Purpose
To study the association of ARMS2 A69S genotype with the development of exudative age-related macular degeneration (AMD) in the unaffected fellow eye and to estimate the duration until the development of AMD in the second eye.
Design
Retrospective cohort study.
Methods
We retrospectively reviewed 326 patients who had exudative AMD in at least 1 eye, genotyping of ARMS2 A69S, and a minimum follow-up of 2 years. Survival analysis and Cox proportional hazard regression analysis were used to examine the association between candidate factors and the duration until the development of AMD in the second eye.
Results
One hundred nineteen patients (36.5%) had bilateral exudative AMD at the initial visit. A risk allele of ARMS2 A69S was more frequently seen in patients with bilateral AMD ( P = .0270) than in those with unilateral AMD. Of the 207 unilateral AMD patients, 23 (11.1%) had AMD in the fellow eye after a mean duration of 56.3 ± 40.4 months. Fellow-eye involvement was associated with ARMS2 A69S genotype (hazard ratio [HR], 2.673; P = .0013), age (HR, 1.102; P = .0005), and smoking history (HR, 0.680; P = .3663). As HRs indicate, correlation of genotype (2.673) was as high as that of 10-year aging (1.102 10 = 2.641). Survival analysis revealed that patients with risk homozygous (TT) genotype had second-eye involvement significantly earlier than those with other genotypes ( P = .0028). When the observation duration reached 120 months, second-eye involvement had developed in 50%, 6.6%, and 11.2% of the TT, GT, and GG cohorts, respectively.
Conclusion
ARMS2 A69S genotype is associated with second-eye involvement of exudative AMD and with the period between first- and second-eye involvements.
Exudative age-related macular degeneration (AMD) is one of the most common vision-threatening eye diseases currently seen in developed countries. Although its exact pathogenesis remains unknown, authors of population-based studies have reported various factors associated with the development of exudative AMD, including age, cataract, sunbathing, sex, history of smoking, hypertension, and soft drusen. In the clinical setting, some patients with unilateral exudative AMD maintain good visual function in the fellow eye for a long time, while others have development of exudative AMD in the fellow eye. When visual disturbance attributable to AMD is seen in 1 eye, the impairment of quality of life (QOL) may be limited, but the involvement of exudative AMD in the second eye, when accompanied by a visual disturbance, often causes a severe decrease in QOL. The rate of bilateral involvement of exudative AMD in whites has been reported to vary from 6% to 9% annually. In Japanese patients, the rate is relatively low, with a cumulative incidence of only 11% to 12% over 5 years having been documented.
Recently, many genetic factors have been reported in the development of exudative AMD, including ARMS2/HTRA1 , CFH , and C2/CFB. Although CFH is the most prevalent susceptibility gene in whites, ARMS2/HTRA1 is the most prevalent gene associated with AMD in Asians. Andreoli and associates have shown that ARMS2/HTRA1 is associated with phenotypic attributes of AMD, while CFH is not. A higher risk for bilateral advanced disease has been shown in several articles, and a higher risk of ARMS2/HTRA1 for exudative disease than for atrophy has also been described. An increasing number of reports have shown that ARMS2 A69S is strongly associated with exudative AMD as well as with typical AMD and polypoidal choroidal vasculopathy (PCV). In addition, HTRA1 polymorphism has been significantly associated with bilateral involvement of exudative AMD, and Sakurada and associates recently reported a significant association between ARMS2 A69S polymorphism and bilaterality of PCV. Accordingly, it might follow that patients with unilateral exudative AMD have a higher risk for the development of exudative AMD in the fellow eye if they have a risk allele of ARMS2 A69S. It would be a great help for both physicians and patients to be better able to estimate the risk of fellow-eye involvement by exudative AMD in order to determine visit frequency and treatment strategy. However, limited information is available about genetic risk factors for fellow-eye involvement of exudative AMD. In the study described herein, we assessed the association of the genotype of ARMS2 A69S and fellow-eye involvement by exudative AMD. In addition, survival analysis was conducted to estimate the elapsed time from the initial visit for first-eye involvement until second-eye involvement, depending on the particular genotype of ARMS2 A69S.
