Glaucomas Associated with Intraocular Hemorrhage
Intraocular hemorrhage is most commonly caused by trauma or surgery. Hyphemas may occur spontaneously in association with several ocular disorders, most of which are discussed in other chapters. Whatever the initial cause, intraocular hemorrhage frequently leads to intraocular pressure (IOP) elevation when the aqueous outflow channels become obstructed by blood in various forms. In this chapter, we consider the mechanisms and management of the blood-induced glaucomas and some specific causes of intraocular hemorrhage that are not covered in other chapters.
GLAUCOMAS ASSOCIATED WITH HYPHEMA
Blunt Trauma
A common source of hyphema, or blood in the anterior chamber, is blunt trauma. This usually results from a tear in the iris or ciliary body, causing bleeding from the small branches of the major arterial circle.
General Features
Young age and male sex appear to be risk factors for blunt ocular trauma. In one large series, 77% of the patients with traumatic hyphemas were younger than 30 years of age (1). In another large study, the annual incidence of traumatic hyphema was significantly increased among men, and sports-related injuries were identified as a cause for a recent rise in the incidence rate (2).
The initial clinical finding may be a microscopic hyphema, which is characterized by red blood cells circulating in the aqueous. In other cases, the quantity of blood may be sufficient to create a layered hyphema. These range in size from a small layer of blood in the inferior quadrant of the anterior chamber, which is the more common situation, to a total hyphema, in which the entire anterior chamber is filled with blood (Fig. 24.1). In most cases, the blood clears within a few days, primarily through the trabecular meshwork, and the prognosis is good unless the associated trauma has caused other ocular injuries. However, complications may occur during the postinjury course that can have devastating results.
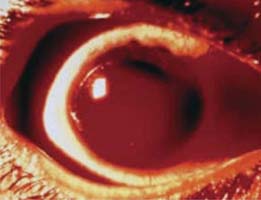
Figure 24.1 A total hyphema in an 11-year-old boy. Although initially normal, the IOP increased to 40 mm Hg within 24 hours. (From Crouch ER Jr, Crouch ER. Trauma: ruptures and bleeding. In: Tasman W, Jaeger EA, eds. Duane’s Clinical Ophthalmology. Vol. 4. Lippincott Williams & Wilkins; 2009:chap 61.)
Complications
Recurrent Hemorrhage
The reported frequency with which eyes rebleed after traumatic hyphema ranges from 4% to 35%, with a rate of fewer than 10% in most reported series (2–28). Rebleeding usually occurs during the first week after the initial injury, which is probably related to the normal lysis and retraction of the clot. Studies vary considerably on risk factors for recurrent bleeding. Some investigators have found no identifiable factors (22,23); others have observed that an increased frequency is associated with the size of the initial hyphema, the degree of reduced visual acuity, and delayed medical attention (24). Use of aspirin can increase the frequency of rebleeding (9,14,29). Elevated IOP is a risk factor for recurrent hemorrhage, but hypotony may also increase the risk for rebleeding (3,5,24). Studies have also suggested that black race is another risk factor for recurrent hemorrhage (21,30).
Although studies may differ regarding the frequency of rebleeding and the risk factors by which this complication can be predicted, nearly all reported series agree that recurrent hemorrhage, as compared with the initial hyphema, is associated with significantly more complications and a more-frequent need for surgical intervention.
Associated Glaucoma
Although IOP elevation may occur after the initial bleed, it is more common after a recurrent hemorrhage and constitutes the most serious complication of a traumatic hyphema. The incidence of glaucoma associated with a traumatic hyphema is partially related to the size of the hemorrhage. In one study of 235 cases, glaucoma occurred in 13.5% of the eyes in which the hyphema filled less than one half of the anterior chamber, in 27% of those with a bleed involving more than one half of the chamber, and in 52% of the eyes with a total hyphema (17). In another study, in addition to hyphema, factors that predicted chronically elevated IOP after blunt trauma included increased angle pigmentation, a higher baseline IOP, angle recession, and lens displacement and cataract (31). It is important to distinguish between a total hyphema with bright red blood and an “eight-ball” hyphema, characterized by dark red-black blood, because the latter carries a worse prognosis relative to associated glaucoma (4). In one series of 113 cases, IOP was elevated in one third of those with a rebleed but in all cases with “eight-ball” hyphemas (4).
