Principles of Medical Therapy and Management
This chapter covers the management of the patient with glaucoma using an evidence-based approach. The following factors will be considered: (a) making an accurate clinical diagnosis; (b) assessing the stage of disease; (c) assessing the risk factors for disease progression; (d) understanding the patient’s access to health care and related factors; (e) considering the patient’s lifestyle, health status, and life expectancy; and (f) implementing a treatment strategy on the basis of these factors and other considerations. All of these factors influence how aggressive the physician should be to achieve the target intraocular pressure (IOP) range to minimize the progression of glaucomatous optic neuropathy. In addition, given the current resources for information and technology through the Internet, literature, glaucoma support groups, and other sources, patients are better informed today (although sometimes misinformed) about their glaucomatous conditions. It is essential to make the patient a part of the team in his or her care, which includes discussion of treatment options from medical to laser or surgical interventions.
GATHERING EVIDENCE TO EVALUATE THE PATIENT
After gathering the data on the patient and making the clinical diagnosis (as described in the preceding chapters), the physician must understand the evidence from the results of major epidemiology studies (see Chapter 9) and glaucoma clinical trials to develop a management plan for the individual patient. Four major prospective National Institutes of Health–sponsored glaucoma clinical trials have shown that lowering IOP is important for “protecting” the susceptible optic nerve in patients with glaucoma (see Chapters 10 and 11, and the National Eye Institute’s Web site: www.nei.nih.gov/neitrials/topics.asp#glaucoma). In addition, another glaucoma clinical trial reported on the benefit of reducing IOP in patients with normal-tension glaucoma (1). These large-scale randomized clinical trials were designed to study outcomes in a cohort of patients with different forms and stages of chronic open-angle glaucoma (COAG) or suspected glaucoma. Results of these studies can be used as we recommend management and treatment for the individual patient. The results are summarized below.
Ocular Hypertension Treatment Study
The Ocular Hypertension Treatment Study (OHTS) evaluated the safety and efficacy of topical ocular hypotensive medication in delaying or preventing the onset of COAG in participants with no initial glaucomatous damage and an IOP between 24 and 32 mm Hg (2,3). An important aspect of the design of this study was to evaluate the risk for glaucoma in patients of both European ancestry and black African descent. The 1636 participants were randomly assigned to observation or treatment with a target IOP reduction of 20%. The investigators found that topical ocular hypotensive medications can delay the onset of COAG in patients with elevated IOP, although not all patients with ocular hypertension require treatment. In the multivariate analysis, race was not a significant risk factor, which can be explained by the fact that African Americans in OHTS had overall thinner central corneas and larger optic discs. The clinical risk factors that increased the risk for glaucoma included older age, large cup-to-disc ratio, early visual field loss, thin central cornea, and elevated IOP.
Early Manifest Glaucoma Trial
The Early Manifest Glaucoma Trial (EMGT) assessed treatment versus observation without treatment in patients with early glaucoma and found that progression was less frequent and occurred later in treated patients (4,5). The EMGT demonstrated that the following factors were predictors of glaucoma progression: elevated IOP, older age, bilaterality, exfoliation, disc hemorrhages, and relatively thin central cornea. In addition, lower systolic perfusion pressure, lower systolic blood pressure, and cardiovascular disease history emerged as new predictors, suggesting a vascular role in glaucoma progression (6).
Collaborative Initial Glaucoma Treatment Study
The Collaborative Initial Glaucoma Treatment Study (CIGTS) evaluated the efficacy and safety of surgery versus medical treatment in patients with newly diagnosed, early glaucoma and found similar outcomes with the two treatment approaches (7). Investigators also found that when patients receive a glaucoma diagnosis, they may have symptoms that are not elicited by routine clinical testing, requiring discussion with the patient to reduce worries and unnecessary concerns about blindness and improving their quality of life (8).
