Over 40% of laryngeal cancers present as advanced stage disease. The 5-year survival also varied according to clinical stage (Table 124.1). Stage III supraglottic and glottic cancer had a 5-year survival of 50% to 60%. Stage IV supraglottic cancer has a 5-year survival of less than 50%, and Stage IV glottic cancer has a 5-year survival of 30% to 57% (3).
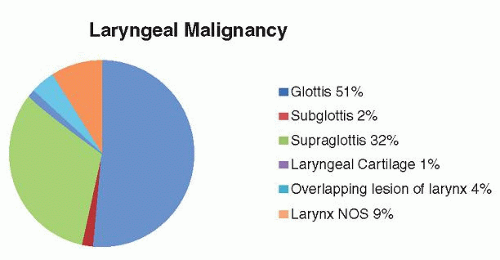 Figure 124.1 Incidence of laryngeal cancer by subsite. Compiled from the National Cancer Database and Hoffman et al. (8). Approval for use obtained. |
|
preepiglottic space by traveling through lacunae on the laryngeal surface of the epiglottis into the space. The paraglottic space is another potential space bound laterally by the perichondrium of the thyroid cartilage and cricothyroid membrane and posteriorly by the mucosa of the pyriform sinus. Medially, the paraglottic space is bound by the quadrangular membrane above the ventricle and the conus elasticus below the ventricle. The conus elasticus extends superiorly from the superior border of the cricoid cartilage to merge with the inferomedial border of the vocal ligament. This ligament resists the extralaryngeal spread of early glottic and subglottic tumors. The paraglottic space allows tumors to become transglottic (superior and inferior to the ventricle) tumors and also to impair the movement of the true vocal cord. The preepiglottic space and paraglottic space communicate with each other.
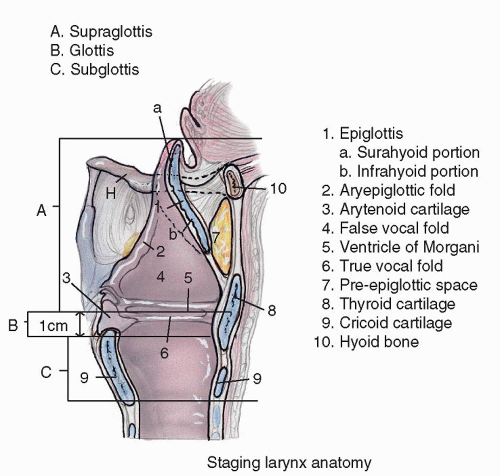 Figure 124.2 Midline sagittal section of the larynx demonstrating the supraglottis, glottis and subglottis. |
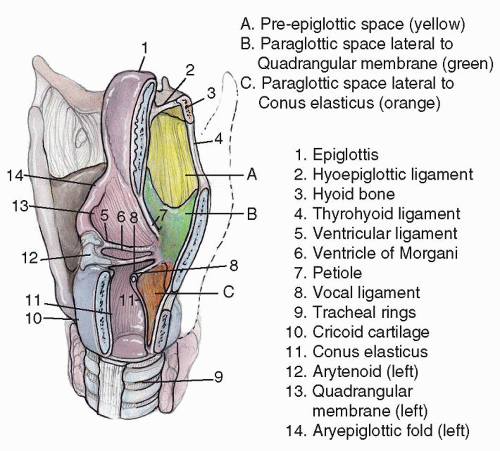 Figure 124.3 Parasagittal view of larynx demonstrating laryngeal spaces and cartilages and ligaments that compose them. |
TABLE 124.2 STAGING LARYNGEAL CARCINOMA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
cancers due to the early change in voice. Supraglottic tumors present with fewer symptoms in the early stage and therefore are usually diagnosed at a later stage. Patients with supraglottic tumors tend to present with pain, dysphagia, and cervical metastasis. Subglottic tumors typically present as advanced stage disease with airway obstruction.
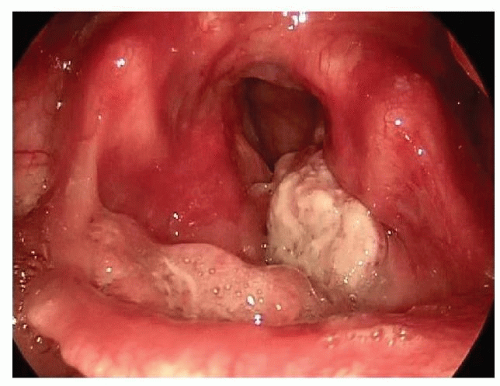 Figure 124.4 Seventy-degree fiberoptic view of T3N0M0 SCCA supraglottis. Tumor extends from epiglottis onto left false fold. |
extent of the primary tumor; however, it is becoming the standard in the assessment of regional cervical metastasis as well as distant metastasis (21). The sensitivity and specificity of PET in detecting cervical metastatic disease range from 74% to 94% and from 82% to 100%, respectively, compared with the sensitivity and specificity of CT, which ranged from 67% to 81% and from 47% to 81%, respectively (22,23,24,25,26,27). Kyzas et al. (28) found that the overall sensitivity of PET in detecting cervical nodal metastasis was 79%; however, in tumors with nodal status as N0, the sensitivity decreased to 50%. Yamazaki et al. (27) found that metastatic nodes measuring greater than 1cm were detected 100% of the time whereas nodes measuring less than 5 mm were not detected. In the posttreatment period, PET has become useful in the identification of residual and recurrent disease (21). In the assessment of distant metastasis, Gourin et al. (29) reported that the sensitivity and specificity of PET-CT detecting distant metastasis were 86% and 84%, respectively.
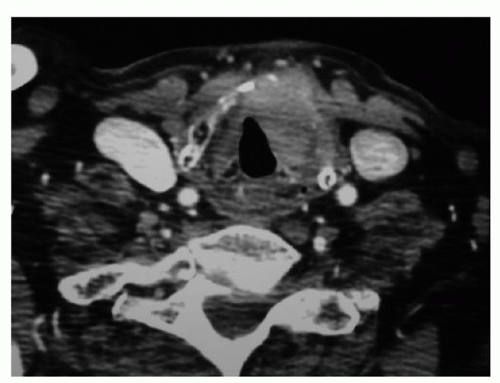 Figure 124.5 Axial contrast CT scan, T4N0M0 transglottic SCCA demonstrating paraglottic space involvement and thyroid cartilage invasion with extralaryngeal spread. |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