Patients and Methods
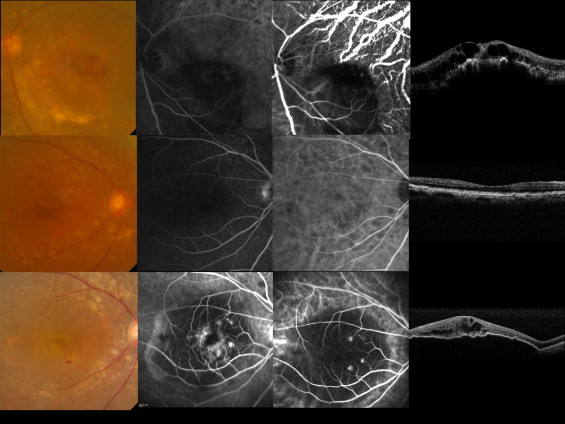
For this observational case study, we reviewed retrospectively the medical records of 326 patients with exudative AMD who visited the Macular Service of the Department of Ophthalmology at Kyoto University Hospital between May 1, 2004 and April 30, 2007. Inclusion criteria of this study were 1) exudative AMD in at least 1 eye, 2) initial comprehensive ophthalmic examination of both eyes, and 3) minimum follow-up of 2 years after the initial presentation. The diagnosis of exudative AMD was based primarily on indirect ophthalmoscopy and fluorescein angiography, according to the definition of the International Classification System for Age-Related Maculopathy, but we also used indocyanine angiography and optical coherence tomography (OCT) to make the diagnosis. The current study of AMD included patients with PCV and retinal angiomatous proliferation (RAP). However, patients with other macular abnormalities (ie, pathologic myopia, idiopathic choroidal neovascularization [CNV], presumed ocular histoplasmosis, angioid streaks, and other secondary CNV) were excluded from the study. If detailed examination of either eye was difficult because of ocular disease other than AMD, the patient was also excluded from the study.
Baseline characteristics of the patients were obtained from their medical charts, including age, sex, presence of hypertension and diabetes, and history of smoking. Each patient’s smoking status was categorized into never smoker, former smoker, and current smoker, according to the classification by Nakanishi and associates. At the initial visit, each patient underwent a comprehensive ophthalmic examination, including determination of best-corrected visual acuity (VA), intraocular pressure measurement, indirect ophthalmoscopy, slit-lamp biomicroscopy with a contact lens, and OCT examination. After fundus photographs were taken, fluorescein angiography and indocyanine green angiography were performed on each patient, using a confocal laser scanning system (HRA-2; Heidelberg Engineering, Dossenheim, Germany). At each scheduled follow-up visit, each patient underwent a complete ophthalmic examination, including VA measurement, slit-lamp biomicroscopy, indirect fundus ophthalmoscopy, and OCT examination. Fluorescein and indocyanine green angiography was performed if necessary.
Preparation of genomic DNA was carried out from peripheral blood using a DNA extraction kit (QuickGene-610L, Fujifilm, Minato, Tokyo, Japan). CFH Y402H rs1061170, I62V rs 800292, and ARMS2 A69S rs10490924 were genotyped via the Taqman SNP assay with the ABI PRISM 7700 system (Applied Biosystems, Foster City, California, USA).
All values are presented as mean ± standard deviation. Statistical analysis among genotypes was performed using χ 2 test for trend or its exact counterpart. In the current study, the date of occurrence of exudative AMD in the second eye was regarded as the date when the physicians documented AMD newly developing in the fellow eye. A Cox proportional hazard regression analysis was conducted to analyze the association between genotype, smoking history, or age with involvement of this fellow eye. In the current study, survival analysis, with the AMD-free period in the better eye after initial visit, was conducted using Kaplan-Meier methods to analyze the relationship between genotype and second-eye involvement. Of the 207 patients, 29 (14%) were lost to follow-up. A difference was considered statistically significant when the P value was less than .05.
Results
In the current study, we examined 326 patients (227 male and 99 female) with exudative AMD. The patients ranged in age from 50 to 90 years (71.6 ± 8.0 years) and all were Japanese. Of the 326 patients, 119 (36.5%) were diagnosed as having bilateral exudative AMD at the initial visit. Table 1 shows the general and ocular characteristics of patients with either unilateral or bilateral AMD at the initial visit. There was no significant difference in sex distribution or in coexisting diabetes mellitus or hypertension between patients with unilateral AMD and those with bilateral AMD ( P = .1987, P = .4798, and P = .4650). The mean age of patients with bilateral AMD was significantly higher than that of patients with unilateral AMD ( P < .0001), and the proportion of current smokers among bilateral AMD patients was significantly greater than in unilateral AMD patients ( P = .0076). A risk allele of ARMS2 A69S was associated significantly with bilaterality of AMD ( P = .027). In addition, polypoidal lesions were more commonly seen in patients with unilateral AMD than in those with bilateral AMD ( P = .0068) at the initial visit.