Advanced Glaucoma Intervention Study
The Advanced Glaucoma Intervention Study (AGIS) investigated two surgical sequences in patients with advanced glaucoma. One sequence began with argon laser trabeculoplasty followed by trabeculectomy, if necessary; the other began with trabeculectomy and was followed by argon laser trabeculoplasty if the trabeculectomy failed. The study provided a weak suggestion that an initial argon laser trabeculoplasty delays the progression of glaucoma more effectively in black patients than in white patients (9). Retrospective evaluation of the study data suggest that consistent, low IOPs with minimal IOP variation is associated with reduced progression of visual field in patients with advanced glaucoma (10).
APPROACH TO THE PATIENT WITH GLAUCOMA
These clinical trials and other evidence from epidemiology studies have led to a better understanding of the management of patients with ocular hypertension and with early and advanced glaucoma (11,12). Given these evidence-based risk factors, it is important to consider that an individual patient may not be comparable with the study participants, who had to meet specific inclusion and exclusion criteria to be enrolled in the clinical trials. Thus, the recommendations to treat patients with various forms and disease stages of glaucoma should be guided by the results of these important clinical trials, while keeping in mind potential differences between the individual patient and the average clinical trial participant (13). Overall, when treating the individual patient, physicians should remember that IOP is a surrogate clinical endpoint and that the long-term goal is to preserve vision and the best quality of life for the patient. In the perspective of glaucoma and the vision spectrum (Fig. 27.1), our approach must change along the vision continuum to accommodate the knowledge gained from epidemiology studies, clinical trials, and long-term clinical experience.
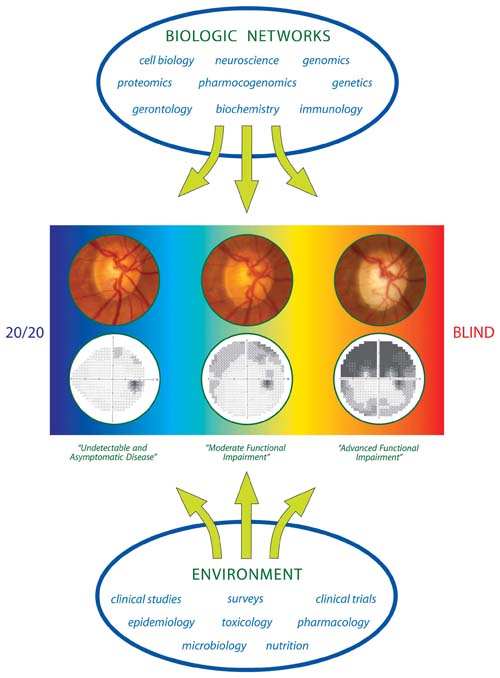
Figure 27.1 There is a broad spectrum of glaucoma, ranging from asymptomatic to advanced disease with optic nerve damage and visual field loss. This case demonstrates progression of glaucoma based on right optic disc photos and right visual fields over 18 years despite medical and surgical treatments with IOP reduction and fluctuation between 7 and 13 mm Hg. For such a patient, we need further advances beyond the risk factors identified in well-designed clinical trials. Such advances will develop from the research areas represented in the “biologic networks” and “environment.” (Modified from Moroi SE, Richards JE. Glaucoma and genomic medicine. Glaucoma Today. 2008;1: 16–24.)
The New Patient
A new patient may or may not know whether he or she has glaucoma or know the type of glaucoma. If the patient has not been seen by an ophthalmologist previously, a full eye examination is indicated. This entails addressing the chief complaint; obtaining an ocular and medical history; testing visual acuity and refraction; performing tonometry and pachymetry; conducting an external examination with evaluation of the pupillary reaction, slitlamp biomicroscopy, and gonioscopy; assessing the retina and optic nerve head with photographic documentation; and testing the visual field.
