EXTERNAL EAR
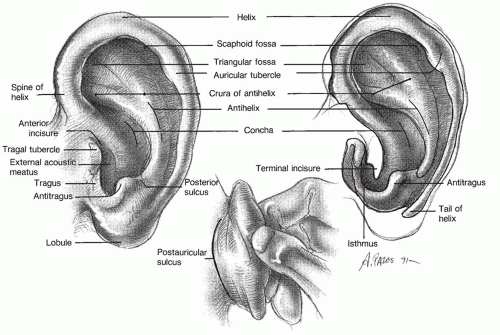
The external ear consists of the pinna (auricle) and the external auditory canal (EAC) from the meatus to the tympanic membrane (
Fig. 141.1). The pinna of humans is composed mostly of cartilage and has no useful muscles. The center of the pinna, the concha, leads to the external auditory meatus, which is about 2.5 cm long. The lateral third of the canal is the cartilaginous portion. It contains cerumen-producing glands and hair follicles. The remaining medial two-thirds is the bony portion, including an epithelial lining over the tympanic membrane (
1).
The external ear and the head have a passive but important role in hearing because of their acoustic properties. The concha, or bowl of the auricle, has a resonance of about 5 kHz, and the irregular surface of the pinna introduces other resonances and antiresonances. These acoustic features are useful to help differentiate whether sound sources are in front of the listener or behind.
The EAC is essentially a tube that is open at one end and closed at the other; thus, the EAC behaves like a quarter-wave resonator. The resonant frequency (f0) is determined by the length of the tube; the curvature of the tube is irrelevant. For a tube of 2.5 cm, the resonant frequency is approximately 3.5 kHz:
f0 = Velocity of sound @ 350 m/s/(4 × 2.5 cm)
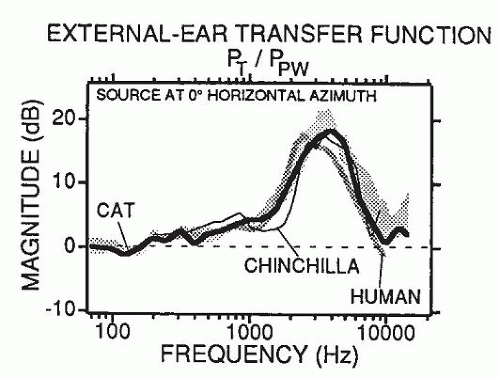
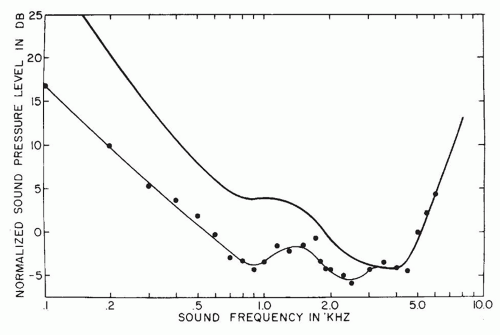
A flat, wide-band sound measured in a sound field is changed considerably by the acoustic properties of the head and external ear. As
Figure 141.2 demonstrates, a gain of about 15 dB occurs in the 3-kHz range of the human, cat, and chinchilla, and 10 dB between 2 and 5 kHz. The acoustic properties of the external ear are one of the reasons noise-induced hearing losses occur first and most prominently at the 4-kHz frequency region (boilermaker notch).
In addition to the prominence of noise-induced hearing loss in the 4-kHz region, the acoustic properties of the head and external ear have an important role in several hearing functions. In localization of sound sources, the head acts as an attenuator at frequencies at which the width of the head is greater than the wavelength of the sound. Thus at frequencies greater than 2 kHz, a head shadow effect occurs, in which interaural intensity differences of 5 to 15 dB are used to localize sound sources. At lower frequencies, at which the wavelength of the sound is larger than the width of the head, little attenuation is provided by the head. Interaural time differences (˜0.6 ms for sound to travel across the head) are the salient cues for localization. The head-shadow effect is the reason right-handed hunters using rifles and shotguns have larger hearing losses in their left ears than in their right ears and vice versa. The muzzle of the gun, where the acoustic energy is greatest, is closer to the left ear, and the right ear is protected by the headshadow effect.
