Glaucomas Associated with Disorders of the Retina, Vitreous, and Choroid
Several types of glaucoma are associated with diseases of the retina. The most common of these is neovascular glaucoma, which is usually associated with one of several retinal disorders, although some cases are associated with other ocular or extraocular conditions. Retinal detachments and a variety of less common disorders of the retina, vitreous, or choroid may cause or occur in association with various forms of glaucoma.
NEOVASCULAR GLAUCOMA
Terminology
In 1906, Coats (1) described new vessel formation on the iris in eyes with central retinal vein occlusion. This neovascularization of the iris is commonly known as rubeosis iridis and is now recognized as a complication of many diseases of the retina and other ocular and extraocular disorders. Rubeosis iridis is frequently associated with a severe form of glaucoma, which has been given different names on the basis of various clinical features: hemorrhagic glaucoma, referring to the hyphema that is present in some cases; congestive glaucoma, describing the frequently acute nature of the condition; and thrombotic glaucoma, implying an underlying vascular thrombotic cause. However, none of these terms accurately describes the glaucoma in all cases, and more nonspecific names are preferable, such as rubeotic glaucoma or neovascular glaucoma, which was proposed by Weiss and colleagues and is the term found most often in the current literature (2,3).
Factors Predisposing to Rubeosis Iridis
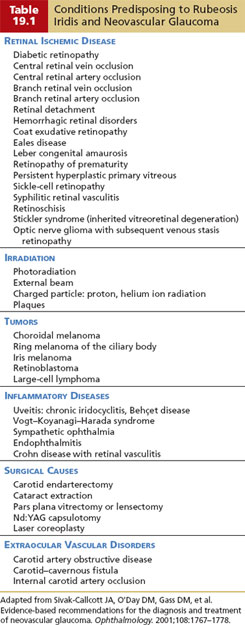
Most cases of rubeosis iridis are preceded by hypoxic disease of the retina. Diabetic retinopathy, central retinal vein occlusion, and carotid ischemic disease are the most common causes (4). However, many additional retinal diseases and certain other ocular or extraocular disorders have been recognized, resulting in a long list of conditions that may predispose to rubeosis iridis (Table 19.1).
Diabetic Retinopathy
Approximately one third of patients with rubeosis iridis have diabetic retinopathy. Tight metabolic control of blood glucose results in delayed onset of diabetic retinopathy and slows or prevents progression to nonproliferative and proliferative retinopathy (5). The frequency with which rubeosis iridis is associated with diabetic retinopathy is greatly influenced by surgical interventions. After pars plana vitrectomy for diabetic retinopathy, the reported incidence of rubeosis iridis ranges from 25% to 42%, whereas that for neovascular glaucoma ranges from 10% to 23% (6–8), with most of these cases developing during the first 6 months after surgery (9). In these cases, rubeosis iridis and neovascular glaucoma occur more often in aphakic eyes (7,8,10). In one series, vitreous cavity lavage of hemorrhage after pars plana vitrectomy for diabetic retinopathy was associated with rubeosis iridis in 76% of aphakic eyes and 14% of phakic eyes (10). Postoperative neovascular glaucoma is also more common when rubeosis iridis is present before vitrectomy (11).
An unrepaired retinal detachment after vitrectomy for diabetic retinopathy is also a risk factor for postoperative rubeosis iridis. The acute onset or exacerbation of rubeosis iridis after diabetic vitrectomy can indicate the presence of a peripheral traction retinal detachment (12). Successful surgical reattachment of the retina during vitrectomy for diabetic retinopathy often leads to regression of preoperative rubeosis iridis, especially in phakic patients (13). A completely attached retina and aggressive anterior or peripheral photocoagulation therapy are the most important factors in controlling or preventing neovascular glaucoma after vitrectomy for proliferative diabetic retinopathy (12,14). Intraocular silicone oil also reduces the incidence of anterior segment neovascularization, possibly by acting as a diffusion or convection barrier to the posterior movement of oxygen from the anterior chamber or the anterior movement of an angiogenesis factor (15).