When making a new diagnosis of glaucoma, physicians should explain the basics of glaucoma to the patient and help him or her understand that glaucoma can lead to irreversible blindness, but that this can be prevented with early diagnosis and proper care. It is also important to explain the type of glaucoma and to show the patient photographs of the optic disc or a computerized topographic analysis report and copies of the visual fields. It is worth explaining to patients that, unless the glaucoma is diagnosed in the very advanced stage, the prognosis for retaining their vision is excellent, especially with a good understanding of their disease combined with adherence to recommended treatment and frequent follow-up visits. In one study, white patients with glaucoma had an approximately 25% chance of monocular blindness and a 9% chance of bilateral blindness in 20 years, even if the glaucoma remained uncontrolled (14).
The Established Patient
For the established patient, the critical components of continuity of care are to evaluate adherence to glaucoma medical treatment, evaluate tolerance to the treatment, and assess stability of the optic nerve head and visual function. If the patient has had a surgical intervention, the surgical site should be examined carefully for signs of tissue breakdown or infection. Many patients are more interested in their IOP levels, often remembering the numbers from the previous visit. Although sharing this information with the patient is helpful, a discussion of the complete clinical picture allows the patient to be better educated, which may result in better adherence to the treatment plan.
THE TREATMENT PLAN
In general, the overall goal to managing all patients with glaucoma is to preserve visual function while maintaining the best possible quality of life. This goal can be achieved by preventing or slowing the progression of glaucomatous damage by lowering IOP to a level at which further damage is minimal. Although not universally accepted, guidelines for glaucoma treatment are available from various professional societies (e.g., American Academy of Ophthalmology, at http://one.aao.org/CE/PracticeGuidelines/PPP.aspx; International Council of Ophthalmology, at www.icoph.org; European Glaucoma Society, at www.eugs.org; and South East Asia Glaucoma Interest Group, at http://seagig.org), with approaches for managing the various clinical forms of glaucoma or ocular hypertension. In the medical management of a patient with glaucoma, physicians should consider when to initiate treatment, how to start, how to follow the patient, when to change the treatment, and when to move on to surgical intervention.
When to Treat
To avoid unnecessary treatment, physicians must decide whether treatment is really indicated. When elevated IOP is present without glaucomatous damage (i.e., ocular hypertension), the physician must evaluate the risk factors for progression to glaucoma before deciding whether to treat. Most patients who have ocular hypertension appear to do well without treatment (2). When the patient presents with established glaucomatous damage or dangerously high IOP, the indication to initiate treatment is usually clear.
How to Start
Initiating treatment involves establishing the target IOP or IOP range, selecting the appropriate medication, educating and instructing the patient, and establishing the efficacy and safety of the treatment at follow-up evaluations.
Establishing the Target Pressure
Elevated IOP is the most important causative risk factor for glaucoma development and progression, and it is the only one for which we have proven treatment. However, no single pressure value is appropriate for all patients. Rather, the physician must establish a target pressure or target IOP range that can prevent further glaucomatous damage. The IOP target is based on the status of the optic nerve head and other risk factors for progression. In most cases, reducing the IOP by 20% to 30% from baseline is recommended, which should result in a target IOP in the middle to high teens (mm Hg) for eyes with minimal damage (e.g., early neural rim thinning without visual field loss), the low to middle teens for eyes with moderate damage (e.g., cupping to the disc margin in one quadrant with early field loss), and the high single digits to low teens for eyes with advanced damage (e.g., extensive cupping and field loss).
Other risk factors that should be considered in establishing the target pressure include the central corneal thickness, with thin corneas having been identified as a major risk factor for patients with ocular hypertension (3,15). Older age, family history of glaucoma, African heritage, and myopia are also indicators of an increased risk for the presence and progression of COAG (11), whereas Asian heritage and hyperopia suggest an increased risk for angle-closure glaucoma (16). Growing evidence suggests that vascular factors, such as ocular ischemia and vasospastic disease, may contribute to the pathogenesis and increased risk for glaucoma (6,17,18), especially in normal-tension glaucoma (19). (Risk factors for specific forms of glaucoma are considered in more detail in Chapter 9.)