The mechanism of pressure elevation is related to obstruction of the trabecular meshwork in most cases of traumatic hyphema. Although fresh red blood cells are known to pass through the conventional aqueous outflow system with relative ease, it appears to be the overwhelming numbers of cells, combined with plasma, fibrin, and debris, that may lead to a transient obstruction of aqueous outflow (Fig. 24.2) (32). On the basis of the effect of melanin on traumatic hyphema in rabbits, one study suggested that in darker-pigmented individuals, release of melanin into the anterior chamber with trauma may prolong the course of hyphema and affect the rate of rebleeding (33). In cases of eight-ball hyphema, it is presumably the formation of a clot, occasionally with degenerated red blood cells from an associated vitreous hemorrhage, that further impedes the outflow.
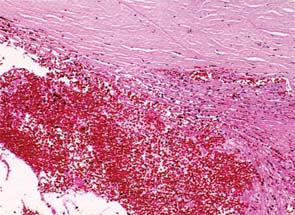
Figure 24.2 Red blood cells and their products of degeneration obstruct the trabecular meshwork in hyphema (hematoxylin–eosin, ×40). (From Callahan CE, Sassani JW. Pathology of glaucoma. In: Tasman W, Jaeger EA, eds. Duane’s Foundations of Clinical Ophthalmology. Vol. 3. Lippincott Williams & Wilkins; 2008: chap 19.)
Sickle cell hemoglobinopathies, including sickle cell trait, increase the incidence of IOP elevation in association with hyphema (34–36). Erythrocytes in these disorders have an increased tendency to sickle in the aqueous humor (34,35,37), and the elongated, rigid cells pass more slowly through the trabecular meshwork (38), leading to IOP elevation even with small amounts of intracameral blood (34,35). Even moderately elevated pressure may affect the optic nerve head more deleteriously in patients with sickle cell anemia than in other patients, possibly because of reduced vascular perfusion (34,35). Given that IOPs that would normally be considered safe to simply watch carefully (e.g., in the range of 20– to 30–mm Hg) can result in severe vision loss, such patients need to be observed diligently and treated more aggressively to keep IOP in the normal range. Another mechanism of glaucoma associated with sickle cell hemoglobinopathies is obstruction to aqueous outflow due to sickled erythrocytes in the Schlemm canal, which has been observed after blunt trauma and in one case with no antecedent trauma (36).
Diabetes mellitus may be associated with delayed clearing of blood from the anterior chamber. Erythrocytes from patients with diabetes have decreased deformability and increased adherence, resulting in delayed clearance time from the rabbit anterior chamber, compared with red blood cells from healthy human participants (39).
Corneal Bloodstaining
Corneal bloodstaining typically results from a prolonged total hyphema that is usually but not always associated with elevated IOP (40). This complication occurred in 6 of 289 patients (2%) with traumatic hyphema, all of whom had a recurrent total hyphema (38). The earliest pathologic event may be corneal endothelial decompensation associated with the passage of hemoglobin and hemoglobin products into the stroma (41). The cornea may initially have a red discoloration, which in rabbit studies is associated with extracellular hemoglobin particles and oxyhemoglobin (41). The hemoglobin is apparently phagocytized by keratocytes and degraded to hemosiderin (41,42). The cornea takes on a brownish discoloration at this stage, which is associated with methemoglobin in the stroma (41). Clearing of the corneal bloodstaining begins in the peripheral and posterior stroma, apparently because of diffusion of hemoglobin breakdown products out of the cornea, and may take up to 3 years to clear completely (42,43).