Nonproliferative and preproliferative diabetic retinopathy may progress after cataract surgery (4). Intracapsular cataract surgery alone in eyes with diabetic retinopathy has been associated with an increased incidence of postoperative rubeosis iridis and neovascular glaucoma. The incidence is similar with extracapsular extraction and a primary capsulotomy. Leaving the posterior capsule intact appears to reduce the likelihood of this complication, although a subsequent laser capsulotomy in patients with diabetes may lead to neovascular glaucoma.
Retinal Vascular Occlusive Disorders
Central retinal vein occlusion accounted for 28% of all cases of rubeosis iridis in one series (16). Elevated intraocular pressure (IOP), with or without glaucomatous damage, is thought by most (17–19) investigators to be a predisposing factor for retinal vein occlusion. Optic disc cupping was reported to be a significant risk factor for central and branch retinal vein occlusions in the Beaver Dam Eye Study (20). Other risk factors for central or branch retinal vein occlusion include systemic hypertension, diabetes, and male sex (19). Retinal vein occlusion may occur in a wide range of ages (14 to 92 years in one large study, although 51% of these patients were 65 years or older) (21).
Rubeosis iridis and neovascular glaucoma are associated with central retinal artery occlusion, and they are less commonly associated with central vein occlusion. In two series of patients with central retinal artery occlusion, the incidence of rubeosis iridis was 16.67% and 18.2%, respectively (22,23). Patients who develop neovascular glaucoma in association with central retinal artery occlusion are usually elderly with severe carotid artery disease and atherosclerosis, which may be predisposing factors for retinal artery occlusion and, in some cases, ocular neovascularization (24,25). Branch retinal vein occlusion may rarely cause rubeosis iridis and neovascular glaucoma (16). Branch retinal artery occlusion has also been reported as a rare cause of rubeosis iridis (25,26), although the association with neovascular glaucoma is uncertain.
Other Retinal Disorders
Rubeosis iridis may be associated with a rhegmatogenous retinal detachment (27), especially when complicated by proliferative vitreoretinopathy (28). In some cases, the detachment may overlie a choroidal melanoma. A chronic retinal detachment with associated glaucoma should always raise the suspicion of melanoma. Neovascular glaucoma may also be associated with sickle-cell retinopathy and many other retinal disorders, which are listed in Table 19.1.
Other Ocular Disorders
Uveitis was present in 11% of rubeotic eyes in one series and in 1.5% of another study (16,29). An iris melanoma has also been associated with neovascular glaucoma, which resolved after the tumor was excised (30). End-stage glaucoma (open-angle or angle-closure) has been said to give rise to rubeosis iridis (16), which may be related to associated central retinal vein occlusion.
Extraocular Vascular Disorders
Carotid artery obstructive disease is probably the third most common cause of neovascular glaucoma, accounting for 13% of all cases in one series (29). These eyes may initially be normotensive or even hypotensive as a result of decreased perfusion of the ciliary body with reduced aqueous production, and fluorescein angiography may reveal an increased arm-to-retina time and leakage from the major retinal arterioles. A carotid–cavernous fistula may also cause rubeosis iridis and neovascular glaucoma as a result of decreased arterial flow and subsequent reduction in the ocular perfusion pressure, which may occur before or after treatment of the fistula (31,32). Internal carotid artery occlusion may create an ophthalmic artery steal phenomenon with associated rubeosis iridis (33).
Theories of Neovasculogenesis
The mechanisms by which the aforementioned clinical situations lead to rubeosis iridis are not fully understood, although the following theories have been proposed (4).
Retinal Hypoxia
Because most of the conditions associated with rubeosis iridis involve diminished perfusion of the retina, retinal hypoxia may be one factor in the formation of new vessels on the iris and anterior chamber angle and on the retina and optic nerve head. This concept is supported by the clinical observation that rubeosis iridis in association with proliferative diabetic retinopathy or central retinal vein occlusion is more likely to occur when significant capillary nonperfusion is present.