Selecting Initial Medication
In the United States, initial medical therapy remains the standard for most patients with newly diagnosed glaucoma. The treatment of most forms of open- and closed-angle glaucoma includes the use of topical and occasionally orally administered agents that lower IOP by enhancing aqueous outflow, reducing aqueous production, or both. Currently, five different classes of glaucoma medications are available for the long-term treatment of glaucoma: prostaglandin-related agents, or hypotensive lipids; β-adrenergic receptor antagonists, or β-blockers; adrenergic receptor agonists; carbonic anhydrase inhibitors; and cholinergics, or miotic agents. An overview of currently available glaucoma drugs is provided in Figure 27.2 and their basic mechanism of action on aqueous humor dynamics is shown in Figure 27.3.
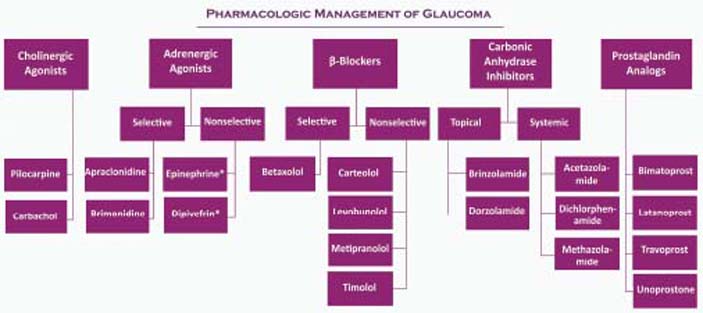
Figure 27.2 Medical therapy options for glaucoma treatment. Agents with an asterisk are no longer commercially available.
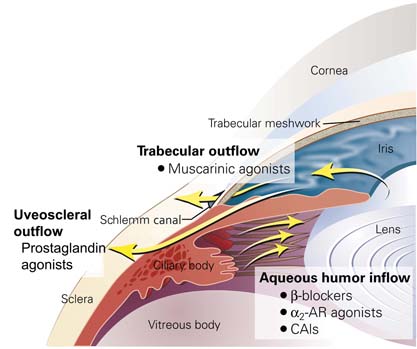
Figure 27.3 Schematic of anterior portion of the human eye highlighting anatomy relevant to current glaucoma therapeutics. The aqueous humor is produced by the ciliary body and secreted by the ciliary body epithelial bilayer into the posterior chamber (three small arrows indicate the aqueous humor inflow pathway). The aqueous humor bathes and nourishes the crystalline lens and then circulates into the anterior chamber through the pupil (indicated by the single arrow going across the iris). The aqueous humor leaves the eye through the trabecular meshwork and into the Schlemm canal (trabecular outflow) and through the peripheral base of the iris, into the ciliary body, and through the sclera (uveoscleral outflow). Current glaucoma medical therapy modulates intraocular pressure by decreasing aqueous humor inflow (β-blockers, α2-adrenergic receptor [AR] agonists, and carbonic anhydrase inhibitors [CAIs]), by enhancing trabecular outflow (muscarinic agonists), or by enhancing uveoscleral outflow (prostaglandin agonists). (Modified from McLaren NC, Moroi SE. Clinical implications of pharmacogenetics for glaucoma therapeutics. Pharmacogenomics J. 2003; 3:197–201.)
Exceptions to initiating glaucoma therapy with medications include patients with very high IOPs, which pose an immediate threat to vision; a history of medical treatment without success or with intolerable side effects; and problems with adherence to therapy. Acute angle-closure glaucomas and many forms of childhood glaucoma are managed with initial or early surgery.