Management
Conservative Management of Hyphema
There is general agreement that the uncomplicated hyphema should be managed nonsurgically with the aims of accelerating resorption of the hyphema and minimizing rebleeding. Historically, bed rest and patching were advised for patients with hyphemas. However, little evidence supports these adjuncts to treatment. In most cases, all that is needed is limiting the patient’s ambulation, avoiding use of aspirin and nonsteroidal anti-inflammatory agents, and using a shield simply to protect the injured eye. Hospitalization is rarely necessary unless the hyphema is large, there is associated ocular trauma, sickle cell disease or trait is present, or the patient cannot be relied on to maintain limited activity or to return for recommended follow-up visits.
Acceleration of Hyphema Clearance
Various drugs have been used by some physicians to accelerate resorption of the hyphema, but no agent has proven to be efficacious for safely accelerating hyphema resorption. Rabbit studies have not supported the efficacy of atropine, pilocarpine, or acetazolamide use for this purpose (44–46), although the use of hyperosmotic agents may accelerate resorption of a clotted hyphema (47). Intracameral tissue plasminogen activator, a clot-specific fibrinolytic agent, has been shown to accelerate the clearance of experimental hyphema in rabbits, although it may also increase the risk of rebleeding (47–49).
Prevention of Rebleeding
Numerous drugs have also been evaluated regarding their ability to prevent rebleeding, with reported results that have been conflicting. Some investigators found that use of oral prednisone significantly reduced the rebleeding rate (8), whereas others found neither steroids nor estrogen to be of value in this regard (4,15). Antifibrinolytic agents (including tranexamic acid and aminocaproic acid (11–13,21,26–28)) have been used in an effort to minimize rebleeding by delaying the natural lysis of the clot. Most reports indicate that the use of either drug is associated with a significant reduction in rebleeds (11,13,26,28). Tranexamic acid has been evaluated in several series of traumatic hyphemas in children (28,50,51). In a study of hospitalized children, the rebleeding rate was 3% with use of tranexamic acid (25 mg/kg every 8 hours for 5 days), compared with 8% without use of antifibrinolytic therapy (28). One group of children with small hyphemas treated with systemic tranexamic acid therapy and limited activity at home had no recurrent hemorrhage (51). A randomized, placebo-controlled trial of 238 patients found tranexamic acid to be more effective than oral prednisolone or no treatment in preventing rebleeding (rebleed rates, 10%, 18%, and 26%, respectively) (52).
Some studies have found no significant difference between tranexamic or aminocaproic acid and placebo therapy (12,53). Aminocaproic acid is typically given as 100 mg/kg every 4 hours, up to a maximum of 30 g daily for 5 days, which is associated with frequent side effects, including lightheadedness, nausea, vomiting, and systemic hypotension. A half dose of 50 mg/kg reduced the incidence of dizziness and hypotension without adversely affecting the reduced rate of recurrent hemorrhage, but it did not lower the incidence of nausea and vomiting (54). In a randomized comparison of aminocaproic acid (50 mg/kg every 4 hours for 5 days) with oral prednisone (40 mg daily), the rebleed rate was 7.1% in each group (27). Another reported complication is elevated IOP associated with the accelerated clot dissolution (55). Use of topical aminocaproic acid has been found to be a safe and effective alternative to systemic administration to prevent secondary hemorrhage. The use of the topical formulation (30% aminocaproic acid in 2% carboxypolymethylene gel) was associated with no systemic side effects (56).
The influence of hydrostatic pressure on the damaged vessels has also been studied, and one report described fewer rebleeds with medical reduction of systemic blood pressure and elevation of the head of the bed (57). The use of aspirin may increase the chances of recurrent hemorrhage (9,14), and therefore, any drug that may increase the risk of bleeding should be avoided for the first week after the trauma or until the hyphema has cleared completely.