The 10- to 15-dB gain provided by the external ear in the 3- to 5-kHz region is useful for improving the detection and recognition of low-energy, high-frequency sounds such as voiceless fricatives. The importance of the acoustic properties of the external ear and head is reflected in hearing-aid design and evaluations. Finally, the resonance of the external canal is approximately 8 kHz in infants and decreases to adult values after approximately 2.5 years of age. This developmental feature has several clinical implications, especially for sound-field testing and for hearingaid design and evaluation of infants.
MIDDLE EAR
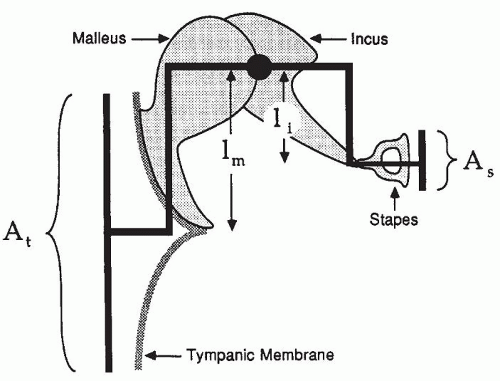
The middle ear transmits acoustic energy from the air-filled EAC to the fluid-filled cochlea. It functions as an impedance- matching device inasmuch as it couples the low impedance of air to the high impedance of the fluid-filled cochlea. The impedance match is achieved in three ways. The first and most important factor is that the effective vibratory area of the tympanic membrane is approximately 17 to 20 times greater than the effective vibratory area of the stapes footplate (
Fig. 141.3). A second factor involves the lever action of the ossicular chain. The arm of the long process of the incus is shorter, by a factor of 1.3, than the length of the manubrium and neck of the malleus. A third and minor factor is the shape of the tympanic membrane. The combined result of these three factors is a pressure gain of approximately 25 to 30 dB. The variance in published
measurements of the transformer ratio is noteworthy. With the exception of studies of acoustic impedance of the ear, most data are from studies of human cadavers, with all of their shortcomings, or of animals, usually cats. In addition to its role in the transfer of power to the inner ear, the tympanic membrane protects the middle ear space from foreign material of the ear canal and maintains the air cushion that prevents insufflation of foreign material from the nasopharynx though the eustachian tube.
The vibratory behavior of the ossicular chain is described in
Figure 141.3. The transformer action of the tympanic membrane and ossicular chain provides for relatively efficient transfer of power to the inner ear, and the fidelity of sound transmission across the middle ear is outstanding. Distortion of sound signals does not occur in the middle ear, even for input signals with sound levels greater than 130 dB sound pressure level (SPL).