One may initiate therapy using a uniocular trial; however, some investigators have recently questioned this approach (20). If the fellow untreated eye is used as a control, then physicians must keep in mind that some antiglaucoma eyedrops may produce a small consensual reduction in IOP in the untreated eye (21). Once-daily dosing has the benefits of increased adherence and possibly decreased side effects. However, if once-daily medication is not achieving the target IOP, then reassessment of treatment may be the next step. In general, the therapeutic goal is to use the least amount of medication that will accomplish the desired therapeutic effect with the fewest adverse reactions and that is affordable for the patient.
Pharmacokinetics of Topical Drugs
In using topical glaucoma medications, one should consider the basic pharmacokinetics, which deals with the absorption, distribution, metabolism, and elimination of an administered drug (22). The availability of these topically applied pharmacologic agents at the receptor site is influenced by (a) drug kinetics in the conjunctival cul-de-sac, (b) corneal and transconjunctival–scleral penetration, and (c) the distribution and rate of drug elimination within the eye (22).
Drug Kinetics in the Conjunctival Cul-de-Sac
Following topical instillation, a medication first mixes with the tears in the cul-de-sac, which normally contains 7 to 9 µL of fluid and has a maximum capacity of about 30 µL. The precorneal tear film is a dynamic trilaminar fluid layer conceptually described as an outer lipid layer, a middle aqueous layer, and an inner mucin layer (23,24). The drop size of commercial glaucoma medications ranges from 25.1 to 56.4 µL, with an average of 39 µL (25). Therefore, as much as one half of the medication may spill out from the lids at the time of instillation.
A large percentage of that which remains in the cul-de-sac enters the lacrimal drainage system as a result of the pumping action created by blinking of the eylids. The rate of loss of the drug in the tears is rapid, with the peak time occurring within the first few minutes after instillation. This drug loss not only reduces the amount of drug available for the pharmacologic effect within the eye but also increases the potential for systemic side effects by absorption into the systemic circulation via the nasopharyngeal mucosa. The degree to which this occurs can be influenced by nasolacrimal occlusion, which is discussed later in this chapter. Thus, the bioavailability of the drug for corneal and conjunctival–scleral penetration is significantly influenced by the degree to which a drug saturates the tear film and by the retention time in the cul-de-sac.
Corneal and Transconjunctival–Scleral Penetration
In order for the drug to reach the intraocular targets, the drug must penetrate the cornea and anterior conjunctival and scleral tissues (26). The cornea may be conceptualized as a “lipid–water–lipid” sandwich in that the lipid content of the epithelium and endothelium is approximately 100 times greater than that of the stroma (27). Consequently, the epithelium and endothelium are readily traversed by lipid-soluble substances (i.e., compounds in a nonionized or nonelectrolyte form) but are impermeable to water-soluble agents (i.e., ionized compounds or electrolytes). These permeability characteristics create a selective barrier, in that only drugs that can exist in both a water-soluble and lipid-soluble state are able to penetrate the intact cornea. This has been referred to as the differential solubility concept. On the basis of these biologic properties of the cornea, drugs tend to concentrate in various layers of the cornea. Some of the drug may be degraded at this level while another portion is temporarily stored in the cornea. The cornea therefore acts as a depot and a limiting factor for transfer of the drug to the aqueous humor (28). Another consideration for drug design and delivery takes advantage of the endogenous enzymes in the corneal epithelium that activate some glaucoma prodrugs into their active form, such as bimatoprost and presumably the other prostaglandin-like agents (29).