Management of Associated IOP Elevation
Medical treatment of elevated IOP is occasionally needed to protect the optic nerve head and enhance the resorption of the hyphema. IOP reduction is best accomplished with use of an aqueous suppressant, such as a topical β-blocker or carbonic anhydrase inhibitor. However, caution should be exercised with the use of carbonic anhydrase inhibitors in patients with sickle cell hemoglobinopathies, because use of these agents increases the concentration of ascorbic acid in the aqueous humor, leading to more sickling in the anterior chamber (58). In the management of patients with sickle cell trait, control of the IOP during the first 24 hours was associated with a good prognosis, and lack of control in that period was associated with continued difficulty in managing the pressure (59). Hyperbaric oxygen therapy can significantly reduce the percentage of sickled cells injected intracamerally in rabbits by raising the aqueous Po2, which may be of value in patients with sickle cell hyphema (60). In one study, using transcorneal oxygen therapy (humidified oxygen 1 to 3 L/min) dramatically reduced the IOP in patients with glaucoma due to sickle cell hyphema (61).
Surgical intervention becomes necessary when a sustained IOP elevation cannot be controlled medically and threatens to damage the optic nerve or is associated with corneal bloodstaining. The critical pressure level depends on the status of the optic nerve head (if this is known): Healthy discs usually tolerate pressures of 40 to 50 mm Hg for 5 or 6 days, but an optic nerve head with preexisting glaucomatous optic atrophy may experience further damage at pressures less than 30 mm Hg within 24 to 48 hours. A total hyphema for more than 4 days is an additional indication for surgical intervention. Special attention must be given to patients with sickle cell anemia or trait because their optic nerve heads are especially vulnerable to damage at minimal to moderate elevations in IOP. A pressure in the mid-20s (mm Hg) for more than 1 day may be an indication to surgically intervene in these patients (59).
The surgical approach most often used is evacuation of the hyphema, which usually includes clotted blood, from the anterior chamber. It is also possible to remove the liquefied portion of the hyphema by gently irrigating the anterior chamber through a paracentesis wound and allowing the residual clot to resorb (62). This technique is of particular value when a sudden increase in pressure requires emergency measures to avoid irreversible loss of vision, as may occur with sickle cell disease (63). A corneal transfixing needle has been developed for simultaneously irrigating the anterior chamber and evacuating a fluid hyphema (64).
Many surgeons prefer to also remove the clot, and fibrinolytic agents such as urokinase and fibrinolysin have been used to facilitate clot lysis and irrigation (65,66). Other reported surgical techniques to remove the clot include cryoextraction, ultrasonic emulsification and extraction, and removal with vitrectomy instruments (67–70). Viscoelastic agents, such as sodium hyaluronate, have also been used to mechanically dissect a clot from the iris and express it through a corneoscleral incision (71,72). The fourth day after injury is said to be the optimum time for removal of the clot because it has usually retracted from the adjacent structures by then (73,74).
Other surgeons have advocated a trabeculectomy and iridectomy combined with gentle irrigation of the anterior chamber (75,76). During any surgical attempt to evacuate the hyphema, the iris may prolapse into the incision because of a pupillary block, necessitating an iridectomy (77). Complete resorption of the hyphema may follow iridectomy alone (78). When recurrent bleeding occurs during clot extraction, raising the IOP to 50 mm Hg for 5 minutes has been used to stop the bleeding (70).
Penetrating Injuries
Intraocular hemorrhage is also frequently associated with penetrating injuries, although associated glaucoma is less common than with blunt trauma in the early postinjury period because of the open wound. However, IOP elevation may follow closure of the wound, especially if meticulous care is not given to reconstruction of the anterior chamber and treatment of the associated inflammation in the early postoperative period (79).
Hyphemas Associated with Intraocular Surgery
Bleeding in the eye can be a serious complication of any intraocular procedure and may occur during the operation or in the early or even late postoperative period.
During Surgery
As an intraoperative complication, bleeding is usually associated with damage to the ciliary body, as can occur when a filtering procedure or iridectomy is performed. Intraoperative bleeding can usually be controlled by placing a large air bubble or viscoelastic agent in the anterior chamber for a few minutes, which raises the IOP and acts as a tamponade. Applying direct, gentle pressure with the tip of a sponge or Gel-Foam, or applying epinephrine (1:1000), to the ciliary body for 1 to 2 minutes can also help stop ciliary body bleeding. Cautery is generally avoided in these cases, although use of an intraocular, bipolar unit may be effective.