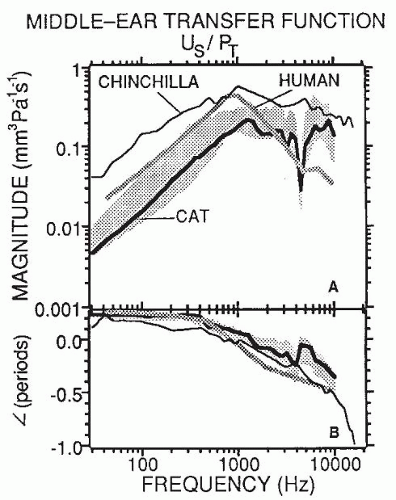
The middle ear, including the tympanic membrane, ossicular chain with supporting ligaments, and middle ear space, can be viewed as a passive mechanical system with both mass and compliant elements and therefore resonant properties. This linear system is coupled to the cochlea, which contributes a large resistance. The result is a middle ear system that is highly damped and linear and has a wide frequency response. The input-output function or transfer function of the middle ear is shown in
Figure 141.4A. The ratio of the volume velocity of the stapes to sound pressure at the tympanic membrane increases in humans to approximately 800 to 900 Hz, which is the resonant frequency of the middle ear, and decreases at higher frequencies. Phase shift or time lag between movement of the tympanic membrane and the stapes generally increases with frequency (
Fig. 141.4B). Although the middle ear is an impressive system in terms of frequency response, linearity, and transformer properties, considerably less than half of the power entering the middle ear actually reaches the cochlea because of the absorption of energy by the ligaments and middle ear. As shown in
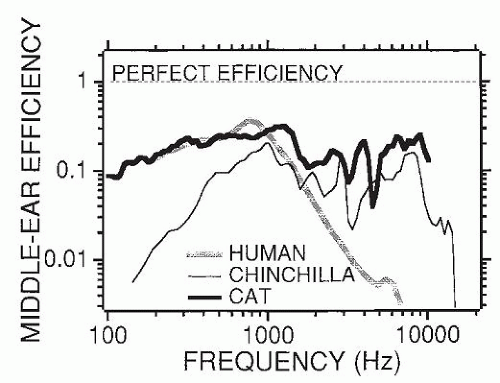
Figure 141.5, the human middle ear is particularly inefficient at frequencies greater than 2 kHz, especially in comparison with the ears of cats and chinchillas. It also is important to recall that a 50% loss of power is a loss of only 3 dB.
Auditory function is profoundly affected by cochlear impedance as well as the combined acoustic effects of the head, external ear, and middle ear. The combined effects of the acoustic properties of the head, external ear, and middle ear, as well the input impedance of the cochlea, have a profound effect on auditory function. For example, these factors determine the shape of the audibility curve and therefore the frequency range of human hearing (
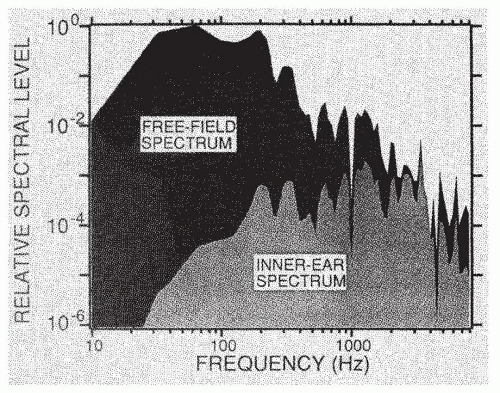
Fig. 141.6). For example, humans do not detect and recognize sounds greater than approximately 20 kHz because such high-frequency sounds are not transmitted efficiently through the middle ear to the cochlea. A second example of this sound transformation is shown in
Figure 141.7, in which the spectrum of a cannon measured in a sound field is compared with the spectrum by the time it is transformed and shaped by the acoustic properties of the external ear, head, middle ear, and input impedance of the cochlea. Low-frequency energy is not transmitted to the cochlea, and the frequency region of greatest energy concentration is 3 to 4 kHz. Thus, these acoustic properties are primarily responsible for the ability of intense lowfrequency sounds (measured in a sound field) to produce high-frequency hearing losses and injuries in the basal region of the cochlea.
Two striated muscles, the tensor tympani and the stapedius, are located in the middle ear. The former attaches to
the malleus and is innervated by the trigeminal nerve. The stapedius muscle attaches to the stapes and is innervated by the stapedial branch of the facial nerve. Noticeably the stapedius and tensor tympani muscles are the smallest striated muscles in the body and also have a high innervation ratio, that is, nerve fibers per muscle fiber. Although no question remains that contraction of these muscles affects sound transmission through the middle ear, the details of the effect and the extent of the influence of the middle ear muscles are still not fully understood. A number of disparate functions have been attributed to the middle ear muscles.