Intraocular Factors Influencing Drug Bioavailability
After penetrating the cornea, conjunctiva, and sclera, the drug must distribute to the appropriate structures in the anterior segment of the eye. The bioavailability of the drug is affected by local tissue binding (30,31), by local tissue metabolism, and by diffusion into the vascular system via the aqueous outflow system. In vitro studies with synthetic melanin revealed a binding rate up to 85% for β-blockers, compared with 40% for pilocarpine, 50% for epinephrine, and almost none for prostaglandins (32). These findings corresponded to results of in vivo studies, in which timolol and pilocarpine had a greater IOP-lowering effect in albino than pigmented rabbits, whereas prostaglandins had the same effect in both groups of animals (32). Another effect of tissue–drug binding is a potential drug depot effect whereby β-adrenergic inhibitors can be released very slowly from the pigmented uveal tissues, accounting for the longer duration of effect in pigmented eyes (33). Other drugs, including pilocarpine, are metabolized in the ocular tissues (34). Thus, the small portion of the instilled drop that escapes extraocular or intraocular elimination, tissue binding, or inactivation may finally reach the appropriate target where it exerts its pharmacologic effect.
Formulation of Topical Drugs
The pharmacokinetics of a particular drug can be greatly influenced by the manner in which it is formulated. This includes the vehicle, pH, concentration, and additives of the formulation. The vehicle in which a drug is delivered affects the amount of medication available for ocular penetration by influencing the rate of drug loss in the tears, the precorneal tear film saturation, and the length of time that the drug remains in contact with the cornea (35). Commonly used vehicles are soluble polymers, including methylcellulose and polyvinyl alcohol. They reduce the initial rapid drainage and prolong the drug–cornea contact time presumably by increasing tear viscosity, providing solution homogeneity (uniform suspension of drug particles in solution), and reducing surface tension.
Ointments significantly increase drug bioavailability by reducing loss in the tears, inhibiting dilution by the tears, providing a higher effective concentration of the drug, and increasing tissue contact time (36). They are limited, however, by interference with vision and aesthetic considerations. Soluble gels, emulsions, and suspensions have also been examined. Although no longer available, pilocarpine gel was formulated as a high-viscosity acrylic soluble gel that delivered a 24-hour pilocarpine dose following a single, nighttime application in the cul-de-sac (37).
Liposomes may be designed as small, homogenous unilamellar structures composed of phospholipids and water, which can be suitable for incorporating topical medications (35), but none is currently in clinical use. Various solid materials have been evaluated for their ability to release a drug at a sustained rate from a location on the cornea or in the conjunctival cul-de-sac. Although no longer available, a diffusional system released pilocarpine from between two polymeric membranes (38), which delivered the drug with zero-order kinetics because the amount of drug delivered per unit time was independent of the amount left undelivered.
As discussed earlier, the lipid–water solubility ratio (or partition coefficient) of a compound influences corneal penetration. Design of topically applied drugs must take into account both the ionized (i.e., more water-soluble) and nonionized (i.e., more lipid-soluble) forms. The two forms of the drug exist in equilibrium, with one form penetrating a particular layer of the cornea and then replenishing the other form to maintain the equilibrium. The pH at which a drug is formulated influences this ratio, with weak bases (which include most glaucoma medications) being absorbed through the cornea at a higher pH, whereas weak acids are absorbed better at a lower pH. Solution pH also affects drug stability and patient comfort on instillation. Fortunately, most weak bases at physiologic pH of 7.4 exist predominantly in the nonionized form (39).
Compounds with a molecular weight greater than 500 g/mol have poor corneal absorption. However, this is not a major factor because most ophthalmic drugs have a lower molecular weight. There is an optimal drug concentration to increase bioavailability of the drug at the target site, but this is balanced by the concern that at higher drug concentrations, a greater amount of drug is lost in the lacrimal drainage system, which increases the potential for systemic side effects.
The topic of additives used in glaucoma medications has received much attention relative to concerns of ocular surface irritation, dry eye, and long-term safety to the ocular tissues (40–43). The main additive is benzalkonium chloride, which not only serves as a “preservative” by providing bacteriostatic activity but also influences corneal penetration by decreasing the surface tension of nonpolar drugs, allowing them to mix more with the tear film, leading to enhanced corneal absorption. (More specific information on modifications in the additives is discussed in the individual chapters discussing specific glaucoma medications.) Additives do not, however, prevent bacterial contamination, and physicians must educate patients on the proper handling of eyedrop dispensers.
