When discussing access to an investigational gene therapy product by way of a clinical trial or otherwise, clinicians should address sources of information, the goals of different phases of clinical trials, the credibility of the clinical trial (including the site, sponsor, and research team), and that clinical trials represent research, not therapy. Clinicians may point patients and families to credible sources of information. For example, the Foundation Fighting Blindness website, while acknowledging patient hopes for a treatment, explicitly states: “The word intervention is used instead of the word treatment to help people remember that the drug and/or surgery that the participant is receiving is not an established treatment. A clinical trial is not a treatment it is an experiment.”
Participation in Clinical Research is Not Risk Free
Media coverage of ocular gene therapy has been highly positive; like most coverage of genetics, it emphasizes benefits and provides minimal information on risks. While most published accounts of gene therapy have shown positive safety profiles, some risks are apparent. General risks for gene therapy, which remain hypothetical risks for ocular applications, include oncogenic risks from insertional mutagenesis and a severe immune response. Specific risks include surgical complications, loss of an eye owing to endophthalmitis, loss of remaining vision, and a theoretical risk of brain toxicity owing to integration of the viral vector in the optic nerve. Reported adverse events include a macular hole, foveal thinning, and retinal detachment. Other risks to participants include associated financial burdens and psychological stress. While disclosure of risks is an essential component of the informed consent process for clinical trial participation, urgency to access a trial may divert patient attention from the risks.
Recommendation
When discussing possible participation in a clinical trial, clinicians should highlight that potential risks exist. Clinicians may direct patients to carefully consider and question descriptions of both magnitude and likelihood of risks during the process of informed consent.
Potential Treatment, Not Cure
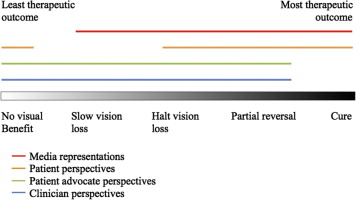
If proven safe and efficacious, gene therapy may present a treatment but not a cure for patients who have already experienced vision loss. While gene therapy may restore the function of dormant yet viable photoreceptors and thereby improve visual outcome measures, unlike proposed cell therapies, it is not regenerative. The LCA trials demonstrate that there may be a therapeutic window for efficacy, with earlier intervention being better, especially if prior to the onset of retinal degeneration. Nevertheless, the media commonly represent gene therapy as a cure for inherited eye diseases. One popular book on LCA gene therapy went so far as to be titled The Forever Fix .
Other stakeholder perspectives tend to be more nuanced. Participants in one choroideremia study anticipated outcomes that ranged from no benefit, through slowing or halting vision loss, to a cure for choroideremia ( Figure 1 ). All stakeholder groups acknowledged the possibility of no benefit. However, although clinicians and advocacy organizations did not express expectations for a cure and considered slowing vision loss as a realistic outcome, patient perspectives ranged from halting vision loss to a cure. In other words, patients overestimated the potential benefits of gene therapy research, which represents a “therapeutic misestimation.” Such overestimation of benefit may be ethically tolerable if its probability was difficult to convey or perceive, but in the case of ocular gene therapy, a cure is highly unlikely. Its long-term effects are unknown at present.