One function of the middle ear muscles is to protect the cochlea from loud sounds (
2). When sounds louder than approximately 80 dB SPL are presented monaurally or binaurally, consensual (bilateral) reflex contraction of the stapedius muscle occurs. This contraction increases the stiffness of the ossicular chain and tympanic membrane, attenuating sounds less than approximately 2 kHz. Although the tensor tympani contracts as part of a startle response, acoustic reflex data from human subjects with neurologic involvement of cranial nerves V and VII suggest that the tensor tympani does not normally respond to intense acoustic stimulation. Laboratory and field studies of noise-induced hearing loss have shown convincingly that the stapedial reflex protects the cochlea, particularly from low-frequency (<2 kHz) sounds in excess of 90 dB. Inasmuch as the latency of the acoustic reflex is greater than 10 ms, the cochlea may be unprotected from shortduration, unanticipated impulsive sounds.
The following functions have been attributed to the middle-ear muscles. Some of these functions include providing strength and rigidity to the ossicular chain; contributing to the blood supply of the ossicular chain; reducing physiologic noise caused by chewing and vocalization; improving the signal-to-noise ratio for high-frequency signals, especially high-frequency speech sounds such as voiceless fricatives, by means of attenuating high-level, low-frequency background noise; functioning as an automatic gain control and increasing the dynamic range of the ear; and smoothing out irregularities in the middle-ear transfer function.
COCHLEA
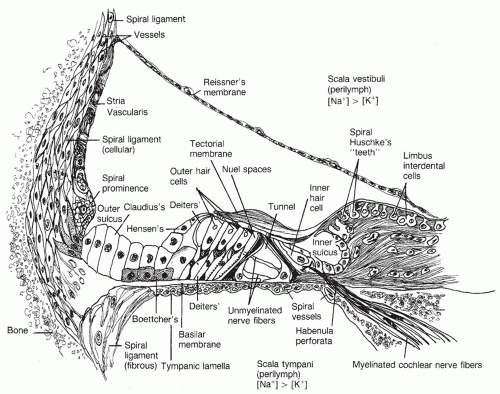
The human cochlea is a coiled, bony tube approximately 35 mm long, divided into the scala vestibuli, scala media, and scala tympani (
Fig. 141.8). The scalae vestibuli and tympani contain perilymph, an extracellular fluid-like material with a potassium concentration of 4 mEq/L and a sodium concentration of 139 mEq/L. The scala media
is bounded by the Reissner membrane, the basilar membrane and osseous spiral lamina, and the lateral wall. It contains endolymph, an intracellular-like fluid with a potassium concentration of 144 mEq/L and a sodium concentration of 13 mEq/L. The scala media has a positive direct current (DC) resting potential of approximately 80 mV that decreases slightly from the base to apex. This endocochlear potential is produced by the heavily vascularized stria vascularis of the lateral wall of the cochlea. The sodium-potassium-adenosine triphosphatase (Na
+-K
+-ATPase) pumps in a number of specialized cells of the stria vascularis contribute to this potential (
3).
Acoustic energy enters the cochlea through the pistonlike action of the stapes footplate on the oval window and is coupled directly to the perilymph of the scala vestibuli. The perilymph of the scala vestibuli communicates with the perilymph of the scala tympani through a small opening at the apex of the cochlea known as the
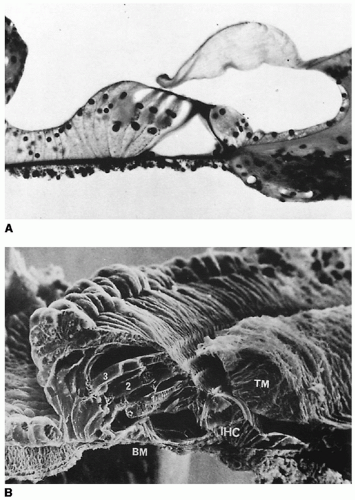
helicotrema. The organ of Corti rests on the basilar membrane and osseous spiral lamina (
Fig. 141.9). The basilar membrane is approximately 0.12 mm wide at the base and increases to approximately 0.5 mm at the apex. The major components of the organ of Corti are the outer and inner hair cells, supporting cells (Deiters, Hensen, Claudius), tectorial membrane, and the reticular lamina-cuticular plate complex (
Fig. 141.10). Supporting cells provide structural and metabolic support for the organ of Corti. The phalangeal processes of the Deiters cells form tight cell junctions of the reticular lamina.