Glaucoma Medications
Prostaglandins (Hypotensive Lipids)
The prostaglandin-like compounds, or “hypotensive lipids,” are the most recent class of drugs to be introduced for the long-term management of glaucoma. Although high concentrations of prostaglandins are associated with ocular inflammation and elevated IOP, very low concentrations effectively reduce the pressure by increasing uveoscleral outflow (44). In the United States, the first of these drugs to be released was latanoprost, followed by unoprostone, travoprost, and bimatoprost. (For further details reg arding mechanism of action, side effects, and cost issues, see Chapter 28.)
β-Blocking Drugs
Of all the glaucoma drug classes, we have the most experience with topical β-blockers, the first of which was timolol maleate, which was approved for use by the U.S. Food and Drug Administration in 1978. These drugs, which lower IOP by reducing aqueous production, are available as nonselective (i.e., blocking β1 and β2 receptors) or selective (i.e., primarily blocking β1 receptors) agents. Concentrations of β-blockers used in treating glaucoma range from 0.25% to 1.0% and are usually instilled once or twice per day. The β1-selective β-blocker betaxolol causes fewer pulmonary and cardiovascular side effects, but it is less effective in lowering IOP than the nonselective β-blockers. (For further details regarding mechanism of action, side effects, and cost issues, see Chapter 29.)
α-Selective Adrenergic Drugs
The α2-adrenergic agonists lower IOP primarily by reducing aqueous production. Apraclonidine in a 1% concentration was first used to prevent IOP spikes after anterior segment laser procedures but was later approved for treatment of chronic glaucoma. Apraclonidine in a 0.5% concentration can also be used short term in patients with glaucoma receiving maximally tolerated medical therapy who require additional reduction in IOP; however, its long-term use is limited by frequent allergic reactions.
Brimonidine is more selective than apraclonidine and appears to elicit a lower incidence of ocular allergic reactions. Like apraclonidine, brimonidine can be used to prevent IOP spikes after argon laser trabeculoplasty. It has been reported to cause clinically significant pulmonary depression in children (45–48). (For further details on mechanism of action, side effects, and cost issues, see Chapter 30.) Epinephrine compounds are no longer available to treat glaucoma, given the availability of newer drugs that are more effective and have fewer side effects.
Carbonic Anhydrase Inhibitors
This is the only class of systemic medications that can be used both topically and systemically for the treatment of glaucoma. These agents lower the IOP by reducing aqueous production through inhibition of the carbonic anhydrase enzyme. Acetazolamide is the prototype carbonic anhydrase inhibitor and may be administered orally or intravenously. Methazolamide is the other oral carbonic anhydrase inhibitor. The topical carbonic anhydrase inhibitors dorzolamide, 2%, and brinzolamide, 1%, are available for the long-term treatment of glaucoma. (For further details regarding mechanism of action, side effects, and cost issues, see Chapter 31.)
Miotic Agents
Pilocarpine is the oldest known therapy for glaucoma, which was introduced in the 1870s. It is a cholinergic agent most commonly used in the treatment of open-angle glaucoma. It is occasionally used in solution with concentrations ranging from 0.5% to 6%, typically administered four times daily. (For further details regarding mechanism of action, side effects, and cost issues, see Chapter 32.)
Educating and Instructing the Patient
The physician is ultimately responsible for educating the patient; the physician or another member of the health care team must discuss the basic aspects of the disease, prognosis, and treatment. This effort is an important investment for the physician–patient relationship. In addition to the physician, office personnel and educational resources should also be used. The most important ancillary individual is the office nurse or technician, who should be able to restate and expand on what the physician has said, instruct the patient on the instillation of eyedrops, work out the therapeutic regimen, and provide the necessary reinforcement of these matters at each follow-up visit. That individual should also be able to answer questions about use of the medications or possible side effects. Other important resource materials are available from several reliable health-related organizations. In some centers, classes or meetings are provided for patients to broaden their understanding of their condition. These are usually conducted by nurses, technicians, or social workers and may incorporate lectures, videos, question-and-answer sessions, or group discussions.