Angiogenesis Factors
The existence of an angiogenic substance regulating normal development of retinal blood vessels was hypothesized in 1948 (34). It has since been demonstrated that tumors possess a diffusible factor, tumor angiogenesis factor, that can elicit new vessel growth toward the tumor (35). Subsequent studies have suggested that human and animal retinas and other vascular ocular tissues have similar angiogenic activity related to a key angiogenic peptide, vascular endothelial growth factor (VEGF), which explains why ocular neovascularization can occur in areas remote from the site of retinal capillary nonperfusion. Several cell types in the retina synthesize VEGF, but under conditions of retinal ischemia, Müller cells appear to be the primary source. Four VEGF isoforms (VEGF121, VEGF165, VEGF189, and VEGF206) have been identified, which are generated by alternative mRNA splicing from the same gene (36). VEGF165 is the most abundant form in the majority of tissues. VEGF is a potent angiogenic stimulator, promoting several steps of angiogenesis, including proliferation, migration, proteolytic activity, and capillary tube formation, thus playing a crucial role in both normal and pathologic angiogenesis. It is also known as a vascular permeability factor on the basis of its ability to induce vascular hyperpermeability and endothelial cell proliferation as well as migration.
Vasoinhibitory Factors
It has been postulated that ocular tissues may produce substances that inhibit neovascularization. The vitreous and lens are possible sources of these vasoinhibitory factors (37,38), which could explain why vitrectomy or lensectomy increases the risk for rubeosis iridis in eyes with diabetic retinopathy. Retinal pigment epithelial cells release an inhibitor of neovascularization (39).
Clinicopathologic Course
The clinical and histologic events that lead from a predisposing factor through rubeosis iridis to advanced neovascular glaucoma may be thought of in four stages (Fig. 19.1).
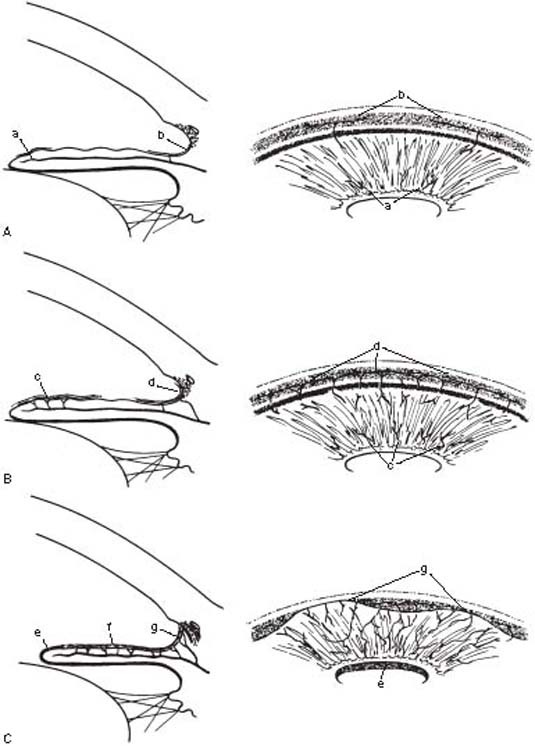
Figure 19.1 Clinicopathologic stages of neovascular glaucoma A: Preglaucoma stage (i.e., rubeosis iridis), characterized by new vessels on the surface of the iris (a) and in the anterior chamber angle (b). B: Open-angle glaucoma stage, characterized by an increase in neovascularization and a fibrovascular membrane on the iris (c) and in the anterior chamber angle (d). C: Angle-closure glaucoma stage, characterized by contracture of the fibrovascular membrane, causing corectopia, ectropion uvea (e), flattening of the iris (f ), and peripheral anterior synechiae (g).
Prerubeosis Stage
In patients with a predisposing factor, such as diabetic retinopathy or central retinal vein occlusion, it is helpful to understand what the likelihood is for development of rubeosis iridis and what the chances are for progression to neovascular glaucoma. Additional circumstances, especially with the two mentioned predisposing factors, may increase the risk for neovascular glaucoma to the extent that treatment may be indicated even before rubeosis is detected.
Diabetic Retinopathy
The prevalence of rubeosis iridis among patients with diabetes mellitus ranges from 0.25% to 20% according to various reports (40). Diabetes usually exists for many years before rubeosis develops, and concomitant proliferative diabetic retinopathy is usually found. In populations of patients with proliferative diabetic retinopathy, rubeosis iridis is reported to occur in approximately one-half of the cases (40,41). Rubeosis iridis may rarely occur in an eye with nonproliferative retinopathy (40), although other predisposing factors, such as carotid artery disease, should be considered in these cases.
