42
Combination Therapy in the Treatment of Choroidal Neovascularization due to Age-Related Macular Degeneration
Albert J. Augustin
INTRODUCTION
Over the past decade, many new treatment options are available for the management of choroidal neovascularization (CNV) secondary to age-related macular degeneration (AMD). These treatments help delay the progress of CNV, offering a temporary slowing, or even some reversal, of the new blood vessel growth. Some provide improvements in visual acuity (VA). The difficulty in finding a more robust and lasting treatment for this condition is multifactorial; it may be due, in part, to the fact that no single action or chain of events is responsible for the damage to the retina. Multiple pathways or factors are involved, and treatments must address each of these pathways in order to be efficacious. At the same time, care must be taken not to harm the normal vasculature of the retina in any way. For these reasons, it may be that combination therapies that target two or more of these factors could be the most ideal way to treat CNV.
NEOVASCULARIZATION AND THE ANGIOGENIC CASCADE
In normal, healthy humans, angiogenesis plays a crucial physiological role in tissue and organ growth, wound healing, and many other processes. However, the irregular and uncontrolled proliferation of new blood vessels can manifest itself as cancerous tumors in many parts of the body. In the context of eye disease, this proliferation presents as CNV.
As the human eye ages, choroidal vascular atrophy leads to a metabolic breakdown where the choriocapillaris underlying the retinal pigment epithelium lose their ability to transfer oxygen and nutrients to the surrounding cells (1, 2). The resulting oxidative stress and hypoxia cause local inflammation beneath the retinal pigment epithelium (RPE), and trigger the upregulation of vascular endothelial growth factor (VEGF), a proangiogenic protein that stimulates the formation of new blood vessels (3). VEGF also upregulates the production and release of matrix metallo-proteinases (MMP) 2 and 9 which degrade the extracellular matrix and lead to endothelial leakage, and vascular endothelial cell migration and proliferation. Plasma proteins are expelled from the vessels and form an inviting bed, on, or around the perimeter of the vessels, where additional new endothelial growth (i.e., lumen and vascular formation) can take place (4, 5).
These newly formed vessels extend into the RPE and Bruch’s membrane, where they create edema and scarring (6) of the retina which can lead to permanent central vision loss.
Other proangiogenic factors that are upregulated and which trigger a strong neovascular response include transforming growth factors-α (acidic) and β (basic), and insulin-like growth factor 1 (7). These factors are not found at detectable levels in the normal RPE, but are expressed in the tissue of newly formed blood vessels.
CAUSES OF NEOVASCULARIZATION
Although VEGF is the most important growth factor in causing new vessel growth, it is not the initiating factor. After normal aging, smoking (8) is one of the most significant contributing factors in the development of AMD because it induces oxidative stress in the retinal vasculature. Other risk factors known to induce trauma or stress in the blood vessels include high body mass index, waist circumference, lack of physical exercise, (9) elevated systolic blood pressure and subclinical atherosclerosis (10). Evidence from epidemiological and observational studies has been inconsistent, but the majority of studies suggest that a diet high in polyunsaturated, monounsaturated, and vegetable fats may be protective against AMD (11).
Importantly, it must also be noted that VEGF expression occurs as an epi-phenomenon secondary to other processes which have different triggers. It has been shown, for example, that the expression of VEGF is upregulated in the endothelial layer of the choriocapillaries as well as in larger choroidal vessels in eyes that have undergone photodynamic therapy. This expression of VEGF was not seen in areas of the same eye that had not been exposed to photodynamic therapy (PDT), or in the eyes of age-matched controls (12). Thus, a procedure that is widely used to treat CNV on one hand, also stimulates additional CNV on the other. Also, CNV itself triggers inflammation in the adjoining tissue, and this again, upregulates the expression of VEGF.
TREATMENT OPTIONS AND THEIR LIMITATIONS
Anti-VEGF Drugs
In the therapeutic setting, treating CNV is not as simple as targeting the angiogenic cascade or any one of its stages. Three antiangiogenic therapies are now widely available: the VEGF aptamer pegaptanib sodium, and the monoclonal antibodies ranibizumab and bevacizumab. Each of these acts by inhibiting the formation of new blood vessels, and has shown some efficacy in reducing CNV.
Rosenfeld et al. (13) showed that ranibizumab could reverse some of the loss in vision caused by CNV, with VA improving in one-quarter to one-third of patients who received monthly intravitreal injections. Gragoudas et al. (14) showed that loss of VA could be slowed with injections of intravitreal pegaptanib every six weeks, and that approximately 20% of patients had a gain of at least 5 ETDRS letters.