| Unilateral n = 207 | Bilateral n = 119 | P Value | |
|---|---|---|---|
| Sex, n (%) | .1987 | ||
| Female | 68 (32.9) | 31 (26.1) | |
| Male | 139 (67.1) | 88 (73.9) | |
| Age (y; mean ± standard deviation) | 70.1 ± 7.9 | 74.0 ± 7.7 | <.0001 |
| Smoking, n (%) | .0076 | ||
| None | 97 (46.9) | 40 (33.6) | |
| Former | 51 (24.6) | 49 (41.2) | |
| Current | 42 (20.3) | 28 (23.5) | |
| Diabetes mellitus, n (%) | 20 (9.7) | 8 (6.7) | .4798 |
| Hypertension, n (%) | 49 (23.7) | 24 (20.2) | .4650 |
| Genotype of ARMS2 A69S (GG/TG/TT) | .0270 | ||
| GG | 43 (20.8) | 22 (18.5) | |
| TG | 88 (42.5) | 33 (27.7) | |
| TT | 76 (36.7) | 64 (53.8) | |
| Polypoidal lesion in either eye, n (%) | 144 (69.6) | 65 (54.6) | .0068 |
To determine those factors associated with fellow-eye involvement, we further examined 207 patients (139 male and 68 female) with unilateral AMD at the initial visit ( Table 2 ). The mean follow-up duration was 56.0 ± 30.2 months (range, 24–182 months). In 23 of these 207 patients (11.1%), exudative AMD developed in the fellow eye during the follow-up period ( Fig. 1 ). The mean elapsed time from the initial visit until the development of exudative AMD in the fellow eye was 56.3 ± 40.4 months (range, 2–149 months). Table 3 shows general and ocular characteristics of patients with and without fellow-eye involvement. There was no significant difference in sex distribution, smoking, coexisting diabetes mellitus or hypertension, or detection of polypoidal lesion in the first eye between the 2 groups ( P = .6192, P = .8353, P = .9769, and P = .7679, respectively). The mean age of the fellow eye (−) group was higher than that in the fellow-eye involvement (+) group ( P = .0110). Regarding the distribution of ARMS2 A69S genotypes, the GG, TG, and TT genotypes were seen in 3, 4, and 16 patients with fellow-eye involvement, respectively, while seen in 40, 84, 60 patients without fellow-eye involvement, respectively. The risk allele of ARMS2 A69S was significantly associated with fellow-eye involvement ( P = .0054). In contrast, no association was observed with CFH Y402H rs1061170 or I62V rs 800292 in the current study.
| Fellow-Eye Involvement (+) n = 23 | Fellow-Eye Involvement (−) n = 184 | P Value | |
|---|---|---|---|
| Sex | .6192 | ||
| Female | 6 (26.1) | 62 (33.7) | |
| Male | 17 (73.9) | 122 (66.3) | |
| Age (y; mean ± standard deviation) | 69.8 ± 7.9 | 72.4 ± 7.7 | .0110 |
| Smoking (none/former/current) | .0619 | ||
| None | 13 (56.5) | 84 (45.7) | |
| Former | 10 (43.5) | 51 (27.7) | |
| Current | 0 (0) | 42 (22.8) | |
| Diabetes mellitus | 3 (13.0) | 17 (9.2) | .8353 |
| Hypertension | 6 (26.1) | 43 (23.4) | .9769 |
| Polypoidal lesion in the first eye | 15 (65.2) | 130 (70.7) | .7679 |
| ARMS2 A69S genotype (GG/TG/TT) | .0054 | ||
| GG | 3 (13.0) | 40 (21.7) | |
| TG | 4 (17.4) | 84 (45.7) | |
| TT | 16 (69.6) | 60 (32.6) |
| Variables | Fellow-Eye Involvement (+) | Fellow-Eye Involvement (−) | Hazard Ratio | 95% CI | P Value |
|---|---|---|---|---|---|
| Genotype | 2.673 | 1.443–5.489 | .0013 | ||
| GG | 3 | 40 | |||
| TG | 4 | 84 | |||
| TT | 16 | 60 | |||
| Smoking | (Never & Former) vs Current | 0.680 | 0.286–1.573 | .3663 | |
| Never & former | 23 | 135 | |||
| Current | 0 | 42 | |||
| Age (y), mean ± standard deviation | 69.8 ± 7.9 | 72.4 ± 7.7 | 1.102 | 1.043–1.169 | .0005 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


