This article presents the complexity of asthma and its ensuing management. The author details the clinical presentation of asthma, discussing its variability of presentation over time and within and among individuals. Pathophysiology is discussed, including immunohistopathologic features and inflammatory mediators, risk factors, comorbid conditions, natural history, and triggers. Diagnosis is presented in light of the challenging history of patients suspected of the disorder; pulmonary function testing, symptom assessment, impairment assessment, and risk assessment. Categories of asthma severity, controlling triggers, pharmacotherapy, stepwise approaches, and ongoing assessment are discussed, with a review of a clinical trial assessing efficacy of management.
Asthma is a common, chronic disorder of the airways, but it is a complex disorder. Asthma is characterized by:
- 1.
Variable and recurring symptoms
- 2.
Airflow obstruction
- 3.
Bronchial hyperresponsiveness
- 4.
Underlying inflammation.
The comorbid conditions of allergic rhinitis and asthma have been well documented, are frequently observed, and may represent a spectrum of the same disease entity (1-airway hypothesis). Health care providers managing allergic rhinitis need to be comfortable with their ability to recognize and treat asthma and inflammation of the lower airway.
The clinical presentation of asthma can be highly variable over time and is often dependent on several factors:
- •
The inflammation of asthma in the airway of susceptible individuals causes recurrent episodes of wheezing, breathlessness, chest tightness, and coughing, which often worsen at night and in the early morning
- •
Episodes of inflammation are usually associated with widespread, but often variable, airflow obstruction that is frequently reversible
- •
This inflammation can also cause an increase in the existing bronchial hyperresponsiveness typical of asthma.
The interaction of these features of asthma determines the clinical manifestations and severity of asthma and the response to treatment.
The relationship between the upper and lower airway inflammation should not be underestimated. Rhinitis occurs in 75% to 90% of adult subjects with allergic asthma and 80% of those with nonallergic asthma. The incidence of asthma is high in developed nations, constituting the most common chronic disease of childhood, affecting more than 7 million children in the United States. Allergic disease is associated with the development, severity, and persistence of asthma. As an example, up to 80% of children with atopic dermatitis develop asthma and/or allergic rhinitis later in childhood. Conversely, asthma occurs in 25% to 50% of individuals with rhinitis. Adults with perennial rhinitis are more likely to have asthma than those without rhinitis. The odds of developing asthma are eightfold higher in patients with allergic rhinitis and almost 12-fold higher in patients with nonallergic rhinitis.
Pathophysiology of asthma
The immunohistopathologic features of asthma include an inflammatory cell infiltration into the respiratory tract. Neutrophils, eosinophils, lymphocytes, mast cell activation, and epithelial cell injury are common cellular findings. In some patients with progression of disease, persistent changes in airway structure occur, including subbasement fibrosis, mucus hypersecretion, injury to epithelial cells, smooth muscle hypertrophy, and angiogenesis. Numerous inflammatory mediators have also been implicated in the inflammatory process, and management of these mediators has held some promise in the progression and management of the reactive process of asthma. Chemokines, cytokines, cysteinyl-leukotrienes, nitric oxide, and immunoglobulin E (IgE) all play a role in the development of airway inflammation.
Central to the variable clinical presentations of asthma is the presence of underlying airway inflammation, which is also varied over time within the same individual and among individuals. This inflammatory variability has distinct, but overlapping, patterns that reflect different aspects of the clinical presentation of the disease, such as intermittent versus persistent asthma or acute versus chronic manifestations.
Acute symptoms of asthma usually arise from bronchospasm and require and respond to bronchodilator therapy. Acute and chronic inflammation can affect not only the airway caliber and airflow but also underlying bronchial hyperresponsiveness, which enhances susceptibility to bronchospasm.
Risk factors for asthma
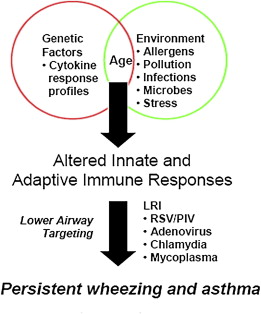
The variables that result in the initiation of the inflammatory process in some individuals are being investigated. Although no definitive pattern exists, there is evidence that the origin of the airway inflammation occurs early. The expression of the clinical presentation of asthma is complex and involves interaction between genetic host factors and environmental exposures occurring at a critical time in the development of the immune system. The development of asthma likely results from complex interactions between multiple genetic and environmental influences ( Box 1 ). The onset of asthma for most patients begins early in life, with the pattern of disease persistence determined by early, recognizable risk factors including atopic disease, recurrent wheezing, and a parental history of asthma.
- •
Innate Immunity
- •
Genetics
- •
Sex
- •
Allergens/type and timing of exposure
- •
Respiratory infections
- •
Environmental exposures
The genetic predisposition for the development of an IgE-mediated response to common aeroallergens is the strongest identifiable predisposing factor for developing asthma. The Third National Health and Nutrition Examination Survey (NHANES) found that one-half of asthma cases were attributable to atopy.