The risk for rubeosis iridis and neovascular glaucoma in patients with diabetic retinopathy is greatly increased when arteriolar or capillary nonperfusion is present or after vitrectomy or lensectomy. There is also a highly significant correlation between rubeosis iridis and optic disc neovascularization (42) as well as a rhegmatogenous retinal detachment (13,14). The demonstration of peripupillary leakage by iris fluorescein angiography correlates with the presence of abnormal iris vessels and the risk for rubeosis iridis after vitrectomy for diabetic retinopathy (Fig. 19.2). Slitlamp biomicroscopy is less reliable than angiography in detecting the presence of diabetic iris lesions (43). It is important to pay close attention to the pupillary margin of the iris, where neovascularization is typically seen first (44), when looking for the earliest biomicroscopic evidence of anterior segment rubeosis. However, gonioscopy is also important, because angle neovascularization may occasionally precede that of the iris (45).
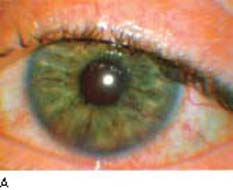
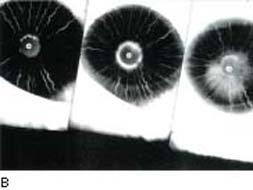
Figure 19.2 A: Slitlamp view of a diabetic patient with neovascular glaucoma. Neovascularization of the iris is seen through the edematous cornea that is secondary to uncontrolled IOP. B: Fluorescein angiographic view of another patient demonstrates fluorescein leakage along the superior papillary margin. In subsequent frames, the area of fluorescein leakage enlarges. (From Reiss GR, Sipperley JO. Glaucoma associated with retinal disorders and retinal surgery. In: Tasman W, Jaeger EA, eds. Duane’s Clinical Ophthalmology. Vol. 3. Philadelphia, PA: Lippincott Williams & Wilkins:chap 54E.)
Central Retinal Vein Occlusion
During the early months after a central retinal vein occlusion, hypotony may develop (46). The explanation for this is unclear, although the possible influences of anterior segment ischemia or an angiogenic factor have been proposed.
As in diabetic retinopathy, the incidence of rubeosis iridis and neovascular glaucoma in eyes with central retinal vein occlusion is significantly correlated with the extent of retinal capillary nonperfusion. In one study, the incidence of rubeosis iridis after central retinal vein occlusion was 60% when retinal ischemia was demonstrated by fluorescein angiography, compared with 1% in eyes with good capillary perfusion (47).
Fluorescein angiography is the most direct method of evaluating capillary nonperfusion but is not always feasible because of obstruction of visualization by blood or other media opacities. The ophthalmoscopic findings may be helpful in determining the risk for neovascular glaucoma, which has been reported in 14% to 27% of eyes with hemorrhagic retinopathy (complete venous occlusion) but in no cases of venous stasis retinopathy (incomplete occlusion) (48,49). Several other techniques have predictive value. Fluorescein angiography of the iris reveals abnormal, leaking vessels in virtually all eyes with extensive retinal capillary closure after central retinal vein occlusion (50). Aqueous protein and cell concentrations, as indicated by a laser flare–cell meter, have been shown to correlate with fluorescein angiographic findings and the severity of retinal vein occlusion (51). A relative afferent pupillary defect also indicates an increased risk for rubeosis iridis after central retinal vein occlusion (52), and infrared pupillometry is an objective method of documenting this finding (53). Electroretinography also has useful predictive value (54). The most diagnostic findings include a B-wave implicit time delay and a reduced B-wave–A-wave amplitude ratio. Flicker electroretinography also has diagnostic value (55). Blood-flow velocities of the central retinal vein and artery can be measured with color Doppler imaging and provide a high degree of predictability regarding the risk for iris neovascularization (56).
