Classification of the Glaucomas
APPROACHES TO CLASSIFICATION OF GLAUCOMA
There are several systems by which the glaucomas have been classified. The most commonly used ones are based on (a) etiology—that is, the underlying disorder that leads to an alteration in aqueous humor dynamics or retinal ganglion cell loss or (b) mechanism—that is, the specific alteration in the anterior chamber angle that leads to a rise in intraocular pressure (IOP). One disadvantage of both systems is that they incorrectly suggest that elevated IOP is the primary risk factor in the glaucomas. A second disadvantage is that neither system incorporates the underlying genetic architecture that contributes to the majority of glaucomas. However, until we understand the causes and mechanisms of the glaucomas more completely, these systems provide the most useful ways to classify the glaucomas.
CLASSIFICATION BASED ON ETIOLOGY
The glaucomas have traditionally been divided on the basis of primary and secondary forms. This division is arbitrary and artificial, however, in that all glaucomas are secondary to some abnormality, whether inherited or environmental. The historical basis for this division was the assumption that the initial events leading to outflow obstruction and IOP elevation in those glaucomas called primary (e.g., open-angle, angle-closure, and congenital) are confined to the anterior chamber angle or conventional outflow pathway, with no apparent contribution from other ocular or systemic disorders. These conditions typically are bilateral and probably have a genetic basis. In contrast, other glaucomas have been classified as secondary because of a partial understanding of underlying, predisposing ocular or systemic events. These latter glaucomas may be unilateral or bilateral, and some may have a genetic basis, whereas others are acquired.
In reality, the concept of primary and secondary glaucomas largely reflects our incomplete understanding of the pathophysiologic events that ultimately lead to glaucomatous optic atrophy and visual field loss. As our knowledge of the mechanisms underlying the causes of the glaucomas continues to expand, the primary and secondary classifications become increasingly artificial and inadequate. Furthermore, glaucomas caused by developmental anomalies of the anterior chamber angle do not fit neatly into either category. For these reasons, we recommend replacing traditional concepts with a new scheme that provides a better working foundation for the concepts of mechanism, diagnosis, and therapy that will shape the management of the glaucomas for the foreseeable future. This classification is used in this text for the discussion of the various forms of glaucoma.
Stages of Glaucoma
Glaucomas can be considered to consist of five stages:
- Stage 1—initiating events
- Stage 2—structural alterations
- Stage 3—functional alterations
- Stage 4—retinal ganglion cell and optic nerve damage
- Stage 5—visual loss
The initiating events (stage 1) include the condition or series of conditions that set in motion the chain of events that may eventually lead to optic nerve damage and visual loss, but which precede any pathologic or physiologic alterations related to aqueous humor dynamics or optic nerve function. Structural alterations (stage 2) are those tissue changes that precede, but may eventually lead to, alterations of aqueous humor dynamics or optic nerve function. Functional alterations (stage 3) are those physiologic abnormalities that may lead indirectly or directly to optic nerve damage. Optic nerve damage (stage 4) represents the loss of retinal ganglion cells and their associated axons as a result of the events in stage 3, which eventually leads to progressive loss of vision (stage 5). The first three stages can be further subdivided into events that are pressure related and those that are pressure independent (Table 7.1).
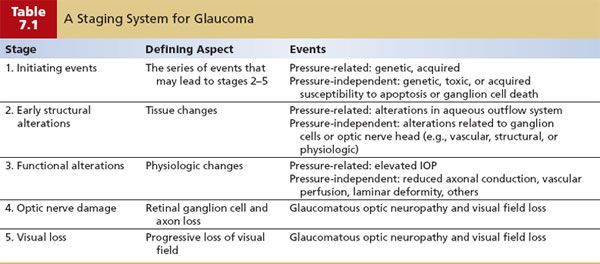
In the pressure-related subdivision, the initiating events (stage 1) are the conditions that may lead to structural alterations in the aqueous outflow system. In some glaucomas, this could be a genetic defect with an associated protein abnormality; in other glaucomas, this may be due to acquired events, such as trauma, inflammation, or a retinal vascular disorder, some of which may also have a genetic predisposition or may be indirectly influenced by genetic disorders. The structural alterations (stage 2) are tissue changes in the outflow system, which may lead to increased resistance to aqueous outflow and subsequent elevation of the IOP. Such changes might be subtle alterations in the endothelial cells or extracellular matrix of the trabecular meshwork or more obvious obstructive mechanisms, such as membranes over the anterior chamber angle, scar tissue in the meshwork, intertrabecular debris, or developmental anomalies. These changes can sometimes be detected by gonioscopy. The functional alterations (stage 3) include obstruction to aqueous outflow that is sufficient to increase the IOP, which (in pressure-related glaucoma mechanisms) may lead to the glaucomatous optic neuropathy (stage 4) and eventually to progressive loss of visual field (stage 5). Another emerging possibility is that cerebrospinal fluid pressure may be abnormally low or high, thus affecting the translamina cribrosa pressure gradient, weakening structural support to axons (as described in Chapter 4).