Additional precipitating events may be important variables for exacerbation of asthma and may also contribute to the development of asthma. These events include viral respiratory infections, exposure to environmental tobacco smoke, and obesity ( Fig. 1 ).
Risk factors for asthma
The variables that result in the initiation of the inflammatory process in some individuals are being investigated. Although no definitive pattern exists, there is evidence that the origin of the airway inflammation occurs early. The expression of the clinical presentation of asthma is complex and involves interaction between genetic host factors and environmental exposures occurring at a critical time in the development of the immune system. The development of asthma likely results from complex interactions between multiple genetic and environmental influences ( Box 1 ). The onset of asthma for most patients begins early in life, with the pattern of disease persistence determined by early, recognizable risk factors including atopic disease, recurrent wheezing, and a parental history of asthma.
- •
Innate Immunity
- •
Genetics
- •
Sex
- •
Allergens/type and timing of exposure
- •
Respiratory infections
- •
Environmental exposures
The genetic predisposition for the development of an IgE-mediated response to common aeroallergens is the strongest identifiable predisposing factor for developing asthma. The Third National Health and Nutrition Examination Survey (NHANES) found that one-half of asthma cases were attributable to atopy.
Additional precipitating events may be important variables for exacerbation of asthma and may also contribute to the development of asthma. These events include viral respiratory infections, exposure to environmental tobacco smoke, and obesity ( Fig. 1 ).
Natural history of asthma
Persistence and severity of asthma seems to involve a progression of airway inflammation resulting in airway remodeling and eventual irreversible airway obstruction. An important question is whether antiinflammatory medication (ie, inhaled corticosteroids [ICS]) given early in the course of disease might interrupt this process and prevent permanent declines in lung function, or whether the progression to more persistent and severe disease is predetermined by other variables.
Early studies have indicated that, although current treatments are effective in controlling symptoms, reducing airflow limitations, and preventing exacerbations, present treatments for persistent disease (eg, ICS) do not seem to prevent the underlying severity of asthma from progressing, which suggests that currently available therapy controls, but does not modify, the underlying disease process.
In the pediatric population, age 3 years or younger, patients most at risk for developing progressive disease were most likely to have had 4 or more episodes of wheezing during the previous year, and either a parental history of asthma, a diagnosis of atopic dermatitis, or evidence of sensitization to aeroallergens.
Data on adult onset asthma are inconclusive on the ability of ICS to alter progression of disease, but seem promising. In 2 long-term observational studies, an association was noted between ICS therapy and reduced decline in forced expiratory volume in 1 second (FEV 1 ) in adults who had asthma.
If ICS can change the natural course of asthma, some continued improvement in lung function might be expected once ICS are discontinued. The best available evidence in children aged 5 to 12 years and aged 2 to 3 years showed that, although ICS provide superior control and prevention of symptoms and exacerbations during treatment, symptoms and airway hyperresponsiveness worsen when treatment is withdrawn. This evidence suggests that currently available therapy controls, but does not modify, the underlying disease process.
Asthma Management: Assessment and Monitoring
Key elements of assessment and monitoring include evaluation of severity of symptoms, control of symptoms, and responsiveness to therapy. Severity, defined as the intrinsic intensity of the disease process, can often be easily measured and should be assessed when the patient is not receiving long-term control therapy. Controls of symptoms are defined as the degree to which the clinical manifestations of asthma can be minimized. Assessment of symptoms, functional impairment, and risks of future events should be minimized. Responsiveness is defined as the ease with which asthma control is achieved.
The diagnosis of asthma is the initial step in reducing the symptoms, functional limitations, impairment in quality of life, and risk of adverse future events that accompany the disease. The ultimate goal of treatment is to enable patients to live with none of the manifestations of asthma.
The initial assessment of the severity of the disease allows for an estimate of the type and intensity of treatment needed. Responsiveness to asthma treatment is variable; therefore, to achieve the goals of therapy, follow-up assessment must be made and treatment should be adjusted accordingly. Patients who have asthma that is well controlled at the time of a clinical assessment should be monitored over time, as the processes of asthma can vary in intensity, and treatment should be adjusted accordingly.
Historical information
Some patients with asthma report or present with the classic triad of symptoms: wheeze, cough (typically worsening at night), and shortness of breath or difficulty breathing. Wheezing does not have a standard meaning for patients and may be used by those without a medical background to describe a variety of sounds. Cough may be dry or productive. Other patients have only 1 or 2 of these symptoms. Some patients describe chest tightness or a bandlike constriction. In contrast, chest pain is uncommonly used to describe the sensation of asthma.