Despite evidence of good perfusion and a low risk for iris neovascularization by any of the noted techniques, all patients with central retinal vein occlusion should be followed for the possibility of rubeosis iridis and neovascular glaucoma. In some patients, perfused retinas will progress to nonperfusion. In one study, this was seen in 15% of cases (57). Time and age appear to influence this percentage. In one study, the cumulative probability of converting from nonischemic to ischemic central retinal vein occlusion in 6 and 18 months was 13.2% and 18.6%, respectively, in persons 65 years or older and 6.7% and 8.1%, respectively, in persons 45 to 64 years of age (21). The study also found that 83% of patients with indeterminate perfusion eventually developed nonperfusion or neovascularization of the iris or anterior chamber angle (58).
Preglaucoma Stage: Rubeosis Iridis
Clinical Features
The preglaucoma stage is characterized by a normal IOP, unless preexisting chronic open-angle glaucoma (COAG) is present. Slitlamp biomicroscopy early in the disease process typically reveals dilated tufts of preexisting capillaries and fine, randomly oriented vessels on the surface of the iris near the pupillary margin (Fig. 19.3). The new vessels are also characterized by leakage of fluorescein. Neovascularization in most cases is first seen on the peripupillary iris, although it may be first seen in the anterior chamber angle in patients with diabetes and central retinal vein occlusion (45,59). Gonioscopy therefore may reveal a normal anterior chamber angle or may show a variable amount of angle neovascularization. The latter is characterized by single vascular trunks crossing the ciliary body band and scleral spur and arborizing on the trabecular meshwork.
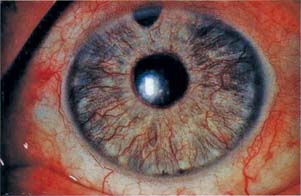
Figure 19.3 Slitlamp view of iris in a patient with rubeosis iridis shows tortuous vessels on the surface of the iris.
Histopathologic Features
The rubeosis iridis begins intrastromally and then develops on the surface of the iris (37,60). Experimental retinal vein occlusion in monkey eyes indicates that the rubeosis iridis begins with dilatation of normal iris vessels and marked increase in metabolism of vascular endothelial cells followed by new vessel formation (61). Silicone-injection studies indicate that the new vessels on the iris arise from normal iris arteries and drain primarily into iris and ciliary body veins, whereas new vessels in the angle arise from arteries of the iris and ciliary body and connect with the peripheral neovascular network on the iris (62). Although the clinical appearance of rubeosis iridis is said to be the same in cases of diabetes and central retinal vein occlusion, the silicone injections show tighter and more evenly distributed neovascularization in the diabetic eye (62). The silicone-injection studies also show that new vessels in the angle run circumferentially in the trabecular meshwork, with branches coursing into the fibrosed Schlemm canal and occasionally into collector channels (62). The new vessels are characterized histologically as having thin fenestrated walls and are arranged in irregular patterns (60). The ultrastructure of iris neovascularization associated with sickle-cell retinopathy is said to be similar to that in diabetes and retinal occlusive disease with open interendothelial cell junctions, attenuated intraendothelial cytoplasm, and pericyte formation (63).
Open-Angle Glaucoma Stage
Clinical Features
Neovascular glaucoma does not invariably follow the development of rubeosis iridis (40,41,63,64), and the latter condition may rarely resolve spontaneously, especially that associated with diabetic retinopathy (40). The reported incidence of neovascular glaucoma in diabetic patients with rubeosis iridis ranges from 13% to 41% (40,41,64), whereas that associated with central retinal vein occlusion is probably significantly higher. The latter condition typically occurs 8 to 15 weeks after the vascular occlusive event (63). It has been called 90-day glaucoma because the average time interval was thought to be 3 months. However, the glaucoma can develop during the first month or any time after a central retinal vein occlusion.
The rubeosis iridis is typically more florid in this stage, and biomicroscopic examination of the aqueous often reveals an inflammatory reaction and sometimes a hyphema (Fig. 19.4). By gonioscopy, the anterior chamber angle is still open, but the neovascularization may be intense (Fig. 19.5). The IOP is elevated and may rise suddenly, causing the patient to present with acute-onset glaucoma.
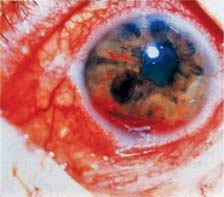
Figure 19.4 Slitlamp appearance of the iris in a patient with neovascular glaucoma shows marked rubeosis and hyphema.