Bevacizumab is also widely used to slow or reverse the progress of CNV, although at present, it has not been approved for this purpose by the Food and Drug Administration in the United States, or by any other licensing authorities worldwide. An attractive feature of bevacizumab is that in small studies, it appeared to reverse CNV and led to an improvement in VA with one to three injections, sustained over several months.
But while anti-VEGF monotherapy appears useful for blocking new blood vessel formation, recent research has suggested that it also stimulates the release of compensatory proangiogenic factors which counteract its anti-VEGF activity, and induce new blood vessel formation. This compensatory reaction was not seen in laboratory animals that received a combination therapy involving three elements: anti-VEGF, an integrin-antagonist, and an antagonist of vascular-endothelial cadherin-mediated adhesion. It was observed in animals treated with anti-VEGF monotherapy (15).
This means that not only will the effectiveness of anti-VEGF therapies be limited when they are used as monotherapy, but that also, whatever role they do play may be temporary. Anti-VEGF will antagonize the CNV, reduce or stop vessel leakage and alleviate the edema, but this effect lasts only as long as the anti-VEGF itself. Once the anti-VEGF effect has worn off, leakage and edema recur. This will happen because the compensatory action of releasing new VEGF will continue to stimulate neovascular growth, even as the anti-VEGF treatment blocks the formation of abnormal vasculature. Patients could be required to undergo anti-VEGF treatments for an unlimited, and unknowable length of time. Furthermore, in view of the upregulation of proangiogenic factors in response to anti-VEGF treatment, this could suggest a rebound effect, should the patient ever discontinue anti-VEGF therapy (16). Therefore, controlling or antagonizing the upregulation of VEGF in any of its isoforms is now thought to be only one of the processes involved in treating CNV.
Photodynamic Therapy
A second aspect of treating CNV is the eradication of the existing neovascularization. No matter how effective an antiangiogenic drug may be at preventing the sprouting of new blood vessels in the retina, it does nothing to address the issue of new vessel growth that is already established. Laser photocoagulation therapy has been used for many years in treating some forms of cancer, but its efficacy in treating CNV related to AMD has been limited. On one hand, the procedure uses a precise and focused laser beam, but the CNV is usually somewhat diffuse. The risk of destroying surrounding tissue, especially the photoreceptors lying on top of the neovascularization, is considerable. Also, the procedure can only be used for extrafoveal lesions, not subfoveal or juxtafoveal lesions. Finally, laser photocoagulation generally has a high recurrence rate. PDT was approved as a treatment for CNV in 1999 and offers a complementary action to that seen with anti-VEGF agents. While anti-VEGF drugs arrest, or slow the development of new blood vessels, PDT eradicates the CNV that has already taken place.
PDT entails the intravenous infusion of verteporfin (benzoporphyrin derivative), which is a red laser activated mono-acid. The photosensitizer, once exposed to the laser light, releases highly reactive short-lived singlet oxygen and other reactive oxygen intermediates which lead to cellular endothelial damage, which then destroys the abnormal blood vessels. There is also further release of vasoactive and thrombogenic signaling molecules, which cause acute thrombosis of the capillaries, followed by decreased vessel leakage. Contrary to laser photocoagulation, only the vessels which have taken up the verteporfin are affected, and therefore, the surrounding tissue is not harmed.
Numerous studies worldwide have now demonstrated the benefit of PDT therapy in patients with classic subfoveal, predominantly classic, and occult CNV (Table 42-1). Most trials with PDT as monotherapy demonstrate a reduction or slowing in vision loss rather than an improvement in VA. Significant improvement of VA in large number of patients has not been seen in any of the major PDT monotherapy trials. The procedure mainly benefits patients with minimally classic CNV or mixed lesions. Smaller lesions respond better to treatment than larger lesions (Table 42-1).
In recent years, certain drawbacks to PDT monotherapy have been identified. First, the therapy requires additional treatments as new CNV arises. Past studies have reported patients receiving an average of 3.1 treatments over 12 months (17) and an average of 5.6 treatments over 24 months (18). Secondly, a small study (12) in elderly patients who had been treated with PDT showed overexpression of VEGF in the endothelial layer of choriocapillaries as well as within larger vessels. Overexpression of VEGF was not evident in eyes that had not been treated with PDT. The same sites showed positive upregulation of vascular endothelial growth factor receptor 3 (VEGFR-3) and pigment epithelium derived growth factor (PEDF). Thus, like anti-VEGF when used as monotherapy, PDT also stimulates VEGF expression, which could, in turn, lead to more neovascular proliferation. Furthermore, PDT is known to cause inflammation which exacerbates the cycle of VEGF upregulation and can trigger neovascularization.