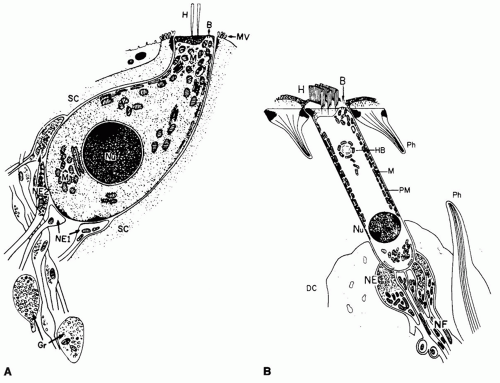
Outer and inner hair cells of the organ of Corti are important in transduction of the mechanical (acoustic) energy into electrical (neural) energy. Outer hair cells are radically different from inner hair cells.
Figure 141.11 and
Table 141.1 detail these differences (
4). In addition to the morphologic differences between outer and inner hair cells, neural innervation is different (
Fig. 141.12). The spiral ganglion, the cell body of the auditory nerve, sends axons to the cochlear nucleus of the brainstem, whereas the dendrite projects through the osseous spiral lamina. Of the 50,000 neurons that innervate the cochlea, 90% to 95% synapse directly on inner hair cells. These are called
type I neurons. Each inner hair cell is innervated by approximately 15 to 20 type I neurons. In contrast, 5% to 10% of the 50,0000 neurons innervate the outer hair cells (
type II neurons). Each type II neuron branches to innervate approximately 10 outer hair cells. In addition to the
afferent innervation pattern of the cochlea, approximately 1,800 efferent fibers, originating from the ipsilateral and contralateral superior olivary complex, project to the cochlea (
Fig. 141.13).
Transduction is initiated by displacement of the basilar membrane in response to displacement of the stapes due to acoustic energy. The displacement pattern of the basilar membrane is a traveling wave (
Fig. 141.14). The basilar membrane is stiffer at the base than in the apex. The stiffness component is distributed continuously. Therefore, the traveling wave always progresses from the base to apex. The maximal amplitude of basilar membrane displacement varies as a function of stimulus frequency. Traveling waves produced by high-frequency sounds (10 kHz) have maximal displacement near the base of the cochlea, whereas the waves to low-frequency sounds (125 Hz) have the maximum toward the apical region. Traveling waves generated by high-frequency sounds do not reach the apical regain of the cochlea, whereas waves to low-frequency sounds can travel the entire length of the basilar membrane.
In the past, the mechanical traveling wave was considered a broadly tuned response, with finer tuning introduced subsequently by transduction, the auditory nerve, and the CNS. Data obtained with sensitive recording and detection methods, however, have shown that the traveling wave has an extremely sharply tuned response (
Fig. 141.15) and that many of the remarkable frequency-selective abilities of the ear can be explained by the mechanical properties of the cochlea.
The mechanism by which the sharply tuned peak is generated within the mechanical traveling wave involves an enhancement known as the
cochlear modifier. This is an activity of the outer hair cells that enhances the motion of the basilar membrane at frequencies near the best frequency of the particular cochlear location. This enhancement contributes to the fine frequency-selective abilities of the ear and to the sensitivity of the ear and ability to detect extremely faint sounds. The notion of an active process in the cochlea, the cochlear amplifier, is supported by the phenomenon of
otoacoustic emissions. That is, when a shortduration signal is presented to the ear, an echo emanating
from the cochlea can be recorded in the external auditory meatus. Because the energy of the echo can be greater than the energy of the short-duration signal, an active process, the cochlear amplifier, is assumed. Factors that may contribute to the cochlear amplifier include motility of outer hair cells and the mechanical properties of the stereocilia and tectorial membrane.