About the Disease
It is essential for patients to be made aware of their disease and its potential seriousness without creating undue apprehension. Patients should be told that they have glaucoma, what glaucoma is, and that it can lead to total and irreversible blindness but that the blindness can be prevented with proper treatment. It is not uncommon for patients to have been taking glaucoma medication for years but be unaware that they have glaucoma or relate it to blindness, whereas others may live in daily fear that they will inevitably go blind. The time taken to correct these misconceptions is one of the most important measures in preventing blindness and improving the general well-being of patients.
Why the Medications
Patients must understand that the purpose of taking their medications is to lower the IOP, which is the main treatable risk factor for slowing loss of vision from glaucomatous optic neuropathy. It should be made clear that this treatment will not improve their visual acuity and that any medication has potential side effects. Many patients stop using their eyedrops because “they did not seem to be helping.”
Poor adherence to recommended medical therapy is a major problem in the prevention of blindness from glaucoma. Most forms of glaucoma represent chronic diseases in which symptoms are mild or absent, treatment is prophylactic, and the consequences of stopping therapy are delayed, all of which are associated with poor adherence to medical therapy. In an older study of 184 patients in which adherence to pilocarpine treatment was monitored electronically in the medication bottle, participants administered 76.0 ± 24.3% of the prescribed pilocarpine doses, but they reported taking 97.1 ± 5.9% of the prescribed pilocarpine doses (49). Another important observation was that the rate of adherence was significantly higher in the 24-hour period preceding the return appointment than in the entire observation period. In a similar study involving 110 patients, patients administered 82.7 ± 19.0% of the prescribed timolol doses (range, 20% to 100%) (50).
What can be done to improve patient adherence to treatment? This question has received serious attention recently, and practical patient-centered communication skills using open-ended questions will best engage the patient to adopt adherent self-management behaviors (51). Once poor adherence is recognized, the cause is usually found to be related to (a) patient factors, (b) medication regimen, (c) provider factors, or (d) situational or environmental factors (52). Learning from the experience in health behavior and hypertension, we should strive to simplify the glaucoma medication regimen, be cost conscious, and educate the patient about the pathophysiology of glaucoma and the importance of various treatments for glaucoma (53).
The Therapeutic Regimen
When told to use eyedrops more than once daily, some patients may not appropriately space the doses unless properly instructed. It is important to work out a daily schedule, especially if patients are using multiple medications. If they are taking more than one drop at the same time of day, they may be instilling them so close together that they are diluting or washing each other from the cul-de-sac. Thus, the physician should instruct the patient to wait at least 5 minutes between instillation of the eyedrops. The physician and patient should work out a schedule that fits the patient’s daily activities and links the use of the eyedrops with specific daily functions, such as meals. It is advisable to record this schedule on a form that is large enough for the patient to see and can be kept in a convenient location (Fig. 27.4). It may also be helpful to make sure that the patient can distinguish between drugs by associating a drug with the color of the bottle top, although this is more difficult today because of a lack of standardized colors for different drug classes.
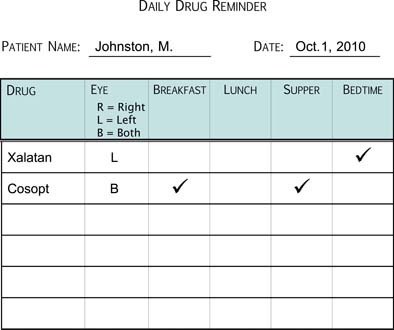
Figure 27.4 Example of a drug schedule that can be used to instruct the patient in the use of glaucoma medications and serve as a reminder for the patient.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