Finally, an in vitro study has demonstrated that PDT resulted in a morphologically and functionally detectable breakdown of the outer blood-retinal barrier function of the RPE, although there was no damage to the RPE cells. Increasing the concentration of verteporfin however, can result in RPE cell damage (19).
STEROIDS
The use of intravitreal corticosteroids has also been proposed as a treatment for CNV. There are several reasons for this. The steroid triamcinolone acetonide has anti-inflammatory properties and so could be beneficial in combating the inflammation that results from both oxidative stress and from PDT. All inflammatory cells are known to make and release VEGF (20), therefore, triamcinolone, and other steroids which have antiangiogenic properties could conceivably be used to block this VEGF expression. Steroids also have antifibrotic and antipermeability characteristics which help maintain the blood-retinal barrier. The results from several studies that have used triamcinolone as monotherapy have produced mixed results and were far from conclusive. However, when used in combination with PDT, triamcinolone seems to offer a synergistic action (21).
A significant drawback to the use of intravitreal triamcinolone is that it dramatically increases intraocular pressure (IOP) (22). Such increases in IOP can be deleterious for patients who either have, or are at risk of primary open-angle glaucoma, since the increases may exacerbate the trauma to the optic nerve head. Such increases in IOP have been known to last for several months (23), and in extreme cases have required paracentesis to alleviate the IOP. A second steroid, dexamethasone, has also been proposed as a useful treatment. It offers much the same benefit as triamcinolone in terms of its angiostatic action, but has fewer side effects as compared to triamcinolone. Dexamethasone is a short-acting steroid, and has the advantage of being administered in solution form, rather than as a suspension; therefore it is cleared from the vitreous much faster than triamcinolone. Dexamethasone also exerts little action on the trabecular meshwork and therefore is not associated with the spikes in IOP that are seen with triamcinolone use.
TABLE 42-1 RESULTS FROM MAJOR STUDIES IN THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION
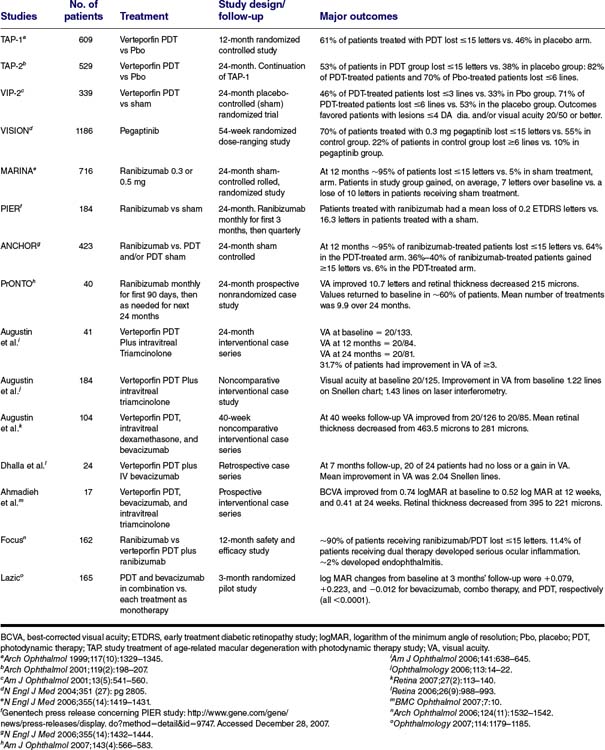
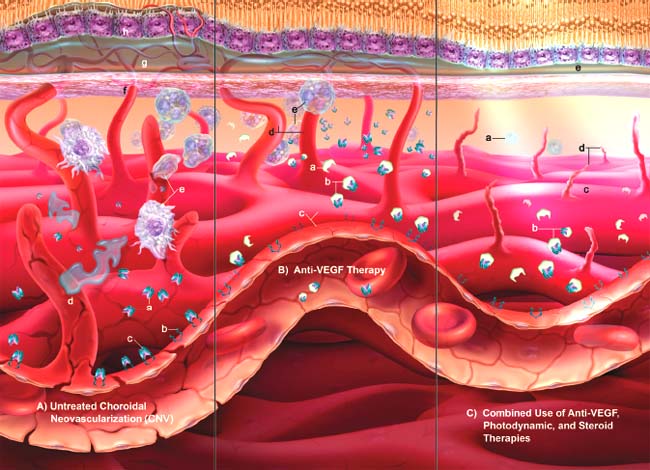
Figure 42-1.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