The specific events in the pressure-independent subdivision are not as well understood and are, in large part, only speculative. The initiating events (stage 1) probably have a genetic basis, however, with alterations in proteins that may lead to structural changes directly related to the ganglion cells or optic nerve head. The structural alterations (stage 2) may be subtle tissue changes in blood vessels supplying the optic nerve head or in supportive elements of the lamina cribrosa or, likely, in additional ways that are not yet understood. The functional alterations (stage 3) may be reduced axonal conduction, vascular perfusion to axons in the optic nerve head, or a progressive deformity of the lamina cribrosa that may lead (alone or in conjunction with a relative IOP elevation) to glaucomatous optic neuropathy (stage 4) and subsequent loss of visual field (stage 5).
Although our knowledge of the first three stages that lead to optic nerve damage and our ability to detect and treat these events are incomplete for most glaucomas, there are some glaucomas for which we have not only a partial knowledge of the events in each stage but also treatments for early intervention at stage 1. In neovascular glaucoma, for example, an initiating event (stage 1) may be a central retinal vein occlusion, which can lead to release of vascular endothelial growth factor and other cytokines that may lead to a structural alteration (stage 2) in the form of a fibrovascular membrane over the anterior chamber angle, which may eventually cause a functional alteration (stage 3) by obstructing aqueous outflow with a rise in IOP, which usually leads to optic nerve damage (stage 4) and eventual loss of visual field (stage 5). An understanding of this sequence provides a rational basis for early intervention with panretinal photocoagulation at stage 1 in selected patients. Such an approach to diagnosis and management should be the ultimate goal for all forms of glaucoma. As the initial events for an increasing number of the glaucomas become known, a complete classification scheme may eventually be developed on the basis of these initial events. However, until continued research provides the answers to these gaps in our knowledge, the etiologic classification scheme shown in Table 7.2 can be developed only partially.
Chronic Open-Angle Glaucomas
This category of glaucomas constitutes at least half of all the glaucomas and has been referred to by various names, including primary open-angle glaucoma, chronic open-angle glaucoma, and chronic simple glaucoma. To de-emphasize use of the “primary” and “secondary” terminology in glaucoma, the term chronic open-angle glaucoma is used in this text. A more appropriate term, however, might be idiopathic open-angle glaucoma, because our failure to provide more precise terminology stems from our lack of knowledge regarding the related mechanisms.
Although the initial events leading to chronic open-angle glaucoma are unknown, there is mounting evidence that inherited susceptibilities lead to increased resistance to aqueous outflow and increased vulnerability of the optic nerve head to a particular IOP level.

Pupillary Block Glaucoma
Among the so-called primary angle-closure glaucomas, the most common variation is pupillary block glaucoma. The term pupillary block glaucoma is best reserved for situations with evidence of optic nerve damage related to the angle-closure mechanism. A considerable amount of information is available regarding the initial events and mechanisms of outflow obstruction in pupillary block glaucoma. Therefore, there is no basis for distinguishing this condition—considered a primary glaucoma—from other disorders previously classified as secondary glaucomas. Pupillary block glaucoma may be divided into acute and subacute forms, although these forms merely represent different clinical manifestations, which can both occur at different times in the same patient. A third form, called chronic angle-closure glaucoma, is characterized by the presence of peripheral anterior synechiae. With a subset of chronic angle-closure glaucomas called creeping angle-closure glaucoma, peripheral anterior synechiae slowly advance forward circumferentially, making the iris insertion appear to become more and more anterior. With another form called combined mechanism glaucoma, IOP elevation persists after peripheral iridotomy for the angle-closure component, despite an open, normal-appearing anterior chamber angle. Some classification schemes have included the plateau iris syndrome with primary angle-closure glaucomas, although recent studies of the mechanism suggest that it might more appropriately be included with glaucomas associated with disorders of the iris and ciliary body (1).
Developmental Anomalies of the Anterior Chamber Angle
Numerous developmental disorders associated with anomalies of the anterior chamber angle can lead to IOP elevation. The initial event is probably a genetic defect in most cases, although some cases may stem from an acquired, intrauterine insult. The developmental anomaly may be a high insertion of the anterior uvea in the trabecular meshwork, incomplete development of the meshwork or Schlemm canal, or broad iridocorneal adhesions. One group of developmental glaucomas was previously classified with the primary glaucomas because it has no apparent consistent association with other ocular or systemic anomalies. This group includes congenital, or infantile, glaucoma and juvenile open-angle glaucoma, which differ primarily by the age of onset. Other conditions in this category have a wide range of associated ocular and systemic developmental abnormalities. Additional developmental disorders, such as those associated with the vitreous and retina, may lead to glaucoma, usually by an angle-closure mechanism; in the present scheme, these disorders are classified as glaucomas associated with abnormalities of a particular ocular structure (e.g., persistent hyperplastic primary vitreous).