Because the symptoms of asthma are also seen in a myriad of other respiratory diseases, it may be difficult to be certain of the diagnosis of asthma based on history alone. However, certain historical features heighten the prior probability of asthma:
- •
Episodic symptoms: asthmatic symptoms characteristically come and go, resolving spontaneously with removal from the triggering stimulus or in response to antiasthmatic medications. Patients with asthma also may remain asymptomatic for long periods of time
- •
Characteristic triggers of asthma include exercise, cold air, and exposure to allergens. Similarly, viral infections are common triggers for asthma, although they can trigger exacerbations in other chronic respiratory conditions as well. A strong family history of asthma and allergies or a personal history of atopic diseases (specifically, atopic dermatitis, seasonal allergic rhinitis and conjunctivitis, or hives) favors a diagnosis of asthma in a patient with suggestive symptoms.
Physical Findings
Widespread, high-pitched, musical wheezes are characteristic of asthma, although these findings are not specific and are often absent between exacerbations of the disease. Wheezes are heard most commonly on expiration, but can also occur during inspiration. The presence or absence of wheezing on physical examination is a poor predictor of the severity of airflow obstruction in asthma. Wheezing may be heard in patients with mild, moderate, or severe airway narrowing, whereas widespread airway narrowing may be present in individuals without wheezing. Thus, the presence of wheezing suggests the likely presence of airway narrowing, but not its severity.
Pulmonary function testing
Variability of patient symptoms and disease exacerbations has resulted in pulmonary function testing becoming a critical tool in the diagnosis and management of asthma. Measurement of peak expiratory flow rate and spirometry are used most frequently.
Peak Expiratory Flow Rate
The peak expiratory flow rate (PEFR) is the measure of airflow during a brief, forceful exhalation. The measurement of peak flow rates can be taught to the patient and routinely used at home to monitor disease severity. As with most pulmonary function testing, the resulting measurements are highly dependent on the patient’s expiratory effort and technique. Thus, it is important that the clinician assesses and monitors the patient’s technique and effort level and corrects any mistakes. The PEFR maneuver can be performed sitting or standing. Proper technique involves taking a maximally large breath in, putting the peak flow meter quickly to the mouth and sealing the lips around the mouthpiece, and blowing as hard and fast as possible into the meter. For PEFR, the effort does not need to be sustained beyond 1 to 2 seconds. The patient should perform the maneuver 3 times and record the highest of the 3 measurements. Patients should perform the test routinely 2 to 3 times per day and keep a log, establishing a personal-best PEFR. PEFRs may be misleading and mild airflow obstruction may be present when the peak flow remains within the normal range. Reduced peak flow measurements do not differentiate between obstructive and restrictive diseases; spirometry and sometimes measurement of lung volumes are necessary to distinguish the two. Peak flow measurements are not sufficient to distinguish upper airway obstruction (eg, vocal cord dysfunction) from asthma. The validity of PEFR measurements depends on patient effort and technique. There is also no standardization among peak flow measurements. Despite the shortcomings, many patients use PEFR to successfully follow the progression of their asthmatic disease.
Spirometry
Spirometry, which includes measurement of FEV 1 and forced vital capacity (FVC), provides valuable information that is useful in the diagnosis of asthma. Spirometry is a simple office procedure that can be completed in 10 to 15 minutes with virtually no risk to the patient. The test is dependent on patient effort, although standard protocols and normative values have been established.
Spirometry can be used to determine normal from abnormal lung function and can categorize the abnormalities into obstructive or restrictive patterns. Spirometry can also be used to characterize the severity of the asthma and can be used to assess the reversibility of the obstructive abnormality if the testing is repeated after administration of a bronchodilator.
Variable airflow obstruction is the hallmark of asthma. An atopic patient with a cough, shortness of breath, and/or wheezing who has expiratory airflow obstruction pattern on spirometry that reverses to normal with treatment almost certainly has asthma. In contrast, the person with cough and chest congestion who has consistently normal spirometry probably does not have asthma, and alternative explanations (eg, recurrent bronchitis) should be sought.
An obstructive pattern on spirometry is identified numerically by a reduction in the ratio of FEV 1 to the FVC. When FEV 1 /FVC is reduced to less than normal (best defined by 95% confidence intervals around the normal values, information that is provided electronically by modern computerized spirometers), airflow obstruction is present. When the FEV 1 /FVC ratio is normal or increased, there is no expiratory airflow obstruction.
Having identified the presence of airflow obstruction by a reduction in FEV 1 /FVC, the severity of airflow obstruction is then categorized by the degree of reduction of the FEV 1 less than normal. By convention, the severity of airflow obstruction based on spirometry is graded as mild, moderate, severe, and very severe based on the reduction in FEV 1 . These categories are used for pulmonary function interpretation and are not the same as categories used to stage asthma severity.
Bronchodilator Response
The ability to acutely reverse airflow obstruction is tested by administering 2 to 4 puffs of a quick-acting bronchodilator (albuterol) and repeating spirometry 10 to 15 minutes later. The variability of the measurement is such that an increase of less than 12% may occur simply because of making repeated measurements. However, an increase in FEV 1 of 12% or more, accompanied by an absolute increase in FEV 1 of at least 200 mL, can be attributed to bronchodilator responsiveness with 95% certainty ( Fig. 2 ).