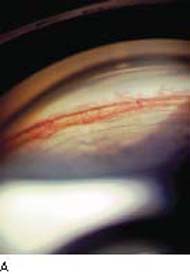

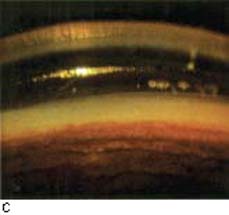
Figure 19.5 A: Angle neovascularization in a patient with a central retinal vein occlusion. Note vessels are superficial and found on the ciliary body band and trabecular meshwork. The angle is open although aqueous outflow is impaired. B: Neovascular glaucoma has progressed to angle closure in this patient. C: A patient with open-angle neovascular glaucoma, with heavy neovascularization of the open angle. The angle, however, is beginning to close, as seen by the low synechia to the left of the view. (A, B, courtesy of Joseph A. Halabis, OD.)
Histopathologic Features
The hallmark of the open-angle glaucoma stage is a fibrovascular membrane that covers the anterior chamber angle and anterior surface of the iris and may even extend onto the posterior iris (60,65). Chronic inflammatory changes are also typically seen on histologic examination (60,65). The glaucoma in this stage probably results from obstruction of the trabecular meshwork by the fibrovascular membrane, with variable contribution from the inflammation and hemorrhage. One histopathologic report of an eye with neovascular glaucoma and without a fibrovascular membrane covering the iridocorneal angle found that the spaces between the trabecular beams were lined by a single layer of vascular endothelium and were filled with red blood cells in this patient, suggesting that neovascular tissue found in the trabecular spaces might be one of the factors responsible for IOP elevation in eyes with neovascular glaucoma (66).
Angle-Closure Glaucoma Stage
Clinical Features
In the angle-closure glaucoma stage, the stroma of the iris has become flattened, with a smooth, glistening appearance. Ectropion uvea is frequently present, and the iris is often dilated and pulled anteriorly from the lens (Fig. 19.6). In the anterior chamber angle, the contracture leads to formation of peripheral anterior synechia, with eventual total synechial closure of the angle. The glaucoma in this stage is typically severe and usually requires surgical intervention.
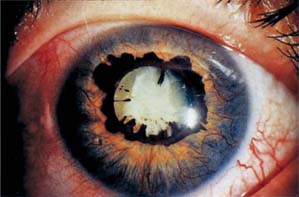
Figure 19.6 Slitlamp view of a patient with the angle-closure stage of neovascular glaucoma shows numerous new vessels on the iris, with pupillary dilatation and ectropion uvea due to contracture of the fibrovascular membrane.
Histopathologic Features
The clinically observed alterations of the iris and anterior chamber angle in this stage result from contracture of tissue overlying these structures. Histopathologic studies reveal peripheral anterior synechiae and flattening of the anterior iris surface by a confluent fibrovascular membrane (67,68). Overlying the new vessels is a clinically inapparent, superficial layer of myofibroblasts (i.e., fibroblastic cells with smooth-muscle differentiation), which may be responsible for the tissue contraction (67). A layer of endothelium, continuous with the corneal endothelium at the pseudoangle, is also seen in some cases and has been observed to possess features of myoblastic differentiation (68,69), which may explain the origin of these cells.
Differential Diagnosis
In the open-angle stage, neovascular glaucoma must be distinguished from other glaucomas with acute onset, such as angle-closure glaucoma and glaucoma associated with anterior uveitis. This differentiation can usually be made on the basis of new vessels on the iris and in the anterior chamber angle with neovascular glaucoma, although eyes with uveitis often have dilatation of normal iris vessels that may be confused with neovascularization, especially with blue irides. Patients with Fuchs heterochromic iridocyclitis also typically have new vessels in the anterior chamber angle (see Chapter 22). In the angle-closure stage of neovascular glaucoma, the new vessels may be less apparent, and the differential diagnosis must include other causes of iris distortion and peripheral anterior synechiae, such as the iridocorneal endothelial syndrome (see Chapter 16) and old trauma (see Chapter 25).
Management
Panretinal Photocoagulation
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