Glaucomas Associated with Other Ocular Disorders
It is this large group of glaucomas that was previously classified as secondary glaucomas. In some cases, the initial events involve an abnormality of a specific ocular structure, such as the corneal endothelium, iris, ciliary body, lens, retina, choroid, or vitreous. In other cases, the initial events may involve a tumor, inflammation, hemorrhage, or accidental or surgical trauma. Many of these initial events are influenced by an inherited susceptibility, whereas others are acquired. Each of these broad categories of glaucoma usually contains subdivisions, based on different series of events that eventually lead to outflow obstruction.
A Note on Molecular Etiology
Now that several genes have been localized for various glaucomas, the hope of being able to reclassify these disorders on the basis of molecular etiology is being realized (2). Although separating patients with adult-onset chronic open-angle glaucoma on the basis of phenotype is difficult, the molecular classification has already allowed us to recognize several genetic forms of glaucoma. Characterizing additional genes and mutations will be extremely helpful in classifying disease in individual patients, with a view to being able to provide tailored prognostic and therapeutic information (as discussed in Chapter 8). Some glaucomas may be associated with more than one gene, such as the Axenfeld–Rieger syndrome, which appears to be caused by at least three different genes located on chromosomes 4, 6, and 13, once again underscoring genetic heterogeneity (3–6). Others are non-Mendelian or complex, and probably involve more than one gene plus environmental factors. An example is exfoliative glaucoma, in which polymorphisms of the LOXL1 gene are strongly associated with this condition in multiple populations (see Chapter 15).
In addition, molecular genetics is helping to associate seemingly disparate diseases. For example, Rieger syndrome and iris hypoplasia can arise from mutations in the same gene on 4q25 (PITX2) (7). Similarly, juvenile open-angle glaucoma and iridogoniodysgenesis can be caused by mutations in the FKHL7 gene on 6p25 (8,9). Improved understanding of the molecular etiology of various glaucomas will permit detailed reclassification of these disorders.
CLASSIFICATION BASED ON MECHANISM
An understanding of the initial events in each form of glaucoma will eventually allow for an improved classification system and a rationale for early glaucoma intervention. Until that information becomes available, however, most treatment strategies will continue to focus on IOP and depend on an understanding of the mechanisms of aqueous outflow obstruction.
As noted previously, one disadvantage of a classification based on the mechanism of aqueous outflow obstruction is that it ignores the causes unrelated to pressure. In addition, many of the glaucomas have more than one mechanism of outflow obstruction at different times in the course of the disease. As a result, some glaucomas must be classified under more than one mechanistic heading. It is for this reason that a classification based on initial events, rather than mechanisms of outflow obstruction, is used for organizing the chapters on clinical forms of glaucoma in this text.
On the other hand, the mechanistic classification has distinct advantages. First, our understanding of the mechanisms of aqueous outflow obstruction is in many cases more complete than our knowledge of the initial events. Second, because most current treatment strategies are directed at reducing IOP, an understanding of the mechanism that leads to aqueous outflow obstruction is important in developing a rationale for controlling the pressure in each form of glaucoma.
Barkan (10) first recognized the distinction between open-angle and closed-angle forms of glaucoma, which led to the basis for the mechanistic classification of the glaucomas (Fig. 7.1). A third group of glaucomas that does not fit well into either the open- or closed-angle mechanisms is the developmental glaucomas. The mechanistic classification, therefore, can be divided into three categories: (a) open-angle glaucoma mechanisms, (b) angle-closure glaucoma mechanisms, and (c) developmental anomalies of the anterior chamber angle (Table 7.3 and Fig. 7.2).

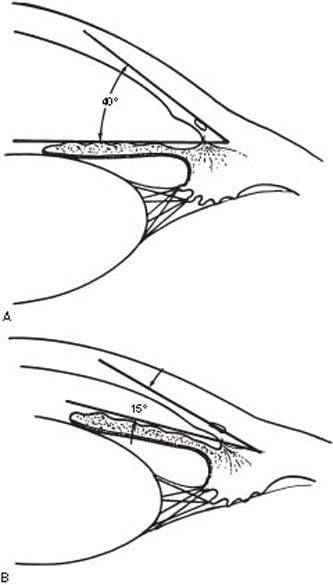
Figure 7.1 The angle of the anterior chamber is formed by the cornea and iris. A: The typical configuration in open-angle forms of glaucoma. B: The narrow angle that typically precedes most forms of angle-closure glaucoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