After Surgery
Bleeding in the early postoperative period is usually not associated with serious sequelae and should be managed conservatively with limited activity and elevation of the head. Small hyphemas after intraocular surgery normally clear rapidly, although the time may be considerably longer in eyes with preexisting glaucoma because of delayed passage of red blood cells through the trabecular meshwork. When a postoperative hyphema is associated with elevated IOP or excessive fibrin, conservative medical management should be instituted as required, by using drugs that lower aqueous production or hyperosmotics if necessary. Frequent topical steroid use can assist in clearing fibrin, and if this is unsuccessful, then intracameral use of tissue plasminogen activator (6.25 µg or 12.5 µg) can be helpful (80). Surgical intervention is reserved for critical cases, although the indications may be somewhat more liberal than with a traumatic hyphema if there is danger of rupturing a corneoscleral wound or causing further atrophy to an optic nerve that has previously been damaged by glaucoma.
Hemorrhage in the late postoperative period may result from the reopening of a uveal wound or from disruption of new vessels growing across a corneoscleral incision (81). In a study of 58 eyes 5 to 10 years after cataract extraction, 12% had vessels in the inner aspects of the incision site and nearly one half of these had evidence of mild intraocular hemorrhage (82). Direct argon laser therapy may be used to treat such vessels when they can be visualized gonioscopically (81), and use of transscleral Nd:YAG laser or diode photocoagulation may be effective if direct argon laser therapy is unsuccessful (83). Fortunately, postoperative hyphema is far less common because of the introduction of small incision and clear cornea cataract surgery.
Spontaneous Hyphemas
Hyphemas may also develop spontaneously in various conditions, most of which are considered in other chapters. In some cases, the hyphema may cause or contribute to an increase in the IOP.
Intraocular Tumors
A spontaneous hyphema may occur in a child with juvenile xanthogranuloma or retinoblastoma, and intraocular hemorrhage may be a manifestation of an ocular malignant melanoma or other intraocular neoplasm (see Chapter 21).
Neovascularization
New blood vessels in the anterior ocular segment, which may lead to a spontaneous hyphema, are seen in neovascular glaucoma (discussed in Chapter 19), Fuchs heterochromic cyclitis (Chapter 22), and other chronic uveitides.
Vascular Tufts at the Pupillary Margin
Vascular tufts at the pupillary margin, also called neovascular tufts or iris microhemangiomas, represent yet another source of spontaneous hyphema. Slitlamp biomicroscopy may reveal multiple vascular tufts along the pupillary margin, and fluorescein angiography of the iris has revealed small areas of staining and leakage from the lesions (84). One histopathologic study revealed thin-walled new vessels at the pupillary margin of the iris with a mild inflammatory cell infiltration (85), and another report described the vascular abnormality as a hamartoma of the capillary hemangioma type (86). Although more common in older adults, this condition occurs in adults of all ages. Most patients have no systemic disease, although associations with diabetes mellitus and myotonic dystrophy have been reported (84,87,88). Spontaneous hyphemas occur in a few of these cases, occasionally causing transient IOP elevation (89,90). Laser photocoagulation has been reported to successfully eradicate bleeding vascular tufts (85,91). However, because having recurrent hyphemas or permanent damage related to the transiently elevated IOP is rare, it is best to withhold treatment until one or more recurrences of bleeding are documented.
Dilatation and Posterior Synechiae
Spontaneous hyphemas may result in individuals with posterior synechiae in whom dilation drops are used. As the iris pulls away from the synechiae, hemorrhage may occur (Fig. 24.3).
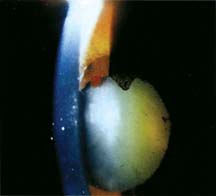
Figure 24.3 Spontaneous hyphema in a patient with posterior synechiae whose eye has been dilated. Note the hemorrhage at the 12-o’clock position of the pupil.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


